Académique Documents
Professionnel Documents
Culture Documents
Cation Anion Worksheet
Transféré par
akbaby514100%(2)100% ont trouvé ce document utile (2 votes)
4K vues1 pageAtoms form ions by gaining or losing electrons to achieve an octet of 8 valence electrons. Atoms with fewer than 4 valence electrons tend to lose electrons and become cations with a positive charge, while atoms with more than 4 valence electrons tend to gain electrons and become anions with a negative charge. The document provides examples of common elements writing out equations showing how many electrons are gained or lost to form cations or anions.
Description originale:
practice sheet for general chemistry
Copyright
© © All Rights Reserved
Formats disponibles
DOC, PDF, TXT ou lisez en ligne sur Scribd
Partager ce document
Partager ou intégrer le document
Avez-vous trouvé ce document utile ?
Ce contenu est-il inapproprié ?
Signaler ce documentAtoms form ions by gaining or losing electrons to achieve an octet of 8 valence electrons. Atoms with fewer than 4 valence electrons tend to lose electrons and become cations with a positive charge, while atoms with more than 4 valence electrons tend to gain electrons and become anions with a negative charge. The document provides examples of common elements writing out equations showing how many electrons are gained or lost to form cations or anions.
Droits d'auteur :
© All Rights Reserved
Formats disponibles
Téléchargez comme DOC, PDF, TXT ou lisez en ligne sur Scribd
100%(2)100% ont trouvé ce document utile (2 votes)
4K vues1 pageCation Anion Worksheet
Transféré par
akbaby514Atoms form ions by gaining or losing electrons to achieve an octet of 8 valence electrons. Atoms with fewer than 4 valence electrons tend to lose electrons and become cations with a positive charge, while atoms with more than 4 valence electrons tend to gain electrons and become anions with a negative charge. The document provides examples of common elements writing out equations showing how many electrons are gained or lost to form cations or anions.
Droits d'auteur :
© All Rights Reserved
Formats disponibles
Téléchargez comme DOC, PDF, TXT ou lisez en ligne sur Scribd
Vous êtes sur la page 1sur 1
Name:_________________________Period:______
Neutral Atoms Forming Ions
Atoms never have more than 8 valence electrons. Atoms with fewer than 4 valence electrons tend to lose these
electrons to become cations (positive). Elements with greater than 4 valence electrons tend to gain more
electrons for a total of eight becoming anions (negative).
Below write out equations showing whether an element gains or loses electron.
electrons are gained or lost. Indicate if the final ion is a cation or anion.
Be sure to indicate how many
Type of Ion
1.
1.
loses 1e-
K+
Cation
2.
gains 2e-
O2-
Anion
3.
Mg
4.
Al
5.
6.
7.
Na
8.
9.
Ba
10.
Br
11.
12.
Ra
13.
Ca
14.
Cl
15.
Sr
16.
anions metals or nonmetals?
2. Are cations metals or nonmetals?
Are
Vous aimerez peut-être aussi
- Electronegativity WorksheetDocument2 pagesElectronegativity WorksheetDn Angel100% (1)
- Activity 3 Valence ElectronsDocument2 pagesActivity 3 Valence ElectronsJan IcePas encore d'évaluation
- Isotope PracticeDocument2 pagesIsotope Practiceliza1207Pas encore d'évaluation
- 6-16a-Names of Ionic Compounds Wkst-KeyDocument1 page6-16a-Names of Ionic Compounds Wkst-KeyAdin Añasco100% (1)
- Worksheet - Orbital Diagrams - TeacherDocument2 pagesWorksheet - Orbital Diagrams - Teacherdela2100% (3)
- Part 2. Activity-Worksheets in Physical ScienceDocument2 pagesPart 2. Activity-Worksheets in Physical Scienceangelo llenes100% (1)
- Types of Chemical Reaction WorksheetDocument2 pagesTypes of Chemical Reaction WorksheetKenneth Myro GarciaPas encore d'évaluation
- Table of SpecificationDocument5 pagesTable of SpecificationRustan LacanilaoPas encore d'évaluation
- Bond Polarity and ElectronegativityDocument2 pagesBond Polarity and ElectronegativityJewel Mae MercadoPas encore d'évaluation
- Summative Assessment (Unit)Document5 pagesSummative Assessment (Unit)Ernesto Berger MarinheiroPas encore d'évaluation
- Atoms Periodic Table Pop QuizDocument2 pagesAtoms Periodic Table Pop QuizAlrianne BatonghinogPas encore d'évaluation
- Multiple Choice. Read Each Question Carefully and Write The Letter of The Correct Answer On The SpaceDocument5 pagesMultiple Choice. Read Each Question Carefully and Write The Letter of The Correct Answer On The SpaceLouie Jane EleccionPas encore d'évaluation
- Summative 1Document3 pagesSummative 1Shine ColterPas encore d'évaluation
- Chem 4.1 Worksheet-R-Atoms, Ions, Isotopes-AnsDocument2 pagesChem 4.1 Worksheet-R-Atoms, Ions, Isotopes-AnsAhmad Johns0% (1)
- Villar - Lesson Plan - Carboxylic Acids, Esters, and Other Acid DerivativesDocument5 pagesVillar - Lesson Plan - Carboxylic Acids, Esters, and Other Acid DerivativesClaire Villar0% (1)
- Protons, Neutrons, and Electrons Practice Worksheet For 8th GradeDocument2 pagesProtons, Neutrons, and Electrons Practice Worksheet For 8th GradeDrama Music67% (3)
- A Lesson Plan in Chemical BondingDocument5 pagesA Lesson Plan in Chemical BondingEllen Grace Dela PeñaPas encore d'évaluation
- General Chemistry 2 Q3 Module 1Document11 pagesGeneral Chemistry 2 Q3 Module 1Jennylyn CariagaPas encore d'évaluation
- Activity 4 - Worksheet On Formation of IonsDocument2 pagesActivity 4 - Worksheet On Formation of IonsJohn Lesther PabiloniaPas encore d'évaluation
- 3rd Periodical Test - Science 8Document10 pages3rd Periodical Test - Science 8Christine Joy Millares GimenoPas encore d'évaluation
- Lesson Plan Ionic Bonding - LessonDocument6 pagesLesson Plan Ionic Bonding - LessonCristina Nicomedes AguinaldoPas encore d'évaluation
- Greenhouse Effect QuizDocument3 pagesGreenhouse Effect QuizSean CampbellPas encore d'évaluation
- Atomic Model WorksheetDocument2 pagesAtomic Model WorksheetTelle Telle100% (1)
- Lesson Plan - Periodic TableDocument3 pagesLesson Plan - Periodic Tableasdfg sdmflsdflsPas encore d'évaluation
- Activity Worksheet On Metallic BondingDocument2 pagesActivity Worksheet On Metallic BondingFrancisca CatacutanPas encore d'évaluation
- Projectile Motion Problem Worksheet: Answer KeyDocument5 pagesProjectile Motion Problem Worksheet: Answer Keyjayrald cruzadaPas encore d'évaluation
- Science 7: First Quarter ExaminationDocument2 pagesScience 7: First Quarter Examinationmacren septemberPas encore d'évaluation
- Unit Learning Plan Science SampleDocument7 pagesUnit Learning Plan Science Sampleshine davidPas encore d'évaluation
- Worksheet-Accuracy and Precision-FinalDocument4 pagesWorksheet-Accuracy and Precision-FinalSabeeh Ul HassanPas encore d'évaluation
- Chemical Bonding ActivitiesDocument12 pagesChemical Bonding Activitiesjoan marie PeliasPas encore d'évaluation
- ANSWERS - Review The AtomDocument5 pagesANSWERS - Review The AtomDayana MoreiraPas encore d'évaluation
- OBTL Chemistry I G12 STEMDocument8 pagesOBTL Chemistry I G12 STEMCelestial Lacambra50% (2)
- Chemistry 1 Performance Task 3: M A N D A L U Y O N GDocument7 pagesChemistry 1 Performance Task 3: M A N D A L U Y O N GAngelo James S. DEJANPas encore d'évaluation
- DLP 7 E's Particle Nature of Matter Group 3a Rtot 2018Document6 pagesDLP 7 E's Particle Nature of Matter Group 3a Rtot 2018KATHRYN CENTINALESPas encore d'évaluation
- Summative Assessment 1 2nd QuarterDocument2 pagesSummative Assessment 1 2nd QuarterJudith DurensPas encore d'évaluation
- D. Isotopes WorksheetDocument2 pagesD. Isotopes WorksheetKarissa100% (1)
- Periodic Table QuizDocument3 pagesPeriodic Table QuizNeptune LopezPas encore d'évaluation
- Physical Science (TM) - PRDocument46 pagesPhysical Science (TM) - PRMary Vi D. Dela CruzPas encore d'évaluation
- The Magnetic Property of An Atom and Atoms Atomic OrbitalsDocument12 pagesThe Magnetic Property of An Atom and Atoms Atomic OrbitalsJanne Lorraine Garcia-EleazarPas encore d'évaluation
- Validated ADM GENERAL CHEMISTRY 1 MODULE 1 3 FIRST QUARTERDocument28 pagesValidated ADM GENERAL CHEMISTRY 1 MODULE 1 3 FIRST QUARTERJun Gabriel Bornales100% (1)
- Formation of IonsDocument2 pagesFormation of Ionserica schoerner100% (1)
- Grade 7 - Lc2 Research I: Sdo Laguna Ste - P WorksheetDocument4 pagesGrade 7 - Lc2 Research I: Sdo Laguna Ste - P WorksheetAnnRubyAlcaideBlandoPas encore d'évaluation
- Atom Quiz PracticeDocument4 pagesAtom Quiz PracticeRejPas encore d'évaluation
- Science 7 Q2 Lesson 2 Biological OrderDocument2 pagesScience 7 Q2 Lesson 2 Biological OrderArianne Jane Mae ManPas encore d'évaluation
- MODULE-1-Gen-Chem-1B-Performance Task PDFDocument1 pageMODULE-1-Gen-Chem-1B-Performance Task PDFJam AicaPas encore d'évaluation
- CarbonylDocument4 pagesCarbonylJulie B. BongcalesPas encore d'évaluation
- DLL Science Grade 8 Week 1Document2 pagesDLL Science Grade 8 Week 1Aika Unica Gabriel100% (1)
- Republic of The Philippines Department of Education Region XiDocument3 pagesRepublic of The Philippines Department of Education Region XiArgie Joy Marie AmpolPas encore d'évaluation
- Worksheets LS2 Layers of The EarthDocument7 pagesWorksheets LS2 Layers of The EarthAngeline Panaligan Ansela100% (1)
- Lesson Plan For Laws of MotionDocument3 pagesLesson Plan For Laws of MotionBarry Landanganon100% (1)
- Reading Comprehension PPT Class VIIDocument4 pagesReading Comprehension PPT Class VIIsadaf hashmiPas encore d'évaluation
- Latitude and Longitude Worksheet WorldDocument8 pagesLatitude and Longitude Worksheet WorldBen Rogaczewski0% (1)
- Balancing Chemical EquationsDocument4 pagesBalancing Chemical EquationsAnonymous AMmNGgPPas encore d'évaluation
- Types of Chemical Reaction Worksheet Practice AnswersDocument1 pageTypes of Chemical Reaction Worksheet Practice AnswersKomalpreet Jeor100% (1)
- Semi-Detailed Daily Lesson Plan in Science 7: Transforming The Learners To BecomeDocument3 pagesSemi-Detailed Daily Lesson Plan in Science 7: Transforming The Learners To BecomeAntonette AlbinaPas encore d'évaluation
- Cation Anion WorksheetDocument1 pageCation Anion WorksheetAryan AgrawalPas encore d'évaluation
- Chemical Bonding Reading MaterialDocument6 pagesChemical Bonding Reading MaterialJohann Carlo C. AldecoaPas encore d'évaluation
- Ions and Bonds PowerpointDocument26 pagesIons and Bonds Powerpointapi-2622192730% (1)
- Valence Electrons, Ions, and The Periodic Table12789Document34 pagesValence Electrons, Ions, and The Periodic Table12789Janelyn GarinPas encore d'évaluation
- Formationn of IonsDocument4 pagesFormationn of IonsMelgeri Aubrey E. UngosPas encore d'évaluation
- Raman Spectroscopy of Two-Dimensional MaterialsDocument270 pagesRaman Spectroscopy of Two-Dimensional MaterialsYahsé Rojas ChallaPas encore d'évaluation
- Atomic Structure & Energy Levels in An AtomDocument15 pagesAtomic Structure & Energy Levels in An AtomhyebibiePas encore d'évaluation
- Helium Atom, Approximate Methods: 22nd April 2008Document17 pagesHelium Atom, Approximate Methods: 22nd April 2008Julian David Henao EscobarPas encore d'évaluation
- Solution Manual For General Organic and Biological Chemistry 2Nd Edition by Janice Gorzynski Smith Isbn 0073402788 9780073402789 Full Chapter PDFDocument36 pagesSolution Manual For General Organic and Biological Chemistry 2Nd Edition by Janice Gorzynski Smith Isbn 0073402788 9780073402789 Full Chapter PDFsteven.biggs159100% (10)
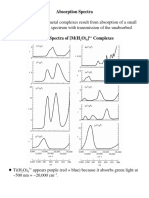
- Ch11 1 Transition Metals Absorption SpectraDocument29 pagesCh11 1 Transition Metals Absorption SpectraTapas GhatakPas encore d'évaluation
- Naming Chemical Compounds: Rules and TipsDocument5 pagesNaming Chemical Compounds: Rules and TipsNoah G.Pas encore d'évaluation
- Gr9 OSSD Chemistry RevisionDocument4 pagesGr9 OSSD Chemistry RevisionocPas encore d'évaluation
- Shimon Sir - AtomsDocument61 pagesShimon Sir - AtomsVenus GirlPas encore d'évaluation
- Yakeen - 2.O Atomic Structure: Xy X yDocument3 pagesYakeen - 2.O Atomic Structure: Xy X yM.Tharun KumarPas encore d'évaluation
- Bonding HL NotesDocument8 pagesBonding HL NotesDhairya GandhiPas encore d'évaluation
- Bohr's Model: Nucleus Electron OrbitDocument5 pagesBohr's Model: Nucleus Electron OrbitCRAZY FOOD LOVERSPas encore d'évaluation
- Electromagnetic Radiation: Visible LightDocument39 pagesElectromagnetic Radiation: Visible LightgeethaPas encore d'évaluation
- General Chemistry I Quarter 2 ModulesDocument91 pagesGeneral Chemistry I Quarter 2 Modulesarnold vallePas encore d'évaluation
- Fermi Surface IIIDocument67 pagesFermi Surface IIIImtiazAhmedPas encore d'évaluation
- MS ChapterDocument80 pagesMS ChapterTarik ChimiePas encore d'évaluation
- EXPERIMENT 3: AAS Determination of Calcium in Commercial Supplement TabletsDocument11 pagesEXPERIMENT 3: AAS Determination of Calcium in Commercial Supplement Tabletsdjambulaziz100% (3)
- SBC 2Document3 pagesSBC 2Erehh JeagerPas encore d'évaluation
- 12.8 Bab 1 - 2 Ting. 4Document10 pages12.8 Bab 1 - 2 Ting. 4izrulPas encore d'évaluation
- Atomic Structure History 1213Document3 pagesAtomic Structure History 1213Milena ŽivkovićPas encore d'évaluation
- Classification of Elements and Periodicity in Properties of ElementsDocument13 pagesClassification of Elements and Periodicity in Properties of ElementsAbhayPas encore d'évaluation
- Class IX QB Chemistry Final Exam 17 18Document5 pagesClass IX QB Chemistry Final Exam 17 18Bosky MendhirattaPas encore d'évaluation
- QQ Highly Efficient Plasmon Induced Hot-Electron TransferDocument8 pagesQQ Highly Efficient Plasmon Induced Hot-Electron TransferdebmallyPas encore d'évaluation
- Radiometric DatingDocument9 pagesRadiometric DatingJohn OsbornePas encore d'évaluation
- Chemistry Paper 1 Revision Mat - Atomic StructureDocument2 pagesChemistry Paper 1 Revision Mat - Atomic StructurekashificetPas encore d'évaluation
- Flashcards - Topic 01 Atomic Structure and The Periodic Table - AQA Chemistry GCSEDocument137 pagesFlashcards - Topic 01 Atomic Structure and The Periodic Table - AQA Chemistry GCSEJaden HalkPas encore d'évaluation
- Test Bank For Introduction To Organic CHDocument19 pagesTest Bank For Introduction To Organic CHClaire TiongsonPas encore d'évaluation
- Lecture 1Document33 pagesLecture 1kiwandaemmanuel21Pas encore d'évaluation
- PhysicalSci12 Q1 Mod1 Week1 2 Formation of Elements v3Document48 pagesPhysicalSci12 Q1 Mod1 Week1 2 Formation of Elements v3Antonio A. Labuguen Jr.Pas encore d'évaluation
- Advanced Periodic TrendsDocument4 pagesAdvanced Periodic TrendsBianca RolstonPas encore d'évaluation
- Chemical Bonding Class11th by PS Sir IIT JEEDocument44 pagesChemical Bonding Class11th by PS Sir IIT JEEMahendra PandaPas encore d'évaluation