Académique Documents
Professionnel Documents
Culture Documents
Chapter Outline: Phase Transformations. Kinetics
Transféré par
mpcd07Description originale:
Titre original
Copyright
Formats disponibles
Partager ce document
Partager ou intégrer le document
Avez-vous trouvé ce document utile ?
Ce contenu est-il inapproprié ?
Signaler ce documentDroits d'auteur :
Formats disponibles
Chapter Outline: Phase Transformations. Kinetics
Transféré par
mpcd07Droits d'auteur :
Formats disponibles
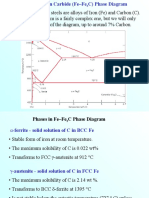
Introduction to Materials Science, Chapter 10, Phase Transformations in Metals
Phase transformations. Kinetics. Chapter Outline: Phase Transformations in Metals
Phase transformations (change of the microstructure) can Heat Treatment (time and temperature) be divided into three categories: Microstructure Mechanical Properties ! Diffusion-dependent with no change in phase composition or number of phases present (e.g. melting, solidification of pure metal, allotropic ! transformations, Kinetics of phase transformations etc.) recrystallization, ! Multiphase Transformations ! Diffusion-dependent with changes in phase ! compositions Phase transformations in Fe-C and/or number ofalloys phases (e.g. eutectoid transformations) ! Isothermal Transformation Diagrams ! phase transformation - produces a ! Diffusionless Mechanical Behavior metastable phase by cooperative small displacements of atoms in structure (e.g. martensitic transformation ! all Tempered Martensite discussed in later in this chapter) Phase transformations do not occur instantaneously. Not tested: Diffusion-dependent phase transformations can be rather 10.6 Continuous Cooling Transformation Diagrams slow and the final structure often depend on the rate of cooling/heating.
We need to consider the time dependence or kinetics of the phase transformations.
University of Virginia, Dept. of Materials Science and Engineering 1 2
Introduction to Materials Science, Chapter 10, Phase Transformations in Metals
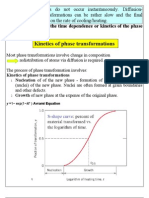
Kinetics of phase transformations
Most phase transformations involve change in composition redistribution of atoms via diffusion is required. The process of phase transformation involves: ! Nucleation of of the new phase - formation of stable small particles (nuclei) of the new phase. Nuclei are often formed at grain boundaries and other defects. ! Growth of new phase at the expense of the original
phase.
S-shape curve: percent of material transformed vs. the logarithm of time.
University of Virginia, Dept. of Materials Science and Engineering
Introduction to Materials Science, Chapter 10, Phase Transformations in Metals
Nucleation
A nucleus is only stable if further growth reduces the energy of the system. For r > rc the nucleus is stable.
University of Virginia, Dept. of Materials Science and Engineering
Introduction to Materials Science, Chapter 10, Phase Transformations in Metals
Rate of phase transformations
Rate of transformation can be defined as reciprocal of time for transformation to proceed halfway to completion: r = 1 / t0.5 Rate increases with temperature according to Arrhenius equation, characteristic for thermally activated processes:
r = A exp (-QA/kT) = A exp (-Qm/ RT)
Per atom Per mole
Percent recrystallization of pure copper at different T
University of Virginia, Dept. of Materials Science and Engineering 5
Introduction to Materials Science, Chapter 10, Phase Transformations in Metals
Superheating / supercooling
! Upon crossing a phase boundary on the compositiontemperature phase diagram phase transformation towards equilibrium state is induced. ! But the transition to the equilibrium structure takes time and transformation is delayed. ! During cooling, transformations occur at temperatures less than predicted by phase diagram: supercooling. ! During heating, transformations occur at temperatures greater than predicted by phase diagram: superheating. ! Degree of supercooling/superheating increases with rate of cooling/heating. ! Metastable states can be formed as a result of fast temperature change. Microstructure is strongly affected by the rate of cooling. ! Below we will consider the effect of time on phase transformations using iron-carbon alloy as an example.
University of Virginia, Dept. of Materials Science and Engineering
Introduction to Materials Science, Chapter 10, Phase Transformations in Metals
Let us consider eutectoid reaction as an example eutectoid reaction: (0.76 wt% C) (0.022 wt% C) + Fe3C
The S-shaped curves are shifted to longer times at higher T showing that the transformation is dominated by nucleation (nucleation rate increases with supercooling) and not by University of occurs faster at higher T).diffusion (whichVirginia, Dept. of
Materials Science and Engineering7
Introduction to Materials Science, Chapter 10, Phase Transformations in Metals
Isothermal Transformation (or TTT) Diagrams (Temperature, Time, and % Transformation)
University of Virginia, Dept. of Materials Science and Engineering
Introduction to Materials Science, Chapter 10, Phase Transformations in Metals
TTT Diagrams
Austenite (stable)
Eutectoid temperature
ferrite
Coarse pearlite
Fe3C Fine pearlite
Austenite pearlite transformation
Denotes that a transformation is occurring
The thickness of the ferrite and cementite layers in pearlite is ~ 8:1. The absolute layer thickness depends on the temperature of the transformation.The higher the temperature, the thicker the layers.
University of Virginia, Dept. of Materials Science and Engineering
Introduction to Materials Science, Chapter 10, Phase Transformations in Metals
TTT Diagrams
! The family of S-shaped curves at different T are used to construct the TTT diagrams. ! The TTT diagrams are for the isothermal (constant T) transformations (material is cooled quickly to a given temperature before the transformation occurs, and then keep it at that temperature). ! At low temperatures, the transformation occurs sooner (it is controlled by the rate of nucleation) and grain growth (that is controlled by diffusion) is reduced. ! Slow diffusion at low temperatures leads to fine-grained microstructure with thin-layered structure of pearlite (fine pearlite). ! At higher temperatures, high diffusion rates allow for larger grain growth and formation of thick layered structure of pearlite (coarse pearlite). ! At compositions other than eutectoid, a proeutectoid phase (ferrite or cementite) coexist with pearlite. Additional curves for proeutectoid transformation must be included on TTT diagrams.
University of Virginia, Dept. of Materials Science and Engineering
10
Introduction to Materials Science, Chapter 10, Phase Transformations in Metals
Formation of Bainite Microstructure (I)
If transformation temperature is low enough (540C) bainite rather than fine pearlite forms.
University of Virginia, Dept. of Materials Science and Engineering 11
Introduction to Materials Science, Chapter 10, Phase Transformations in Metals
Formation of Bainite Microstructure (II)
! For T ~ 300-540C, upper bainite consists of needles of ferrite separated by long cementite particles ! For T ~ 200-300C, lower bainite consists of thin plates of ferrite containing very fine rods or blades of cementite ! In the bainite region, transformation rate is controlled by microstructure growth (diffusion) rather than nucleation. Since diffusion is slow at low temperatures, this phase has a very fine (microscopic) microstructure. ! Pearlite and bainite transformations are competitive; transformation between pearlite and bainite not possible without first reheating to form austenite
Upper bainite
Lower bainite
University of Virginia, Dept. of Materials Science and Engineering
12
Introduction to Materials Science, Chapter 10, Phase Transformations in Metals
Spheroidite
Annealing of pearlitic or bainitic microstructures at elevated temperatures just below eutectoid (e.g. 24 h at 700 C) leads to the formation of new microstructure spheroidite - spheres of cementite in a ferrite matrix. Composition or relative amounts of ferrite and cementite are not changing in this transformation, only shape of the cementite inclusions is changing. Transformation proceeds by C diffusion needs high T. Driving force for the trancsformation - reduction in total ferrite - cementite boundary area
University of Virginia, Dept. of Materials Science and Engineering
13
Introduction to Materials Science, Chapter 10, Phase Transformations in Metals
Martensite (I)
Martensite forms when austenite is rapidly cooled (quenched) to room T. It forms nearly instantaneously when the required low temperature is reached. The austenite-martensite does not involve diffusion no thermal activation is needed, this is called an athermal transformation. Each atom displaces a small (sub-atomic) distance to transform FCC -Fe (austenite) to martensite which has a Body Centered Tetragonal (BCT) unit cell (like BCC, but one unit cell axis is longer than the other two). Martensite is metastable - can persist indefinitely at room temperature, but will transform to equilibrium phases on annealing at an elevated temperature. Martensite can coexist with other phases and/or microstructures in Fe-C system Since martensite is metastable non-equilibrium phase, it does not appear in phase Fe-C phase diagram
University of Virginia, Dept. of Materials Science and Engineering
14
Introduction to Materials Science, Chapter 10, Phase Transformations in Metals
TTT Diagram including Martensite
A: Austenite B: Bainite
P: Pearlite M: Martensite
Austenite-to-martensite is diffusionless and very fast. The amountUniversity of Virginia,formed depends onand Engineering only.of martensite Dept. of Materials Science temperature15
Introduction to Materials Science, Chapter 10, Phase Transformations in Metals
Time-temperature path microstructure
University of Virginia, Dept. of Materials Science and Engineering
16
Introduction to Materials Science, Chapter 10, Phase Transformations in Metals
Mechanical Behavior of Fe-C Alloys (I)
Cementite is harder and more brittle than ferrite increasing cementite fraction therefore makes harder, less ductile material.
University of Virginia, Dept. of Materials Science and Engineering
17
Introduction to Materials Science, Chapter 10, Phase Transformations in Metals
Mechanical Behavior of Fe-C Alloys (II)
The strength and hardness of the different microstructures is inversely related to the size of the microstructures (fine structures have more phase boundaries inhibiting
dislocation motion).
Mechanical properties of bainite, pearlite, spheroidite Considering microstructure we can predict that ! Spheroidite is the softest ! Fine pearlite is harder and stronger than coarse pearlite ! Bainite is harder and stronger than pearlite
Mechanical properties of martensite Of the various microstructures in steel alloys ! Martensite is the hardest, strongest and the most brittle The strength of martensite is not related to microstructure. Rather, it is related to the interstitial C atoms hindering dislocation motion (solid solution hardening, Chapter 7)
and to the small number of slip systems.
University of Virginia, Dept. of Materials Science and Engineering 18
Introduction to Materials Science, Chapter 10, Phase Transformations in Metals
Mechanical Behavior of Fe-C Alloys (III)
University of Virginia, Dept. of Materials Science and Engineering
19
Introduction to Materials Science, Chapter 10, Phase Transformations in Metals
Tempered Martensite (I) Martensite is so brittle that it needs to be modified for practical applications. This is done by heating it to 250-650 oC for some time (tempering) which produces tempered martensite, an extremely fine-grained and well dispersed cementite grains in a ferrite matrix. ! Tempered martensite is less hard/strong as compared to regular martensite but has enhanced ductility (ferrite phase is ductile). ! Mechanical properties depend upon cementite particle size: fewer, larger particles means less boundary area and softer, more ductile material eventual limit is spheroidite. ! Particle size increases with higher tempering temperature and/or longer time (more C diffusion) - therefore softer, more ductile material.
University of Virginia, Dept. of Materials Science and Engineering
20
Introduction to Materials Science, Chapter 10, Phase Transformations in Metals
Tempered Martensite (II)
Higher temperature & time: spheroidite (soft)
Electron micrograph of tempered martensite
University of Virginia, Dept. of Materials Science and Engineering 21
Introduction to Materials Science, Chapter 10, Phase Transformations in Metals
Summary of austenite transformations
Austenite
Slow cooling Moderate cooling
Rapid quench
Pearlite ( + Fe3C) + a proeutectoid phase
Bainite ( + Fe3C)
Martensite (BCT phase) Reheat
Tempered martensite ( + Fe3C)
Solid lines are diffusional transformations, dashed is diffusionless martensitic transformation
University of Virginia, Dept. of Materials Science and Engineering 22
Introduction to Materials Science, Chapter 10, Phase Transformations in Metals
Summary
Make sure you understand language and concepts: ! Alloy steel ! Athermal transformation ! Bainite ! Coarse pearlite ! Fine pearlite ! Isothermal transformation diagram ! Kinetics ! Martensite ! Nucleation ! Phase transformation ! Plain carbon steel ! Spheroidite ! Supercooling ! Superheating ! Tempered martensite ! Thermally activated transformation ! Transformation rate
University of Virginia, Dept. of Materials Science and Engineering
23
Introduction to Materials Science, Chapter 10, Phase Transformations in Metals
Reading for next class:
Chapter 11: Thermal Processing of Metal Alloys ! Process Annealing, Stress Relief ! Heat Treatment of Steels ! Precipitation Hardening
University of Virginia, Dept. of Materials Science and Engineering
24
Vous aimerez peut-être aussi
- Chapter Outline:: Heat Treatment (And Temperature)Document24 pagesChapter Outline:: Heat Treatment (And Temperature)DSGPas encore d'évaluation
- TTT 11Document55 pagesTTT 11MrDOTPas encore d'évaluation
- Fe CDocument34 pagesFe CZaza ArifinPas encore d'évaluation
- Iron-Carbon Phase Diagram (A Review) See Callister Chapter 9Document34 pagesIron-Carbon Phase Diagram (A Review) See Callister Chapter 9Zefa Erliana YullahPas encore d'évaluation
- Fe-C Phase DiagramDocument34 pagesFe-C Phase DiagramYoung-long Choi100% (1)
- TTT N AvramiDocument40 pagesTTT N AvramiAmirul AminPas encore d'évaluation
- Phase Transformation in Metals and AlloysDocument31 pagesPhase Transformation in Metals and AlloysSyafiqah RusdiPas encore d'évaluation
- EMAT 10 (2k21)Document41 pagesEMAT 10 (2k21)Kumail AbbasPas encore d'évaluation
- Kinetics and Heat Treatments in Metals: The IT TTT Diagram For Eutectoid SteelDocument8 pagesKinetics and Heat Treatments in Metals: The IT TTT Diagram For Eutectoid Steelkatchani123Pas encore d'évaluation
- 03 - Iron - Iron CarbideDocument35 pages03 - Iron - Iron CarbidebotobotoakbarPas encore d'évaluation
- Phase Transformation in Metals: Dr. Aneela WakeelDocument29 pagesPhase Transformation in Metals: Dr. Aneela WakeelmazharPas encore d'évaluation
- University of Kirkuk. Department of Mechanical Enginnering.: Name: Ahmed QaisDocument2 pagesUniversity of Kirkuk. Department of Mechanical Enginnering.: Name: Ahmed QaisMostafa HamawandyPas encore d'évaluation
- TTT CurvesDocument4 pagesTTT Curvesmanas310jntuhPas encore d'évaluation
- 02.Iron-Phase DiagramDocument21 pages02.Iron-Phase Diagrampopular102001Pas encore d'évaluation
- 8.heat TreatmentDocument7 pages8.heat Treatmentrohan_n_desai100% (2)
- TTT Phase DiagramDocument9 pagesTTT Phase Diagramhari krishnaPas encore d'évaluation
- Publication 4 11889 199Document9 pagesPublication 4 11889 199Mulia AridhoPas encore d'évaluation
- Computational Materials Science: R. Mahnken, A. Schneidt, S. Tschumak, H.J. MaierDocument7 pagesComputational Materials Science: R. Mahnken, A. Schneidt, S. Tschumak, H.J. MaierJames HaidoPas encore d'évaluation
- Time Temperature Transformation (TTT) Diagrams PDFDocument108 pagesTime Temperature Transformation (TTT) Diagrams PDFSerkan Apay100% (1)
- Fe CdiagramDocument36 pagesFe CdiagramGeorge SingerPas encore d'évaluation
- Heat Treatment 1Document42 pagesHeat Treatment 1Akash TiwariPas encore d'évaluation
- Time-Temperature-Transformation (TTT) DiagramDocument18 pagesTime-Temperature-Transformation (TTT) DiagramGikiTopiPas encore d'évaluation
- 08 Transformasi Fasa - UASDocument35 pages08 Transformasi Fasa - UASDinta PratiwiPas encore d'évaluation
- Material and Heat Treatment 3Document5 pagesMaterial and Heat Treatment 3lastking_king17Pas encore d'évaluation
- TTT DiagramDocument6 pagesTTT DiagramDeepa PujariPas encore d'évaluation
- TTT DiagramDocument17 pagesTTT DiagramGAURAV SINGHPas encore d'évaluation
- 3 Fe-Fe3C Phase DiagramDocument33 pages3 Fe-Fe3C Phase DiagramRajat Mishra100% (1)
- TTT DiagramDocument31 pagesTTT DiagramEducated SmugglerPas encore d'évaluation
- FALLSEM2019-20 MEE1005 ETH VL2019201001078 Reference Material I 29-Aug-2019 Fe-Fe3C Phase DiagramDocument33 pagesFALLSEM2019-20 MEE1005 ETH VL2019201001078 Reference Material I 29-Aug-2019 Fe-Fe3C Phase DiagramFazal KhanPas encore d'évaluation
- TTT and CCT Continuous Cooling TransformationDocument6 pagesTTT and CCT Continuous Cooling TransformationurcojPas encore d'évaluation
- Phase Transformation in Metals1Document3 pagesPhase Transformation in Metals1sridharan.engineer01Pas encore d'évaluation
- Review MetalDocument38 pagesReview MetalnisannisaPas encore d'évaluation
- TTTDocument7 pagesTTTMuhammad Hery Al-GhiffaryPas encore d'évaluation
- TTT and CCT DiagramDocument24 pagesTTT and CCT DiagramArun Raj A C100% (2)
- Microstructure Properties: II Martensitic TransformationsDocument38 pagesMicrostructure Properties: II Martensitic TransformationsRecep VatanseverPas encore d'évaluation
- Platinum Alloys For Shape Memory ApplicationsDocument15 pagesPlatinum Alloys For Shape Memory ApplicationsKursalPas encore d'évaluation
- Kinetics - Heat TreatmentDocument18 pagesKinetics - Heat TreatmentUnknownPas encore d'évaluation
- Heat Treatment of 1045 Steel PDFDocument17 pagesHeat Treatment of 1045 Steel PDFH_DEBIANEPas encore d'évaluation
- ME 216 - Engineering Materials II: Heat Treatment (Part I)Document15 pagesME 216 - Engineering Materials II: Heat Treatment (Part I)ozanPas encore d'évaluation
- Time Temperature Transformation With ReferncesDocument12 pagesTime Temperature Transformation With ReferncesEllie BrooklynPas encore d'évaluation
- Phase Transformations: Ferrite - BCCDocument65 pagesPhase Transformations: Ferrite - BCCAhsan JunaidPas encore d'évaluation
- Iron Carbon Phase DiagramDocument4 pagesIron Carbon Phase DiagramMizanur RahmanPas encore d'évaluation
- The Role of Manganese and Copper in The Eutectoid Transformation of Spheroidal Graphite Cast IronDocument11 pagesThe Role of Manganese and Copper in The Eutectoid Transformation of Spheroidal Graphite Cast IronChanthar SoePas encore d'évaluation
- Deformation-Induced Transformation of Retained Austenite in Transformation Induced Plasticity-Aided Steels: A Thermodynamic ModelDocument10 pagesDeformation-Induced Transformation of Retained Austenite in Transformation Induced Plasticity-Aided Steels: A Thermodynamic ModelAhmed El-SaiedPas encore d'évaluation
- Microstructure-Properties: II Martensitic TransformationsDocument38 pagesMicrostructure-Properties: II Martensitic TransformationsnotsofarPas encore d'évaluation
- Materials Science & Engineering A: SciencedirectDocument11 pagesMaterials Science & Engineering A: SciencedirectDaniel TibataPas encore d'évaluation
- Iron Carbon DiagramDocument43 pagesIron Carbon Diagramhrpatel_165Pas encore d'évaluation
- Ch-8 Compatibility ModeDocument58 pagesCh-8 Compatibility Modedreamgurl9011Pas encore d'évaluation
- Sample Questions For Phase DiagramDocument5 pagesSample Questions For Phase DiagramMohaiminul Islam TalhaPas encore d'évaluation
- DivaCalista 1806187902 CCT&TTTdiagramsDocument7 pagesDivaCalista 1806187902 CCT&TTTdiagramsAlishia LoshPas encore d'évaluation
- Trans FasesDocument6 pagesTrans FasesIveth Carmona GonzalezPas encore d'évaluation
- Trans FasesDocument6 pagesTrans FasesIveth Carmona GonzalezPas encore d'évaluation
- Time-Temperature-Transformation Diagram Within The Bainitic Temperature Range in A Medium Carbon SteelDocument10 pagesTime-Temperature-Transformation Diagram Within The Bainitic Temperature Range in A Medium Carbon SteelAnonymous Fty6OOHlPas encore d'évaluation
- Dataset We UsedDocument11 pagesDataset We UsedDev Raj SoniPas encore d'évaluation
- An Overview On Bainite Formation in SteelsDocument10 pagesAn Overview On Bainite Formation in SteelssajadPas encore d'évaluation
- FeC and TTT DiagramsDocument12 pagesFeC and TTT DiagramsMohamed El-WakilPas encore d'évaluation
- Thermomechanical Processing of High-Strength Low-Alloy SteelsD'EverandThermomechanical Processing of High-Strength Low-Alloy SteelsPas encore d'évaluation
- Materials Data for Cyclic Loading: Aluminium and Titanium AlloysD'EverandMaterials Data for Cyclic Loading: Aluminium and Titanium AlloysÉvaluation : 1 sur 5 étoiles1/5 (1)
- Sundram FastenersDocument4 pagesSundram Fastenersmpcd07Pas encore d'évaluation
- ST Lawrence Ultra-MetDocument6 pagesST Lawrence Ultra-Metmpcd07Pas encore d'évaluation
- Technical Comparision - BoilerDocument2 pagesTechnical Comparision - Boilermpcd07Pas encore d'évaluation
- Boa PromisesbibleDocument26 pagesBoa Promisesbiblempcd07Pas encore d'évaluation
- Upload A Document For Free Download Access.: Select Files From Your Computer or Choose Other Ways To Upload BelowDocument5 pagesUpload A Document For Free Download Access.: Select Files From Your Computer or Choose Other Ways To Upload Belowmpcd07Pas encore d'évaluation
- Get Free Access To This Document by Contributing To Scribd: NTN Handbook of Roller BearingDocument2 pagesGet Free Access To This Document by Contributing To Scribd: NTN Handbook of Roller Bearingmpcd07Pas encore d'évaluation
- Objective Chemistry For ..Document3 pagesObjective Chemistry For ..mpcd070% (1)
- This Document by Contributing To Scribd: 5S ModulDocument2 pagesThis Document by Contributing To Scribd: 5S Modulmpcd07Pas encore d'évaluation
- Objective Chemistry For ..Document3 pagesObjective Chemistry For ..mpcd070% (1)
- 1 150 - Questions Paper I 2012 TetDocument5 pages1 150 - Questions Paper I 2012 Tetmpcd07Pas encore d'évaluation
- Get Free Access To This Document by Contributing To Scribd: Six Sigma, Kaizen & 5SDocument2 pagesGet Free Access To This Document by Contributing To Scribd: Six Sigma, Kaizen & 5Smpcd07Pas encore d'évaluation
- Get Free Access To This Document by Contributing To Scribd: MuthuswamyDocument2 pagesGet Free Access To This Document by Contributing To Scribd: Muthuswamympcd07Pas encore d'évaluation
- Get Free Access To This Document by Contributing To Scribd: QSP-211-02 3C 5S Improvment SheetDocument2 pagesGet Free Access To This Document by Contributing To Scribd: QSP-211-02 3C 5S Improvment Sheetmpcd07Pas encore d'évaluation
- This Document by Contributing To Scribd: Upload NowDocument2 pagesThis Document by Contributing To Scribd: Upload Nowmpcd07Pas encore d'évaluation
- This Document by Contributing To Scribd: Spoken English To TamilDocument1 pageThis Document by Contributing To Scribd: Spoken English To Tamilmpcd07Pas encore d'évaluation
- Get Free Access To This Document by Contributing To Scribd: Kenneth E. Hagin The Art of PrayerDocument1 pageGet Free Access To This Document by Contributing To Scribd: Kenneth E. Hagin The Art of Prayermpcd07Pas encore d'évaluation
- 1 PDFDocument1 page1 PDFmpcd07Pas encore d'évaluation
- 1 PDFDocument1 page1 PDFmpcd07Pas encore d'évaluation
- Get Free Access To This Document by Contributing To Scribd: Bible StoriesDocument2 pagesGet Free Access To This Document by Contributing To Scribd: Bible Storiesmpcd07Pas encore d'évaluation
- Get Free Access To This Document by Contributing To Scribd: (2) Weld Deposit CalculationDocument1 pageGet Free Access To This Document by Contributing To Scribd: (2) Weld Deposit Calculationmpcd07Pas encore d'évaluation
- Related Keywords: 1,689,989 People Like This. To See What Your Friends LikeDocument11 pagesRelated Keywords: 1,689,989 People Like This. To See What Your Friends Likempcd07Pas encore d'évaluation
- 1 PDFDocument6 pages1 PDFmpcd07Pas encore d'évaluation
- Chief Educational Officer: RecentDocument2 pagesChief Educational Officer: Recentmpcd07Pas encore d'évaluation
- This Document by Contributing To Scribd: Bible StoriesDocument2 pagesThis Document by Contributing To Scribd: Bible Storiesmpcd07Pas encore d'évaluation
- Regnet Research Paper SeriesDocument7 pagesRegnet Research Paper Seriesnqdpuhxgf100% (1)
- Vogue 201602 Japan PDFDocument254 pagesVogue 201602 Japan PDFJack Douglas YooPas encore d'évaluation
- Kraken Intelligence's Taproot Primer - An Upgrade For The AgesDocument37 pagesKraken Intelligence's Taproot Primer - An Upgrade For The AgesT. CC ElficPas encore d'évaluation
- Falcon 4 CLHS Cameras: True High-Performance: High-Speed Global Shutter CMOS CamerasDocument4 pagesFalcon 4 CLHS Cameras: True High-Performance: High-Speed Global Shutter CMOS CamerasEkpetorson ChinweikePas encore d'évaluation
- Ubd Planning Template With QuestionsDocument3 pagesUbd Planning Template With Questionsapi-217297849Pas encore d'évaluation
- The Relationship Between Emotional Maturity and Psychosocial Adjustment Among First-Year Undergraduate Students in Amhara Region Public Universities, EthiopiaDocument11 pagesThe Relationship Between Emotional Maturity and Psychosocial Adjustment Among First-Year Undergraduate Students in Amhara Region Public Universities, EthiopiaYared FentawPas encore d'évaluation
- Mothballing Requires More Than Idle ThoughtDocument4 pagesMothballing Requires More Than Idle Thoughtfawmer61Pas encore d'évaluation
- Coding in Communication System: Channel Coding) Will Be AddressedDocument5 pagesCoding in Communication System: Channel Coding) Will Be AddressedDuge PlakolliPas encore d'évaluation
- AC Induction Motors - How AC Motors WorkDocument10 pagesAC Induction Motors - How AC Motors WorkBraulio IrrutiaPas encore d'évaluation
- The Whys and Wherefores of Microphone Array Crosstalk in Multichannel Microphone Array DesignDocument6 pagesThe Whys and Wherefores of Microphone Array Crosstalk in Multichannel Microphone Array DesignmewarulesPas encore d'évaluation
- Alarm ListDocument4 pagesAlarm ListMirela Perenda ŠabićPas encore d'évaluation
- WJMDocument6 pagesWJMAldianivo Nur FaizPas encore d'évaluation
- Netxknx Opc 3 - 5 - 4 EngDocument93 pagesNetxknx Opc 3 - 5 - 4 Engtai_sao_khong_1307Pas encore d'évaluation
- WDM Bi553Document138 pagesWDM Bi553florea_madfoxPas encore d'évaluation
- Whinner y 1990Document5 pagesWhinner y 1990LGPas encore d'évaluation
- FELPRO - Tablas-de-Torque - 035Document1 pageFELPRO - Tablas-de-Torque - 035Clodoaldo BiassioPas encore d'évaluation
- AssociationBetweenBRAFV600EMutationand MortalityDocument9 pagesAssociationBetweenBRAFV600EMutationand MortalityMade RusmanaPas encore d'évaluation
- Grammar 111Document33 pagesGrammar 111Suntithvibol Sem100% (1)
- V-Ray For SketchUp Rendering An Exterior Scene PDFDocument7 pagesV-Ray For SketchUp Rendering An Exterior Scene PDFDevohPas encore d'évaluation
- Direct Synthesis of Sodalite From Kaolin: The Influence of AlkalinityDocument7 pagesDirect Synthesis of Sodalite From Kaolin: The Influence of AlkalinityIsye RahmaenaPas encore d'évaluation
- Chapter Five Decision Theories and AnalysisDocument47 pagesChapter Five Decision Theories and AnalysisAbdela AyalewPas encore d'évaluation
- CNS - Types of CiphersDocument47 pagesCNS - Types of Ciphersmahesh palemPas encore d'évaluation
- Data Rate Limits: Dept. of Computer Engineering Faculty of EngineeringDocument31 pagesData Rate Limits: Dept. of Computer Engineering Faculty of Engineeringআসিফ রেজাPas encore d'évaluation
- Chapter 8Document7 pagesChapter 8Maiane JunqueiraPas encore d'évaluation
- Operating and Maintenance Manual 322375H01 Rev CDocument82 pagesOperating and Maintenance Manual 322375H01 Rev CGustavo BarbosaPas encore d'évaluation
- Clipping Clamping CircuitsDocument7 pagesClipping Clamping CircuitsPavan SaiPas encore d'évaluation
- USN 18CS654: B. E. Degree (Autonomous) Sixth Semester End Examination (SEE)Document2 pagesUSN 18CS654: B. E. Degree (Autonomous) Sixth Semester End Examination (SEE)Sarmi HarshaPas encore d'évaluation
- Chapter 3 Ni ChupaghettiDocument2 pagesChapter 3 Ni ChupaghettiRodemar RosarioPas encore d'évaluation
- 2020 Specimen Paper 1 Mark SchemeDocument16 pages2020 Specimen Paper 1 Mark SchemesarabPas encore d'évaluation
- Flue Gas Analysis in IndustryDocument150 pagesFlue Gas Analysis in Industryblack_knight728Pas encore d'évaluation