Académique Documents
Professionnel Documents
Culture Documents
Examen Final de Quimica II
Transféré par
Renato ProleonDescription originale:
Titre original
Copyright
Formats disponibles
Partager ce document
Partager ou intégrer le document
Avez-vous trouvé ce document utile ?
Ce contenu est-il inapproprié ?
Signaler ce documentDroits d'auteur :
Formats disponibles
Examen Final de Quimica II
Transféré par
Renato ProleonDroits d'auteur :
Formats disponibles
Examen Final de Qumica II
AA-223 E y F
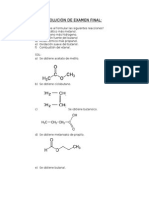
1) Nombras segn IUPAC y escribir las estructuras de todos los ismeros
de formula global C3H6O2
Solucin:
CH3CH2COOH Ac. Propanoico
H-COO-CH2-CH3 Formiato de Etilo
CH3-COO-CH3 Acetato de Metilo
CH2OH-CH=CHOH 1-propen-1,2-diol (Cis, Trans)
CH2OH-CH-=CHOH 1-propen-1,3-diol (Cis, Trans)
CH2OH-COH=CH2
CHOH
CHOH
CH2
2-propen-1,2-diol
1,2- ciclopropanodiol (Cis,
CH3-O-CH=CHOH
CH2=CH-O-CH2OH
2-metoxi etanol (Cis, Trans)
Vinoxi metanol
CH2
CH-CH2OH
CHOH
COH-CH
2-xido de
2-metil-1etileno metanol
hidroxioxido de
etileno (Cis,
Trans)
CH-CH3
CH3-CHOH-CHO
2-hidroxi-propanal
CH3COCH2OH
1-hidroxi-propanona
CH2OH-CH2-CHO
3-hidroxi- propanal
CH2
1-metil-1hidroxioxido de
etileno
2) Seale los nombres IUPAC de los siguientes compuestos poli
funcionales:
NO 2
a) C8H3-C7O-C6H-C5H2-C4H-C3OH-C2H-C1-Cl
O
NH 2
C2H5
Cloruro de 2-amino-6-etoxi-4-fenoxi-3-hidroxi-3-nitro-7-oxooctanilo
b)
O
CH3-O-C3H-C4O-C5O-C6OH-C7H-C8-O-CH2C 2H-COOH CO
C 1OOH
-CH3
N(CH3)2
CH3
Acido 6-cetil-9-dimetilamino-8-p-dimetilbenzoxicarbono-2-carboxi6-hidroxi-3-metoxi-4,5-dioxo-octanoico
c)
SO3
C
Cl-
CO-NH2
CH
CH3
Acido1-acetil-8-carbanil-4-ciano-3-cloroformil-10-formilfenantrenosulfonico
3) Seale las reacciones necesarias para la sntesis del:
A. Acido p-amino benzoico a partir del benceno
B. Eter -antranil--naftilico a partir del antraceno y naftaleno
C. Trans-1,2-dibromo ciclohexano a partir del ciclohexadieno
Solucin:
A.
Alquilaci
n
+ 2CH3Cl
CH
3
NH3
Oxidaci
n
+ KMnO4 H
+
CH
3
Amidaci
n
NH2
COO
H
CONH2
CO
Ac. Paminobenzoico
COO
H
COO
H
COO
H
B.
Cl
Halogenac
in
FeCl3
+
+ Cl2
NaOH
Antrace
no
Cloruro de
Antranilo
O-Na+
O
H
(I)
Na
+
- Antranxido de
Sodio
Antranol
Br
+ Br2
FeCl3
(II)
Naftalen
o
-Bromuro de
Naftilo
Sntesis de
Williamson
(II) + (I)
O
Eter -antranil-naftilico
C.
+ H2
Pt
+Br2
CCl4
B
r
B
r
Trans 1,2-dibromociclohexano
4) A. Cul es el flujo diario que se produce en toneladas de jabn duro s
se saponifican 5x 10
Kg/h de Linolenina y 3x 10
lb/10 min del
lcali, la grasa contiene 90% de pureza y el lcali se encuentra al
70% en masa, adems el rendimiento de procedo de saponificacin
es 80% y el jabn se produce con 95% de pureza? cido Linoleico
C17H29COOH
Solucin:
A.
Reaccin de Saponificacin
M = 8.72
CH2-OOC-C17H29
CH2OH
1CH-OOC-C17H29
CHOH
3 C 17H29-COO+Na + 1
+ 3 NaOH
CH2-OOC-C17H29
CH2OH
872 Kg
0.9 x 5 x
106
lb
min
10
120 Kg
Kg
h
1 Kg
2.2 lb
x
x
900 Kg
1Tn
1000 Kg
60 min
1h
24 h
1 dia
24 h
dia
70
100
1Tn
1000 Kg
Reactivo en Exceso
Limitante
1.08 x
106
1 Tn
Dia
jabn terico
1 Tn
Dia
107
<> 5.7
= 1.03 x
107
Reactivo
x 10
<> 4.5
8
10
COH 95% pureza
Kg
h
1.37 x
Kg
h
1 Tn
Dia
COH Rend. = 80%
<> 4.29
jabn practico
jabn total
x 108
= 8.24 x
= 8.67 x
10
6
10
B.
120 TN de NaOH
x3x
1 TN mol glicerina
Kg
h
6
1 Tn
Dia
1 Tn
Dia
1.37 x
104
1.14 x
Tn
Dia
10
NaOH
TN mol
Rend = 80%
10
n glicerina =
n glicerina = 9.13 x
TN mol
Nitracin (70%)
1 mol glicerina
1 mol nitroglicerina
N nitroglicerina = 0.7 x 9.13 x
10
TN mol
N practica = 6.39 x
10
TN mol
Descomposicin
2 C3H5(NO3)3
6 CO2(g) + 3 N2(g) +
2 mol
6.39 x
10
1
2
O2(g) + 5 H2O(l)
9.5 mol Gases Secos
TN mol
3.035 x
10
TN mol
N gases secos = 0.8 x 3.035 x
4
10 TN mol
N practico = 2.43 x
4
10 TN mol
Con 90% de pureza
4
10 TN mol
n gases secos totales = 2.7 x
Vous aimerez peut-être aussi
- Ejercicios Resueltos de Estructuras HidraulicasDocument58 pagesEjercicios Resueltos de Estructuras HidraulicasJuan Royer TURPO CONDORI82% (123)
- Ingeniería química. Soluciones a los problemas del tomo ID'EverandIngeniería química. Soluciones a los problemas del tomo IPas encore d'évaluation
- Dilatación VolumétricaDocument2 pagesDilatación VolumétricaWILSON MORA100% (1)
- Pa 3 - Algebra MatricialDocument8 pagesPa 3 - Algebra MatricialJAIME IGOR GIL TUANAMAPas encore d'évaluation
- NTP 399 610 Mortero Gral C270Document17 pagesNTP 399 610 Mortero Gral C270Lennyn Quispe ValdiviaPas encore d'évaluation
- Argandoña Grimaldo Carlos Pa1 Introduccion Ingenieria IndustrialDocument5 pagesArgandoña Grimaldo Carlos Pa1 Introduccion Ingenieria IndustrialCarlos Argandoña GrimaldoPas encore d'évaluation
- Examen Final - 2020-10 - CDocument3 pagesExamen Final - 2020-10 - CJeBuz Braulee Lazo Tejeda0% (2)
- Producto Académico 1Document9 pagesProducto Académico 1Jose JGioPas encore d'évaluation
- Autoevaluación #5 - Revisión de IntentosDocument4 pagesAutoevaluación #5 - Revisión de IntentosJOSE ANTONIO SAAVEDRA GUEVARAPas encore d'évaluation
- Examen Final de Química 2Document4 pagesExamen Final de Química 2Juan Carlos Zuñiga JustinianoPas encore d'évaluation
- Tesis Hidrogeno PDFDocument197 pagesTesis Hidrogeno PDFmilser roel campos bautistaPas encore d'évaluation
- Autoevaluacion 1 - Quimica 2Document5 pagesAutoevaluacion 1 - Quimica 2Paul Vidal MunivePas encore d'évaluation
- Antivibrantes ROSTADocument12 pagesAntivibrantes ROSTAGustavo GaunaPas encore d'évaluation
- Evaluacion de Consolidado 1 Matematica Discreta 2022Document3 pagesEvaluacion de Consolidado 1 Matematica Discreta 2022venta examenesPas encore d'évaluation
- 1ra Evaluación Del Consolidado 2Document3 pages1ra Evaluación Del Consolidado 2JHOAN ANTONY PEREZ GARCIA100% (1)
- Examen Final de Quimica 15-2Document6 pagesExamen Final de Quimica 15-2Renato ProleonPas encore d'évaluation
- Examen Parcial de Quimica 2 Fia-Uni (Masguito)Document9 pagesExamen Parcial de Quimica 2 Fia-Uni (Masguito)Ricardo ChuraPas encore d'évaluation
- Producto Académico 3: Química 2Document9 pagesProducto Académico 3: Química 2FREDI HUATAPas encore d'évaluation
- Examen Sustitutorio Quimica 2 2020 2Document3 pagesExamen Sustitutorio Quimica 2 2020 2brandonleo1920Pas encore d'évaluation
- Sem 09 - PRÁCTICA - TIPOS DE CARBONODocument2 pagesSem 09 - PRÁCTICA - TIPOS DE CARBONOconcha luisPas encore d'évaluation
- Producto Academico 3 - Quimica 1Document14 pagesProducto Academico 3 - Quimica 1AlexPas encore d'évaluation
- Producto Académico N°2 (Cuestionario)Document4 pagesProducto Académico N°2 (Cuestionario)JOSE ANTONIO SAAVEDRA GUEVARAPas encore d'évaluation
- T2 AutoevaluacionDocument3 pagesT2 AutoevaluacionDiego Martinez MorenoPas encore d'évaluation
- GUÍA DE APRENDIZAJE UNIDAD 3 - Química 2Document18 pagesGUÍA DE APRENDIZAJE UNIDAD 3 - Química 2Daniel OrellanaPas encore d'évaluation
- Examen Sustitutorio de QuimicaDocument7 pagesExamen Sustitutorio de QuimicaNikolas Jaen100% (1)
- Producto Académico N°1 (Cuestionario)Document4 pagesProducto Académico N°1 (Cuestionario)JOSE ANTONIO SAAVEDRA GUEVARAPas encore d'évaluation
- Producto Academico 3 Quimica 2 ValidadoDocument6 pagesProducto Academico 3 Quimica 2 ValidadoJairo Alania Vilcachagua100% (1)
- Evaluación Final BDocument2 pagesEvaluación Final BLuis AntonioPas encore d'évaluation
- Autoevaluación 2Document5 pagesAutoevaluación 2adolfo diazPas encore d'évaluation
- Producto Académico 01 - TAREADocument13 pagesProducto Académico 01 - TAREAHank Carti Sunga0% (1)
- Pa3 QuimicaDocument3 pagesPa3 QuimicaRyuzaki KuroShin クロしんPas encore d'évaluation
- Autoev 3 y 4 QuimicaDocument3 pagesAutoev 3 y 4 Quimicagloria alvarezPas encore d'évaluation
- Evaluación Consolidado 2 2023 - VirtualDocument2 pagesEvaluación Consolidado 2 2023 - VirtualMabel ChinoPas encore d'évaluation
- Examen Final Del Curso de Química 1Document3 pagesExamen Final Del Curso de Química 1Alexis VPPas encore d'évaluation
- Ariann Diego Alvarez Begazo Quimica ExmDocument4 pagesAriann Diego Alvarez Begazo Quimica ExmDiego BegazoPas encore d'évaluation
- Evaluacion Parcial Quimica 01 CGT 2010 10 BDocument7 pagesEvaluacion Parcial Quimica 01 CGT 2010 10 BAlfredo Secce100% (1)
- PRACTICA #1 Virtual Quimica 2Document6 pagesPRACTICA #1 Virtual Quimica 2Julio Cesar Alarcon Astoquilca0% (1)
- Evaluacion de Consolidado 2 Matematica Discreta 2022 - 2BDocument3 pagesEvaluacion de Consolidado 2 Matematica Discreta 2022 - 2BELVIS ALBERTO RAMIREZ HUANCAPas encore d'évaluation
- Pa3 Algebra 2020Document4 pagesPa3 Algebra 2020Milagros Mamani PumaPas encore d'évaluation
- Solucionario Evaluación Parcial Quimica - PRUEBA 1Document3 pagesSolucionario Evaluación Parcial Quimica - PRUEBA 1ARNALDO ELARD HUILLCA HUARCAPas encore d'évaluation
- Examen ParcialDocument4 pagesExamen ParcialAdela Karen Segura ZegarraPas encore d'évaluation
- Pa2 2022 10a ActualizadoDocument2 pagesPa2 2022 10a Actualizadoruizprado71Pas encore d'évaluation
- PA1 quimiCADocument3 pagesPA1 quimiCALadydiana VilavilaPas encore d'évaluation
- Eval Parcial - Fund - Cáculo - GQT - 2020-10 - A - TIPO - (C)Document2 pagesEval Parcial - Fund - Cáculo - GQT - 2020-10 - A - TIPO - (C)Dageus Skoteinó IppótiPas encore d'évaluation
- Producto Académico QuimicaDocument3 pagesProducto Académico QuimicaGEDEON1100% (1)
- Taller #2 Solucionario GasesDocument3 pagesTaller #2 Solucionario GasesADONY ADY CHURAMPI ANTONIOPas encore d'évaluation
- Pa3 2Document7 pagesPa3 2Bryana Baes Flores100% (1)
- QUIMICA Producto Academico N 2 BDocument4 pagesQUIMICA Producto Academico N 2 BRuben CatariPas encore d'évaluation
- P4 ButaldehidoDocument12 pagesP4 ButaldehidoAriana CastroPas encore d'évaluation
- Producto Académico Nº1Document3 pagesProducto Académico Nº1AlejandroHuacasiCornejoPas encore d'évaluation
- Retroalimentación de Química 2017-20-BDocument6 pagesRetroalimentación de Química 2017-20-BWilliam Ortiz100% (1)
- Solucionario 2Document2 pagesSolucionario 2Xiomara SalvatierraPas encore d'évaluation
- Practica #1 Termoquímica I y IiDocument4 pagesPractica #1 Termoquímica I y IiWYNNY ALICIA BELTRAN QUISPE0% (1)
- Guia de Fundamentos Del Cálculo-2022-20Document97 pagesGuia de Fundamentos Del Cálculo-2022-20Fabrizzio Miguel Vergara DiazPas encore d'évaluation
- Producto Academico 3 DefinitivoDocument10 pagesProducto Academico 3 DefinitivoELIAS ZUNIGA TACACPas encore d'évaluation
- Ejercicios para El Consolidado 1 7655CZDocument1 pageEjercicios para El Consolidado 1 7655CZCARMEN CECILIA LOPEZ TAPARAPas encore d'évaluation
- Evaluación Final Ingeniería de MaterialesDocument4 pagesEvaluación Final Ingeniería de MaterialesANALIZ VIVIANA MARTINEZ BARRETOPas encore d'évaluation
- Álgebra Matricial y Geometría Analítica - Tipo ADocument3 pagesÁlgebra Matricial y Geometría Analítica - Tipo APEDRO BLAS COILLO MAMANIPas encore d'évaluation
- Reporte Semana 11 - Sintesis de Compuestos InorgánicosDocument3 pagesReporte Semana 11 - Sintesis de Compuestos InorgánicosFRANKLIN ROUSBEL ROJAS PACHECOPas encore d'évaluation
- Solucionario Producto Académico 1Document9 pagesSolucionario Producto Académico 1Nayeli PalominoPas encore d'évaluation
- Lab SistAxonometricoOblicuo 04Document111 pagesLab SistAxonometricoOblicuo 04FREDY MENDIGURE LLOCLLEPas encore d'évaluation
- Quimica 2 Solucionario de Los FinalesDocument21 pagesQuimica 2 Solucionario de Los FinalesRenato ProleonPas encore d'évaluation
- Solucionario de Los Problemas de Quimica IiDocument16 pagesSolucionario de Los Problemas de Quimica IiRenato ProleonPas encore d'évaluation
- 30 Problemas de MasgitoDocument30 pages30 Problemas de MasgitoSAKURA ALEXIA �A�EZ MATTAPas encore d'évaluation
- Quimica IIDocument15 pagesQuimica IIMarthaLorenzoCastilloPas encore d'évaluation
- TUTUNADIECOMOTUTUDocument25 pagesTUTUNADIECOMOTUTURenato ProleonPas encore d'évaluation
- JuctionsDocument1 pageJuctionsRenato ProleonPas encore d'évaluation
- C Ultura Tiahuanaco: Se Desarrolló enDocument6 pagesC Ultura Tiahuanaco: Se Desarrolló enRenato ProleonPas encore d'évaluation
- Demanda LlumpaDocument1 pageDemanda LlumpaRenato ProleonPas encore d'évaluation
- Uprobys: Proyecto 1Document1 pageUprobys: Proyecto 1Renato ProleonPas encore d'évaluation
- Infografia Rrss MunicipalesDocument1 pageInfografia Rrss MunicipalesRenato ProleonPas encore d'évaluation
- Microsoft Powerpoint - Sistema ShimantoDocument15 pagesMicrosoft Powerpoint - Sistema ShimantoRenato ProleonPas encore d'évaluation
- 02 Calculo Estructural Barraje Con CanalDocument10 pages02 Calculo Estructural Barraje Con CanalJames Rolando Segura HernándezPas encore d'évaluation
- Tarea 2 Abastos 2 19-3Document3 pagesTarea 2 Abastos 2 19-3Renato ProleonPas encore d'évaluation
- Tarea 1 Abastos 2Document10 pagesTarea 1 Abastos 2Renato ProleonPas encore d'évaluation
- UASBDocument4 pagesUASBRenato ProleonPas encore d'évaluation
- 00Document5 pages00Renato ProleonPas encore d'évaluation
- Expo N°1 - Tuberias PVC - Final 4 PMDocument24 pagesExpo N°1 - Tuberias PVC - Final 4 PMRenato ProleonPas encore d'évaluation
- Monografia 1 Matemática Financiera Conceptos Generales1Document12 pagesMonografia 1 Matemática Financiera Conceptos Generales1Renato ProleonPas encore d'évaluation
- Ley de Los GasesDocument52 pagesLey de Los Gasesodey maroufPas encore d'évaluation
- Microestructuras Del Al, PB, SN II RegDocument31 pagesMicroestructuras Del Al, PB, SN II RegJordan Capa ZamataPas encore d'évaluation
- Adobe - Ensayos Sismicos MurosDocument11 pagesAdobe - Ensayos Sismicos Muroswachuma100% (4)
- Arte GoticoDocument19 pagesArte GoticoEdwin Miguel TumPas encore d'évaluation
- ROXANADocument7 pagesROXANAJose L Huaman GasparPas encore d'évaluation
- Características Del Gas LPGDocument4 pagesCaracterísticas Del Gas LPGCristobal RomoPas encore d'évaluation
- Notas Acero InoxidableDocument6 pagesNotas Acero InoxidableVicky RamosPas encore d'évaluation
- Informe Alcoholes y FenolesDocument2 pagesInforme Alcoholes y FenolesEsteban Ocampo EcheverriPas encore d'évaluation
- FT Geomembrana de Hdpe Lisa Nominal 1 5 MM - Corporación Geo ExtruplastDocument1 pageFT Geomembrana de Hdpe Lisa Nominal 1 5 MM - Corporación Geo ExtruplastBilmaSilvaRojasPas encore d'évaluation
- Tarea 1 Mecanica de MaterialesDocument7 pagesTarea 1 Mecanica de MaterialesWilliam RodriguesPas encore d'évaluation
- Tablastacas PDFDocument7 pagesTablastacas PDFAndres WartanbelPas encore d'évaluation
- Tema 2Document15 pagesTema 2Alan ApalaPas encore d'évaluation
- Décimo - Guía de Aprendizaje Tercer PeriodoDocument14 pagesDécimo - Guía de Aprendizaje Tercer Periodojalcachofa x06Pas encore d'évaluation
- Memoria Descriptiva Estructuras - Manuel PradoDocument65 pagesMemoria Descriptiva Estructuras - Manuel PradoRodrigo Teran MermaPas encore d'évaluation
- Pirólisis FlashDocument4 pagesPirólisis FlashGrace Vera100% (1)
- Tabla Periódica y Enlace QuimicoDocument95 pagesTabla Periódica y Enlace QuimicoAnthony GodoyPas encore d'évaluation
- Fajas TransportadorasDocument35 pagesFajas TransportadorasJaqueline ChambillaPas encore d'évaluation
- Mapa Conceptual Sobre Métodos de Limpieza e Higiene en La Industria AlimentariaDocument1 pageMapa Conceptual Sobre Métodos de Limpieza e Higiene en La Industria AlimentariaRosPas encore d'évaluation
- Ácidos y BasesDocument32 pagesÁcidos y Basesluisa niñoPas encore d'évaluation
- MATRIZ DE IDENTIFICACION Y EVALUACION AeIADocument13 pagesMATRIZ DE IDENTIFICACION Y EVALUACION AeIAyamilPas encore d'évaluation
- ESTRUCTURAS METALICAS - Analisis de CasoDocument7 pagesESTRUCTURAS METALICAS - Analisis de Casoaron100% (1)
- Sistema A Base de PorticosDocument22 pagesSistema A Base de PorticosElizabeth Gamarra RodriguezPas encore d'évaluation
- Taller 4 Termodinamica - 2020Document3 pagesTaller 4 Termodinamica - 2020Harry Zty MedinaPas encore d'évaluation
- 02 Especificaciones SECTOR 7A 9 R3 API-ZLO-13-15Document65 pages02 Especificaciones SECTOR 7A 9 R3 API-ZLO-13-15Yazmin MurilloPas encore d'évaluation
- Quimica-Brayan Pinzon GomezDocument4 pagesQuimica-Brayan Pinzon GomezBrayan PgPas encore d'évaluation
- Práctica 4. Identificación de Cationes Por Vía HúmedaDocument10 pagesPráctica 4. Identificación de Cationes Por Vía HúmedaMartínez García Miriam AriadnaPas encore d'évaluation