Académique Documents
Professionnel Documents
Culture Documents
How To Identify Geometrical Isomers by S.K.sinha See Chemistry Animations at
Transféré par
myiitchemistryDescription originale:
Titre original
Copyright
Formats disponibles
Partager ce document
Partager ou intégrer le document
Avez-vous trouvé ce document utile ?
Ce contenu est-il inapproprié ?
Signaler ce documentDroits d'auteur :
Formats disponibles
How To Identify Geometrical Isomers by S.K.sinha See Chemistry Animations at
Transféré par
myiitchemistryDroits d'auteur :
Formats disponibles
SINHA I. I.T.
CHEMISTRY
How to identify Geometrical Isomers.
Organic compounds which have same molecular formula but different non-interchangable
geometry due to different orientation of atoms in space are called Geometrical Isomers.
Example.
H3C CH3 H3C H
H H H CH3
cis-but-2-ene trans)-but-2-ene
Geometrical Isomers are
1. different compounds i.e non-superimposable i.e. cannot be interconverted without bond
dissociation
2. A type of configurational isomers i.e. they differs in orientation at least at one carbon
atom. Or ( stereo center)
3. Being isomers they have same molecular formula , empirical formule , % composition
4. They have a defined geometry.
5. As they geometrically differs therefore have different dipole moments , mp, bp, vapour
pressure , solubility.
Geometrical Isomerism is found in compounds which have
a. Planner geometry (real or virtual)
b. Different substituent on either side of imaginary or real planner geometry.
c. No interconvertibility ( Presence of Pi bond , continuous pi bond etc.
Planner geometry (real or virtual) :-It is found in following cases.
H3C H
2
sp carbon
1. Alkene. H CH3
2.Alkene (cumulene with odd no of pi bonds)
H3C
C C CH3
H H
.
Z Z
Y
Y
Y
H3C CH3
C C
H H X
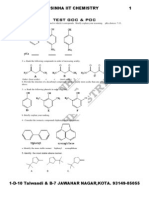
1-D 10 TALWANDI, KOTA 09314905055
SINHA I. I.T. CHEMISTRY
3 Cycloalkanes
XZ plane
H3C CH3
H H
4. Odd no Spiranes
H3C CH3
H H
5.Odd no combination of spiro & cumulene
H3C
CH3
H H
6.Cycloalkene with carbon greater than 8
H
H
H cis H trans
7 Fused rings also have cis & trans orientation
H3C
H
H
trans
CH3
cis
Notation system:
1. cis & trans.
2. Syn and anti
3. E & Z.
1. cis & trans cis and trans notation is used only if there are two identical groups on either
side of the planer geometry.
cis trans none
Ex.
2. Syn and Anti notation is used only if there are C=N or N=N bond with different
substituents ( Historical approach used by fisher for Oximes).
1-D 10 TALWANDI, KOTA 09314905055
SINHA I. I.T. CHEMISTRY
Ex.
H3C OH H OH H3C OH
N N N
H anti H3C syn H3C none
H3C CH3
CH3
N N
N N
syn H3C anti
3.E and Z notations are introduced to designate geometrical isomers of any type.
1. H3C 1. H3C
Cl Br 1.
Ex. H E Br 1. H Z Cl 1. represent high priority group
No. of geometrical units and no of geometrical isomers.
Total no of geometrical isomers depends on
1. no of geometrical units
2. symmetry/ unsymmetry
3. other structural limitations.
CH3
CH3
H3C
H3 C
1. C C CH3 C C CH3
2.
Ex. H3C CH3 H CH3
CH3
H3C H 3C CH3
H 3C CH3
3. C C H H
4.
H
H H H H
H3C H3C CH3
CH36.
H3C
H
5. H
H H
H CH3
H3C H
H3C CH3
H3C CH3
8.
7.
H H
H H H3C H
1-D 10 TALWANDI, KOTA 09314905055
SINHA I. I.T. CHEMISTRY
H3C CH3 H H
9.
CH3
H H CH3
H CH 3
H 3C C C CH 3
10 C C
H 3C H H H H
H
H3C
11. H
H CH3
H H
H3C
12. H
H CH3
CH3
H
H H
14. 15.
13.
1-D 10 TALWANDI, KOTA 09314905055
Vous aimerez peut-être aussi
- How To Find No of Structural Isomers by S.K.sinha See Chemistry Animations at HTTP://WWW - Openchemistry.inDocument2 pagesHow To Find No of Structural Isomers by S.K.sinha See Chemistry Animations at HTTP://WWW - Openchemistry.inmyiitchemistry81% (16)
- Electrochemistry IPEDocument18 pagesElectrochemistry IPEAdiChemAdi100% (3)
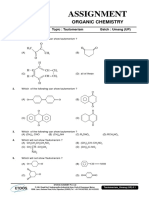
- Assignment: Organic ChemistryDocument6 pagesAssignment: Organic ChemistryWalid EbaiedPas encore d'évaluation
- TEST 2 GOC & POC Tough by S.K.sinha See Chemistry Animations atDocument3 pagesTEST 2 GOC & POC Tough by S.K.sinha See Chemistry Animations atmyiitchemistryPas encore d'évaluation
- H.D.A. 2021Document54 pagesH.D.A. 2021Every Time Chemistry [ ETC]Pas encore d'évaluation
- Solid State-1Document31 pagesSolid State-1ChirAgPas encore d'évaluation
- Organic ChemistryDocument20 pagesOrganic ChemistryGirish RaguvirPas encore d'évaluation
- Solved Example: Chemistry For Neet & AiimsDocument24 pagesSolved Example: Chemistry For Neet & AiimsAnup KPas encore d'évaluation
- GOC Practice Assignment CHCDocument26 pagesGOC Practice Assignment CHCjanviPas encore d'évaluation
- Chem 212 Alkyl Halide Problems 3Document1 pageChem 212 Alkyl Halide Problems 3kevinamyPas encore d'évaluation
- Reaction IntermediatesDocument20 pagesReaction IntermediatesSacchitDShethPas encore d'évaluation
- Dipole Moments in Organic CHEMISTRYDocument18 pagesDipole Moments in Organic CHEMISTRYBalraj Dhillon100% (2)
- Mole Concept 2Document38 pagesMole Concept 2R S.NagiPas encore d'évaluation
- Atomic Structure IITDocument16 pagesAtomic Structure IITAdiChemAdi69% (13)
- Name Reactions-II PDFDocument32 pagesName Reactions-II PDFEstanislao Amadeo Avogadro100% (1)
- Chem 212 Alkyl Halide Problems 2Document1 pageChem 212 Alkyl Halide Problems 2kevinamy100% (1)
- Reaction IntermediateDocument57 pagesReaction IntermediateMohd Shah Faisal75% (4)
- Prince Singh: Physical & Inorganic ChemistryDocument5 pagesPrince Singh: Physical & Inorganic ChemistryJatin SinglaPas encore d'évaluation
- Chem 212 Alkyl Halide Problems 4Document1 pageChem 212 Alkyl Halide Problems 4kevinamyPas encore d'évaluation
- Alkyl HalidesDocument20 pagesAlkyl HalidesShivam Gupta0% (1)
- Reaction IntermediateDocument20 pagesReaction IntermediateSiddarth Singh100% (2)
- Stereochemistry Basic Concepts Useful Notes For StudentsDocument26 pagesStereochemistry Basic Concepts Useful Notes For StudentsReddappaPas encore d'évaluation
- Chemistry 3 Electrochemistry PDFDocument15 pagesChemistry 3 Electrochemistry PDFinfinilifePas encore d'évaluation
- Periodic Table IPEDocument15 pagesPeriodic Table IPEAdiChemAdi100% (4)
- Isomerism - Handwritten NotesDocument7 pagesIsomerism - Handwritten Notesgovind_galamPas encore d'évaluation
- G R Reduction AlkaneDocument43 pagesG R Reduction AlkaneManthan HaritashPas encore d'évaluation
- Part - I: Objective Questions: Section A: Geometrical IsomerismDocument10 pagesPart - I: Objective Questions: Section A: Geometrical IsomerismTejas pawarPas encore d'évaluation
- Solution - Colligative Properties Solutions PDFDocument25 pagesSolution - Colligative Properties Solutions PDFGOURISH AGRAWALPas encore d'évaluation
- Resonance and Inductive Effects PresentationDocument36 pagesResonance and Inductive Effects Presentationeagl33yePas encore d'évaluation
- IsomerismDocument62 pagesIsomerismsubesinghPas encore d'évaluation
- Solid State Chemistry IPEDocument15 pagesSolid State Chemistry IPEAdiChemAdi100% (4)
- Alkene DPPDocument20 pagesAlkene DPPKalyan ReddtPas encore d'évaluation
- Organic 6 CDocument26 pagesOrganic 6 CDr.Rajarshi PatelPas encore d'évaluation
- General Organic Chemistry For IITDocument21 pagesGeneral Organic Chemistry For IITParas Thakur83% (6)
- S BlockDocument84 pagesS BlockPrakhar ShuklaPas encore d'évaluation
- 01 D and F Block Elements Theory Final EDocument17 pages01 D and F Block Elements Theory Final Etech 2 life100% (1)
- Chapter 1 - Introduction To Organic ChemistryDocument102 pagesChapter 1 - Introduction To Organic ChemistryMELVINDO JACOBPas encore d'évaluation
- Problems On Named ReactionsDocument103 pagesProblems On Named ReactionsBapu ThoratPas encore d'évaluation
- Thermodynamics PDFDocument38 pagesThermodynamics PDFUjjwal KumarPas encore d'évaluation
- SKM Organic Chem NotesDocument267 pagesSKM Organic Chem NotesGyanesh DwivediPas encore d'évaluation
- 03ElectronicdisplacementEffects Exercise Send1Document33 pages03ElectronicdisplacementEffects Exercise Send1Aaryan Keshan100% (1)
- Geometrical Isomerism (Animated)Document114 pagesGeometrical Isomerism (Animated)myiitchemistry60% (5)
- NEO JEE 11 P1 CHE E Organic Chemistry - Some Basic Principle and Techniques S17 209Document481 pagesNEO JEE 11 P1 CHE E Organic Chemistry - Some Basic Principle and Techniques S17 209Shubh NawaniPas encore d'évaluation
- Chemical Kinetics (M) PDFDocument41 pagesChemical Kinetics (M) PDFNalla Umapathi Reddy75% (4)
- Coordination Compounds 19-06-2020Document6 pagesCoordination Compounds 19-06-2020Vanshaj GuptaPas encore d'évaluation
- Aldol Reaction - ChemistryDocument7 pagesAldol Reaction - ChemistryGamer HelperPas encore d'évaluation
- Bioinorganic HandoutDocument63 pagesBioinorganic HandoutAL__52Pas encore d'évaluation
- Stereoisomerism VKP SirDocument49 pagesStereoisomerism VKP SirSandeep ReddyPas encore d'évaluation
- CH CH CHCH CH H CH CH CH CH CH CH H CH: Byvineet Khatri SirDocument13 pagesCH CH CHCH CH H CH CH CH CH CH CH H CH: Byvineet Khatri Sirsarvesh goyalPas encore d'évaluation
- Reduction, Oxidation - Hydrolysis Exercise PDFDocument24 pagesReduction, Oxidation - Hydrolysis Exercise PDFGOURISH AGRAWAL100% (3)
- Chapter 13: Nuclear Magnetic Resonance (NMR) SpectrosDocument20 pagesChapter 13: Nuclear Magnetic Resonance (NMR) SpectrosWwe 2Pas encore d'évaluation
- Important Concepts: Conformational Analysis of AlkanesDocument4 pagesImportant Concepts: Conformational Analysis of AlkanesKayla JoezettePas encore d'évaluation
- Steroisomerism: Classification of StereoisomersDocument20 pagesSteroisomerism: Classification of StereoisomersParas BaldotraPas encore d'évaluation
- Stereochemistry: Concepts For Chiral CompoundsDocument6 pagesStereochemistry: Concepts For Chiral CompoundsSankar AdhikariPas encore d'évaluation
- Cis and Trans IsomersDocument2 pagesCis and Trans IsomersNosiphesihle ShanduPas encore d'évaluation
- Chapter 5 ProblemsDocument7 pagesChapter 5 Problemsdicoz diphaPas encore d'évaluation
- Part.4 (رابعه حيوان) Photo 2021Document8 pagesPart.4 (رابعه حيوان) Photo 2021amira emamPas encore d'évaluation
- Section 3 - StereochemistryDocument21 pagesSection 3 - Stereochemistrysf9 fanfarePas encore d'évaluation
- Chem261, B2 Practice Questions For The Midterm Exam, AnswersDocument8 pagesChem261, B2 Practice Questions For The Midterm Exam, Answerschemistry tutorialPas encore d'évaluation
- Set of 50 Obj in General Organic Chemistry by S.K.sinha HTTP://WWW - Openchemistry.inDocument6 pagesSet of 50 Obj in General Organic Chemistry by S.K.sinha HTTP://WWW - Openchemistry.inmyiitchemistry50% (4)
- Solid State / Crystalline State ChemistryDocument26 pagesSolid State / Crystalline State ChemistrymyiitchemistryPas encore d'évaluation
- Unique Approach of Problem Solving in Isomerism by S.K.sinha HTTP://WWW - Openchemistry.inDocument6 pagesUnique Approach of Problem Solving in Isomerism by S.K.sinha HTTP://WWW - Openchemistry.inmyiitchemistry100% (3)
- Geometrical Isomerism (Animated)Document114 pagesGeometrical Isomerism (Animated)myiitchemistry60% (5)
- Mole Concept Solution Practice Set 1 Objective by S.K.sinha See Chemistry Animations atDocument2 pagesMole Concept Solution Practice Set 1 Objective by S.K.sinha See Chemistry Animations atmyiitchemistry100% (4)
- Basic Inorganic Nomenclature FOR IIT-JEE ENTRANCE TEST by S.K.sinha See Chemistry Animations atDocument5 pagesBasic Inorganic Nomenclature FOR IIT-JEE ENTRANCE TEST by S.K.sinha See Chemistry Animations atmyiitchemistry88% (17)
- Mole Concept Solution Practice Set Objective by S.K.sinha See Chemistry Animations atDocument1 pageMole Concept Solution Practice Set Objective by S.K.sinha See Chemistry Animations atmyiitchemistry50% (2)
- IONIC EQUILLIBRIUM FOR IIT-JEE ENTRANCE TEST by S.K.sinha See Chemistry Animations at Http://openchemistry - inDocument31 pagesIONIC EQUILLIBRIUM FOR IIT-JEE ENTRANCE TEST by S.K.sinha See Chemistry Animations at Http://openchemistry - inmyiitchemistry100% (7)
- Mole & Stoichiometry by S.K.sinha See Chemistry Animations atDocument3 pagesMole & Stoichiometry by S.K.sinha See Chemistry Animations atmyiitchemistry50% (2)
- Mole Concept Empirical & Molecular Formula by S.K.sinha See Chemistry Animations atDocument3 pagesMole Concept Empirical & Molecular Formula by S.K.sinha See Chemistry Animations atmyiitchemistry100% (5)
- UNSTABLE FUNCTIONAL GROUPS IN ORGANIC by S.K.sinha See Chemistry Animations atDocument1 pageUNSTABLE FUNCTIONAL GROUPS IN ORGANIC by S.K.sinha See Chemistry Animations atmyiitchemistry100% (2)
- JEE - Similar Test Paper Goc & Alkane by S.K.sinha See Chemistry Animations atDocument4 pagesJEE - Similar Test Paper Goc & Alkane by S.K.sinha See Chemistry Animations atmyiitchemistry100% (9)
- Test Goc 3 by S.K.sinha See Chemistry Animations atDocument6 pagesTest Goc 3 by S.K.sinha See Chemistry Animations atmyiitchemistryPas encore d'évaluation
- Organic Nomenclature HTTPDocument19 pagesOrganic Nomenclature HTTPmyiitchemistry91% (11)
- 6th Central Pay Commission Salary CalculatorDocument15 pages6th Central Pay Commission Salary Calculatorrakhonde100% (436)
- PRACTICAL ORGANIC CHEMISTRY FOR IIT-JEE ENTRANCE TEST by S.K.sinha See Chemistry Animations atDocument2 pagesPRACTICAL ORGANIC CHEMISTRY FOR IIT-JEE ENTRANCE TEST by S.K.sinha See Chemistry Animations atmyiitchemistry100% (10)
- Some Important Organic Information by S.K.sinha See Chemistry Animations atDocument2 pagesSome Important Organic Information by S.K.sinha See Chemistry Animations atmyiitchemistry83% (6)
- Test1 Goc & Poc Tough by S.K.sinha See Chemistry Animations atDocument3 pagesTest1 Goc & Poc Tough by S.K.sinha See Chemistry Animations atmyiitchemistry100% (1)
- Intratec PortfolioDocument279 pagesIntratec Portfolioagarwalashwin32Pas encore d'évaluation
- Dowfax Muy BuenoDocument20 pagesDowfax Muy Buenohugogauna100% (1)
- June 2016 MS - Unit 1 Edexcel Biology A-LevelDocument28 pagesJune 2016 MS - Unit 1 Edexcel Biology A-LevelAyse KerimPas encore d'évaluation
- USP Definition of HPLC Column ClassificationDocument9 pagesUSP Definition of HPLC Column ClassificationLuisPas encore d'évaluation
- ChemiluminescenceDocument7 pagesChemiluminescenceErinaldo CarvalhoPas encore d'évaluation
- Glycine MetabolismDocument28 pagesGlycine MetabolismSuryansh GoyalPas encore d'évaluation
- Structure and Chemical Composition of Raw Hides and SkinsDocument11 pagesStructure and Chemical Composition of Raw Hides and SkinsAnushree Bhattacharya75% (4)
- Guidelines Ventilation Requirements in Buildings: Indoor Air Quality & Its Impact On ManDocument44 pagesGuidelines Ventilation Requirements in Buildings: Indoor Air Quality & Its Impact On ManBruno RochaPas encore d'évaluation
- Amrut MittiDocument8 pagesAmrut Mittidonbosskiss100% (1)
- The Conversion of Carboxylic Acids To KetonesDocument5 pagesThe Conversion of Carboxylic Acids To KetonesKybernetikumPas encore d'évaluation
- Absolute Structure Determination - PLOTONDocument29 pagesAbsolute Structure Determination - PLOTONuday_khandavilliPas encore d'évaluation
- LidocaineDocument6 pagesLidocaineg20kpPas encore d'évaluation
- Class 32Document23 pagesClass 32Ramasubramaniam PalaniswamyPas encore d'évaluation
- Organic Pesticide With The Use of Hot Pepper and GarlicDocument6 pagesOrganic Pesticide With The Use of Hot Pepper and GarlicJen Julianna100% (1)
- Enzyme Technology by Martin F. ChaplinDocument149 pagesEnzyme Technology by Martin F. ChaplinAKSHAYPas encore d'évaluation
- Application of Nanoparticles in Biofuels - An OverviewDocument18 pagesApplication of Nanoparticles in Biofuels - An OverviewYashvir SinghPas encore d'évaluation
- 2011 WGMH New Orleans Moisture Determination Draft RecommendationDocument11 pages2011 WGMH New Orleans Moisture Determination Draft RecommendationKristiene Balatbat SadiwaPas encore d'évaluation
- SQ Presentation 2021-r2Document43 pagesSQ Presentation 2021-r2nadeem4ahmed-805026Pas encore d'évaluation
- D6778 14Document7 pagesD6778 14jenishjsPas encore d'évaluation
- Daftar Bahan Berbahaya Dan Beracun (B3) Rumah Sakit Eka Di BSDDocument78 pagesDaftar Bahan Berbahaya Dan Beracun (B3) Rumah Sakit Eka Di BSDReniPas encore d'évaluation
- The Feasiblity of Using Indigenous DyestuffDocument2 pagesThe Feasiblity of Using Indigenous Dyestuffalvin90% (10)
- Basic Requirement For Dyes:: Department of Chemistry A.M.U., AligarhDocument14 pagesBasic Requirement For Dyes:: Department of Chemistry A.M.U., AligarhHarshita MayalPas encore d'évaluation
- Chapter 3 CrosswordDocument1 pageChapter 3 Crosswordapi-372001543Pas encore d'évaluation
- Pak Pharma IndexDocument54 pagesPak Pharma IndexDar LeePas encore d'évaluation
- University of Cambridge International Examinations General Certifi Cate of Education Advanced Subsidiary Level and Advanced LevelDocument12 pagesUniversity of Cambridge International Examinations General Certifi Cate of Education Advanced Subsidiary Level and Advanced LevelHubbak KhanPas encore d'évaluation
- Gram Negative Positive NotesDocument8 pagesGram Negative Positive NotesKusum Roy100% (1)
- Membranes in CellsDocument25 pagesMembranes in CellsPrakashdev99Pas encore d'évaluation
- Turf Essential PlusDocument2 pagesTurf Essential PlusGladio SeifPas encore d'évaluation
- Lithium CarbonateDocument9 pagesLithium Carbonatehumag143Pas encore d'évaluation
- Introduction To Chemistry General Organic and BiologicalDocument1 236 pagesIntroduction To Chemistry General Organic and BiologicalPamela MathernePas encore d'évaluation