Académique Documents
Professionnel Documents
Culture Documents
Selden
Transféré par
Maham Faisal Khan0 évaluation0% ont trouvé ce document utile (0 vote)
31 vues9 pagesTwo highly organized and discrete bundles of cholinergic fibres extended from the nucleus basalis to the cerebral cortex and amygdala. The medial pathway joined the white matter of the gyrus rectus, while the lateral pathway joined the cerebral cortex.
Description originale:
Copyright
© © All Rights Reserved
Formats disponibles
PDF, TXT ou lisez en ligne sur Scribd
Partager ce document
Partager ou intégrer le document
Avez-vous trouvé ce document utile ?
Ce contenu est-il inapproprié ?
Signaler ce documentTwo highly organized and discrete bundles of cholinergic fibres extended from the nucleus basalis to the cerebral cortex and amygdala. The medial pathway joined the white matter of the gyrus rectus, while the lateral pathway joined the cerebral cortex.
Droits d'auteur :
© All Rights Reserved
Formats disponibles
Téléchargez comme PDF, TXT ou lisez en ligne sur Scribd
0 évaluation0% ont trouvé ce document utile (0 vote)
31 vues9 pagesSelden
Transféré par
Maham Faisal KhanTwo highly organized and discrete bundles of cholinergic fibres extended from the nucleus basalis to the cerebral cortex and amygdala. The medial pathway joined the white matter of the gyrus rectus, while the lateral pathway joined the cerebral cortex.
Droits d'auteur :
© All Rights Reserved
Formats disponibles
Téléchargez comme PDF, TXT ou lisez en ligne sur Scribd
Vous êtes sur la page 1sur 9
Brain (1998), 121, 22492257
Trajectories of cholinergic pathways within the
cerebral hemispheres of the human brain
Nathan R. Selden,
1,2
Darren R. Gitelman,
1
Noriko Salamon-Murayama,
1
Todd B. Parrish
1
and
M.-Marsel Mesulam
1
1
Cognitive Neurology and Alzheimer Disease Center, Correspondence to: M.-Marsel Mesulam, Cognitive
Northwestern University Medical School, Chicago, Illinois Neurology and Alzheimer Disease Center, Northwestern
and
2
Section of Neurosurgery, Department of Surgery, University, 320 E. Superior Ave, Searle 11-453, Chicago,
University of Michigan Medical Center, Ann Arbor, IL 60611, USA. E-mail: mmesulam@nwu.edu
Michigan, USA
Summary
All sectors of the human cerebral cortex receive dense
cholinergic input. The origin of this projection is located
in the Ch4 cell group of the nucleus basalis of Meynert.
However, very little is known about the location of the
pathways which link the cholinergic neurons of the nucleus
basalis to the human cerebral cortex. This question was
addressed in whole-hemisphere sections processed for the
visualization of multiple cholinergic markers. Two highly
organized and discrete bundles of cholinergic bres
extended from the nucleus basalis to the cerebral cortex
and amygdala and were designated as the medial and
lateral cholinergic pathways. These bundles contained
acetylcholinesterase, choline acetyltransferase and nerve
growth factor receptors, conrming their cholinergic
nature and origin within the basal forebrain. The medial
pathway joined the white matter of the gyrus rectus,
Keywords: nucleus basalis; cerebral hemispheres; choline acetyltransferase; nerve growth factor receptor;
acetylcholinesterase
Abbreviations: ACh acetylcholine; AChE acetylcholinesterase; ChAT choline acetyltransferase; nbM nucleus
basalis of Meynert; NGFr nerve growth factor receptor
Introduction
Magnocellular neurons of the primate basal forebrain provide
a massive and widespread cholinergic input to the
telencephalon and inuence nearly all aspects of cortical
function, especially in the domains of attention, memory and
emotion (Mesulam, 1996; Everitt and Robbins, 1997). These
projections originate from the four overlapping Ch1Ch4 cell
groups of the basal forebrain. Cholinergic neurons of the
medial septal nucleus (also known as the Ch1 cell group)
and the vertical limb nucleus of the diagonal band (Ch2)
provide the major cholinergic input of the hippocampus;
cholinergic neurons of the horizontal limb nucleus of the
diagonal band (Ch3) provide the major cholinergic input of
the olfactory bulb; and cholinergic neurons of the nucleus
Oxford University Press 1998
curved around the rostrum of the corpus callosum to
enter the cingulum and merged with bres of the lateral
pathway within the occipital lobe. It supplied the
parolfactory, cingulate, pericingulate and retrosplenial
cortices. The lateral pathway was subdivided into a
capsular division travelling in the white matter of the
external capsule and uncinate fasciculus and a perisylvian
division travelling within the claustrum. Branches of the
perisylvian division supplied the frontoparietal
operculum, insula and superior temporal gyrus. Branches
of the capsular division innervated the remaining parts
of the frontal, parietal and temporal neocortex.
Representation of these cholinergic pathways within a 3D
MRI volume helped to identify white matter lesion sites
that could interfere with the corticopetal ow of
cholinergic pathways.
basalis of Meynert (nbMCh4) provide the principal
cholinergic input of the remaining cerebral cortex and
amygdala (Mesulam et al., 1983; Everitt et al., 1988; Alonso
et al., 1996).
Basal forebrain cholinergic neurons are the only CNS
neurons that continue to express high levels of the p75 nerve
growth factor receptor (NGFr) during adulthood (Hefti et al.,
1986; Mesulamet al., 1992b). The presence of NGFr therefore
leads to the inference that a cholinergic axon originates
within the Ch1Ch4 complex of the basal forebrain (Heckers
et al., 1994). The neuroanatomy, cytochemistry, connectional
topography and cortical distribution of cholinergic pathways
originating in the basal forebrain have been investigated in
2250 N. R. Selden et al.
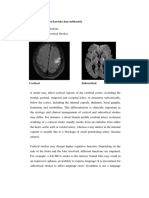
Fig. 1 (A) AChE histochemistry in the insula demonstrates a dense network of AChE-rich cholinergic axons. (B) Photomacrograph of
the region of the external capsule and claustrum revealing dense AChE reaction product over the putamen (P) and a thick network of
AChE-rich bres in the insular cortex (IN). The two components of the lateral cholinergic pathway are shown by the arrowhead
(perisylvian division) and the double arrowhead (capsular division). The microscopic elds shown in (C) and (D) include the part of the
capsular division located at the tip of the double arrowhead in (B) but immunostained in adjacent sections for ChAT in (C) and NGFr in
(D). The correspondence of the AChE-rich bre bundle with ChAT- and NGFr-immunoreactive bres demonstrates that the pathway is
denitively cholinergic and that it originates in the Ch14 cells of the NbM. ec external capsule; LII, LIII cortical layers II and III;
P putamen. Medial is towards the left and dorsal towards the top. Magnications: A, 186; B, 11.3; C and D, 371.
Cholinergic bre pathways 2251
Fig. 2 (A) AChE histochemistry in the frontal lobe reveals two tracts of AChE-rich (cholinergic) bres coursing through the white matter
(arrows). The medial pathway (M) is closely associated with the adjacent cingulate gyrus (CG) and rostrum of the corpus callosum (R).
This anatomical section was used to generate the diagram in Fig. 3A. (B) A higher power photomicrograph of the same section
demonstrating individual AChE-rich bres streaming from the lateral pathway to the cortex of the inferior frontal gyrus (arrows). The
arrowheads in (A) and (B) serve as a reference marker for the identical anatomical area. L lateral pathway; OF orbitofrontal cortex.
Magnications: A, 1.56; B, 6.86.
considerable detail in the macaque monkey with the help of
axonally transported tracers, enzyme assays and
immunohistochemistry (Mesulam et al., 1983, 1986; Satoh
and Fibiger, 1985; Everitt et al., 1988; Kordower et al., 1989;
Geula et al., 1993).
The human brain displays an analogous topographic and
cytochemical organization of cholinergic cells in the basal
forebrain, with perhaps an even greater degree of anatomical
differentiation, especially within the nbM (Mesulam and
Geula, 1988). Furthermore, the density of cholinergic axons in
the human cerebral cortex displays orderly regional variations
which obey the systems-level organization of information
processing streams (Mesulam et al., 1992a). Although direct
axonal tracing experiments are not possible in the human
brain, the study of patients with Alzheimers disease suggests
that the topography of the projections connecting the Ch1
Ch4 cell groups to the cerebral cortex is similar to that
identied in the monkey (Mesulam and Geula, 1988).
Some of the cholinergic pathways emanating from the
basal forebrain travel within easily identiable bre tracks.
For example, cholinergic pathways extending from Ch12 to
the hippocampus travel predominantly within the fornix
(Green and Mesulam, 1988; Alonso et al., 1996), those that
reach the olfactory bulb within the olfactory tract (Kitt et al.,
1987; Luiten et al., 1987) and those to the amygdala within
the ventral amygdalofugal pathway and the stria terminalis
(Carlsen et al., 1985; Heckers and Mesulam, 1994). The
trajectories of connections between the nbM and the cerebral
cortex are less well understood.
Using injections of tritiated amino acids into the midportion
of the nbM and surrounding structures in rhesus monkeys,
Kitt et al. (1987) described a medial projection to the
cingulate cortex that travelled within the cingulum, a lateral
projection to the frontoparietal and insular cortices that
travelled within the external and extreme capsules and a
ventral projection to the temporal cortex and the amygdala
that travelled within the uncinate fasciculus. However, these
studies did not include specic histochemical or immuno-
histochemical markers to ascertain the cholinergic nature of
the pathways. There is currently no information about the
white matter trajectory of the pathways that connect the
cholinergic Ch4 neurons of the nbM with the cerebral cortex
in the human brain. The purpose of the present report is to
investigate the organization of these pathways with the help
of selective cholinergic markers.
Material and methods
Subjects
More than 50 human brains processed for the visualization
of cholinergic markers in whole-hemisphere sections were
examined. Five representative specimens were chosen for
this study. They were among the optimally stained specimens
but did not differ in anatomical detail from the others. The
specimens used in this study came from subjects with no
neurological or psychiatric disease as determined by clinical
records. Their ages ranged from27 to 101 years and three were
2252 N. R. Selden et al.
female. Although cortical cholinergic innervation displays a
minor age-related decline in density (Geula and Mesulam,
1989), the trajectories of the pathways do not change with
age. The post-mortem autolysis interval was between 4 and
12 h. Cause of death included myocardial infarction (two),
congestive heart failure, cardiorespiratory arrest and gunshot
wound. No specic neuropathological alterations were found
in sections stained with haematoxylineosin and cresyl violet.
Thioavin S histouorescence showed some neuritic plaques
and neurobrillary tangles in brains from the oldest subjects,
but in a density and distribution that were belowthe diagnostic
thresholds for Alzheimers disease (Mirra et al., 1991).
Histological procedures
Slabs of whole cerebral hemispheres were xed, cryo-
protected and sectioned coronally at a thickness of 40 m,
as described previously (Mesulam et al., 1991). A set of
Cholinergic bre pathways 2253
matching sections through each brain was stained with cresyl
violet for cytoarchitectonic identication and acetylcholin-
esterase histochemistry using a modication of the direct
colouring KarnovskyRoots method (Tago et al., 1986;
Mesulam et al., 1987). Adjacent sections were processed
immunohistochemically using a monospecic, polyclonal
antibody raised against human placental choline acetyl-
transferase (ChAT, generously donated by Lewis B. Hersh)
and a monoclonal antibody raised against human NGFr
(generously donated by Mark Bothwell) (German et al.,
1985; Marano et al., 1987).
The immunohistochemical procedure was based on
formation of the avidinbiotin complex and used
diaminobenzidine as the chromogen (Mesulam et al., 1991).
In selected sections, additional intensication of the reaction
product was obtained using silver nitrate (Kitt et al., 1988).
For control sections, an irrelevant IgG was substituted for
the primary antibody.
Data analysis
This study is conned to the cholinergic projections that
emanate from the nbM. The more fully investigated
hippocampal, olfactory and amygdaloid cholinergic pathways
(which travel in the fornix, olfactory tract, stria terminalis
and ventral amygdalofugal pathway) will not be addressed.
Acetylcholinesterase (AChE) histochemistry provided the
most intense staining and was used as the principal method
for illustrating the trajectory of cholinergic pathways. The
pattern of this staining was compared with that obtained with
ChAT and NGFr immunohistochemistry to ascertain that
AChE could be used as a specic marker of cholinergic
pathways. The location, course and density of AChE-rich
bres in six representative sections spanning the rostrocaudal
extent of the cerebral hemispheres were then charted using
a plotter electronically coupled to the stage of a compound
microscope. These chartings were imported into the
Photoshop graphics program (Adobe Systems Incorporated,
San Jose, Calif., USA) for differential coloration of pathways.
Fig. 3 Electronic plotter charts of AChE-rich (cholinergic) bre bundles in the hemispheric white matter of six coronal sections. A is
most rostral and F is most caudal. The medial cholinergic pathway is represented in green, and the two divisions of the lateral pathway
in red (capsular division) and orange (perisylvian division). The cholinergic Ch4 neurons of the nucleus basalis of Meynert (nbM) are
represented in blue. (A) The medial pathway remains medial to the rostrum of the corpus callosum (R) and gives off bres to the
cingulate gyrus (CG) and medial frontal cortex, whereas the lateral pathway courses within the white matter of the frontal lobe and gives
off bres to the dorsal and lateral prefrontal cortex. The orbitofrontal cortex receives bres from both the medial and lateral cholinergic
bundles. (B) The medial pathway courses anteriorly under the corpus callosum in the white matter of the parolfactory gyrus (PG) and,
having curved up around the rostrum at more anterior levels, courses posteriorly in the cingulum (Ci), dorsal to the corpus callosum. The
two lateral components course within the external capsule (red) and the claustrum (orange). (C and D) The lateral and medial pathways
emanate from the nbMCh4. The components of the lateral pathway extend dorsally into frontoparietal cortex and ventrally into the
temporal lobe. (E and F) The medial pathway continues its caudad course in the cingulum and curves around the splenium (Sp) to
supply the cingulate and pericingulate cortices. The lateral pathway courses in the form of discrete bundles within the deep white matter
of the parietal, temporal and occipital lobes and gives off AChE-rich bres to adjacent cortical areas. Magnication, 1.5. ac anterior
commissure; Am amygdala; C caudate; CaS calcarine sulcus; CC corpus callosum; CeS central sulcus; CiS cingulate
sulcus; CoS collateral sulcus; dg dentate gyrus; En, entorhinal cortex; GP globus pallidus; H hippocampus; IN insula;
mb mammillary body; P putamen; SF Sylvian ssure; Th thalamus; TP temporal pole; V ventricle.
Three-dimensional imaging
A contiguous, 1 mm, T
1
-weighted MRI volume (TR 15
ms, TE 6 ms, ip angle 20, FOV 165 220 mm
with a 192 256 matrix) was obtained in the coronal plane
froma neurologically normal 62-year-old right-handed female
using a 1.5 Tesla scanner (Siemens Vision, Erlangen,
Germany). The exact plane of image acquisition was
determined to match the plane of section in the anatomical
reference cases. The MRI slices were then exported into
Photoshop and the principal cholinergic pathways were traced
based on their location in matching anatomical sections. Only
dense cholinergic bre bundles coursing within the white
matter were represented. The entire volume was then
reconstructed within AVS (Applied Visual Systems, Waltham,
Mass., USA) in order to enable the visualization of these
pathways in additional planes of section.
Results
All regions of the cerebral cortex displayed a dense net of
AChE-rich bres (Fig. 1A). The white matter contained
AChE-rich bre bundles (Fig. 1B). Adjacent sections showed
these bres to be both ChAT- and NGFr-immunoreactive,
providing denitive conrmation that they are cholinergic
(because of the ChAT) and that they originate in the basal
forebrain (because of the NGFr) (Fig. 1C and D). Individual
AChE-rich axons emanated from these cholinergic bundles
and fanned towards the cerebral cortex (Fig. 2). Cholinergic
bre bundles originated from the nbMCh4 region and
formed a medial and a lateral pathway (Fig. 3). The lateral
pathway was further subdivided into a capsular component
travelling within the external capsule and a perisylvian
component travelling within the claustrum.
The medial cholinergic pathway emanated anteriorly from
the nbMCh4 and joined the white matter of the gyrus
rectus (parolfactory gyrus) and medial orbital gyrus, passed
anteriorly to the rostrum of the corpus callosum, entered the
cingulum, travelled posteriorly to the splenium and curved
ventrally to enter the retrosplenial white matter. Individual
2254 N. R. Selden et al.
Fig. 4 The medial and lateral cholinergic pathways are represented within a volume-rendered MRI format, enabling their visualization in
planes other than that of the anatomical sections. As in Fig. 3, the medial pathway is shown in green and the lateral pathway in red
(capsular division) and orange (perisylvian division). The nbMCh4 is shown in blue (arrowheads). Only the dense bundles within white
matter are included. The ne bres which take off from these bundles to innervate adjacent areas of cortex are not shown in the interest
of clarity. The trajectories shown in this gure also help to identify white matter sites where lesions (for example vascular) could
interrupt the more distal ow of cortical cholinergic pathways. (A) and (B) Horizontal sections at the level of the frontal horns of the
lateral ventricles and the upper level of the corpus callosum, respectively. (C) Parasagittal view obtained 4 mm lateral to the
interhemispheric ssure. The medial pathway originates in the anterior nbMCh4 (arrowhead) and courses around the corpus callosum,
predominantly within in the cingulum. (D) Intersection of a coronal section at the level of the posterior border of the thalamus and a
horizontal section at the level of the anterior commissure. The arrowhead points to the nbMCh4 neurons which give rise to the
corticopetal cholinergic pathways. ac anterior commissure; C caudate; CC corpus callosum; CeS central sulcus; CiS
cingulate sulcus; IPS intraparietal sulcus; OF orbitofrontal gyri; P putamen; R rostrum of corpus callosum; SF Sylvian
ssure; Sp splenium of corpus callosum; Th thalamus.
Cholinergic bre pathways 2255
axons radiated from this pathway to supply medial
orbitofrontal, subcallosal, cingulate, pericingulate and
retrosplenial cortices (Figs 2 and 3).
The capsular division of the lateral cholinergic pathway
coursed in a densely packed and highly organized bundle of
bres immediately adjacent to the putamen in the medial
aspect of the external capsule (Figs 1B and 3). Additional
bres travelled ventrally in the white matter of the uncinate
fasciculus, adjacent to the fundus of the putamen and the
amygdala (Fig. 3). Some of these bres appeared to penetrate
the substance of the amygdala while others continued in a
dense bundle into the white matter of the temporal lobe.
Individual bres radiated from the capsular division to supply
the dorsal frontoparietal neocortex, the middle and inferior
temporal gyri, the inferotemporal cortex and the
parahippocampal gyrus (Fig. 3).
The perisylvian division of the lateral cholinergic pathway
coursed within the claustrum, curving laterally into the white
matter of the inferior frontal and superior temporal gyri.
Individual bres radiated from the perisylvian division to
supply the frontoparietal opercular cortices, superior temporal
gyrus and the insula (Fig. 3).
The medial and lateral cholinergic pathways merged
anteriorly in the white matter of the orbitofrontal gyri and
posteriorly in the occipital lobe beneath the optic radiations
(Fig. 3).
The manual reconstruction of anatomically identied
cholinergic pathways within corresponding MRI sections
allowed a 3D reconstruction of their distribution within the
human cerebral hemispheres (Fig. 4). The entire curved
trajectory of the medial cholinergic pathway was visualized
in a single parasagittal plane of section (Fig. 4C).
Representation of cholinergic pathways within axial sections,
which are commonly used in clinical studies, showed that
these pathways course through regions of white matter that
are frequently involved in many different disease processes,
including cerebrovascular and demyelinating diseases and
neoplasm (Fig. 4A and B).
Discussion
The neurons of nbMCh4 are selectively responsive to novel
and motivationally relevant sensory events (Wilson and Rolls,
1990; Morris et al., 1997). Novel and emotionally salient
events are therefore particularly effective in eliciting the
release of acetylcholine (ACh) in the cerebral cortex. The
major effect of ACh upon neurons of the cerebral cortex is
mediated through the m1 subtype of muscarinic receptors
and causes a prolonged reduction of potassium conductance
so as to make cortical neurons more receptive to other
excitatory inputs (Sato et al., 1987; McCormick, 1990).
Cortical cholinergic pathways also promote long-term
potentiation and experience-induced synaptic remodelling
(Huerta and Lisman, 1993; Cruikshank and Weinberger, 1996;
Baskerville et al., 1997). The cholinergic pathway from the
basal forebrain to the cerebral cortex is therefore in a position
to promote the impact and memorability of novel and
motivationally relevant events. The degeneration of nbM
Ch4 neurons observed in various neurological diseases,
including Alzheimers disease and Parkinsons disease, results
in a depletion of cortical cholinergic innervation and
undoubtedly contributes to the altered mental state that is
observed in these conditions (Geula and Mesulam, 1994).
Although cholinergic projections reach all areas of the
cerebral cortex and are generally described as diffuse, our
studies demonstrate that they travel in discrete, organized
bundles. The medial cholinergic pathway travels within
the cingulum and supplies the orbitofrontal, subcallosal,
cingulate, pericingulate and retrosplenial cortices. The lateral
cholinergic pathway includes a perisylvian division, which
courses within the claustrum to supply opercular and insular
cortices, and a capsular division, which courses within the
external capsule and uncinate fasciculus to supply remaining
areas of the cerebral cortex. Our ndings in the human brain
are generally in concordance with those in the rhesus monkey
(Kitt et al., 1987) and provide additional histochemical and
immunohistochemical validation concerning the cholinergic
nature of these pathways. Such specic validation is important
since the nbM also contains noncholinergic neurons which
project to the cerebral cortex (Gritti et al., 1993).
The discrete topography of cholinergic pathways within the
cerebral hemispheres suggests that corticopetal cholinergic
neurotransmission may be vulnerable to focal pathological
insults of the white matter even when the nbM itself remains
intact. The representation of cholinergic pathways within 3D
MRI volumes allows the identication of the lesion sites that
are most likely to interrupt these pathways. Cingulotomly is
occasionally performed to treat intractable depression or
obsessivecompulsive disorders (Cosgrove and Ballantine,
1998). One unexpected consequence of this procedure may
be to cause a cholinergic denervation of cingulate and
pericingulate cortices caudal to the lesion site.
The centrum semiovale, the external capsule and claustrum
are commonly involved in cerebrovascular disease. Such
lesions could cause a widespread disconnection of remote
cortical areas from their source of cholinergic innervation in
the basal forebrain. The resulting cholinergic denervation
could contribute to the emergence of mental state
impairments. Multi-infarct dementia and Alzheimers disease
may therefore share a common neurochemical pattern of
cortical cholinergic depletion through completely different
mechanisms. The ascending cholinergic axons of the nucleus
basalis are mostly unmyelinated (Wainer and Mesulam,
1990). It therefore remains to be seen if demyelinating
diseases such as multiple sclerosis can also damage the
white matter pathways which carry cholinergic bres to the
cerebral cortex.
Investigation of the human brain is progressing rapidly,
especially as a consequence of remarkably powerful methods
related to functional imaging. However, delineating the
anatomy of neural connections in humans continues to pose
serious challenges. Our results demonstrate that transmitter-
2256 N. R. Selden et al.
specic anatomical markers in post-mortem specimens may
make useful contributions to this area of inquiry.
Acknowledgements
N.R.S. wishes to thank Dr Julian Hoff for advice and
encouragement. This study was supported in part by an
Alzheimers Disease Center Grant (1-P30 AG13854) and
NS20285.
References
Alonso JR, UHS, Amaral DG. Cholinergic innervation of the primate
hippocampal formation: II. Effects of mbria/fornix transection. J
Comp Neurol 1996; 375: 52751.
Baskerville KA, Schweitzer JB, Herron P. Effects of cholinergic
depletion on experience-dependent plasticity in the cortex of the
rat. Neuroscience 1997; 80: 115969.
Carlsen J, Zaborszky L, Heimer L. Cholinergic projections from
the basal forebrain to the basolateral amygdaloid complex: a
combined retrograde uorescent and immunohistochemical study. J
Comp Neurol 1985; 234: 15567.
Cosgrove GR, Ballantine HT. Cingulotomy in psychosurgery. In:
Gildenberg PL, Tasker RR, editors. Textbook of stereotactic and
functional neurosurgery. New York: McGrawHill; 1998. p.
196570.
Cruikshank SJ, Weinberger NM. Evidence for the Hebbian
hypothesis in experience-dependent physiological plasticity of
neocortex: a critical review. [Review]. Brain Res Brain Res Rev
1996; 22: 191228.
Everitt BJ, Robbins TW. Central cholinergic systems and cognition.
Annu Rev Psychol 1997; 48: 64984.
Everitt BJ, Sirkia TE, Roberts AC, Jones GH, Robbins TW.
Distribution and some projections of cholinergic neurons in the
brain of the common marmoset, Callithrix jacchus. J Comp Neurol
1988; 271: 53358.
German DC, Bruce G, Hersh LB. Immunohistochemical staining
of cholinergic neurons in the human brain using a polyclonal
antibody to human choline acetyltransferase. Neurosci Lett 1985;
61: 15.
Geula C, Mesulam MM. Cortical cholinergic bers in aging and
Alzheimers disease: a morphometric study. Neuroscience 1989; 33:
46981.
Geula C, Mesulam M-M. Cholinergic systems and related
neuropathological predilection patterns in Alzheimer disease. In:
Terry RD, Katzman R, Bick KL, editors. Alzheimer disease. New
York: Raven Press; 1994. p. 26394.
Geula C, Schatz CR, Mesulam MM. Differential localization of
NADPH-diaphorase and calbindin-D28k within the cholinergic
neurons of the basal forebrain, striatum and brainstem in the rat,
monkey, baboon and human. Neuroscience 1993; 54: 46176.
Green RC, Mesulam MM. Acetylcholinesterase ber staining in the
human hippocampus and parahippocampal gyrus. J Comp Neurol
1988; 273: 48899.
Gritti I, Mainville L, Jones BE. Codistribution of GABA with
acetylcholine-synthesizing neurons in the basal forebrain of the rat.
J Comp Neurol 1993; 329: 43857.
Heckers S, Mesulam M-M. Two types of cholinergic projections to
the rat amygdala. Neuroscience 1994; 60: 38397.
Heckers S, Ohtake T, Wiley RG, Lappi DA, Geula C, Mesulam M-
M. Complete and selective cholinergic denervation of rat neocortex
and hippocampus but not amygdala by an immunotoxin against the
p75 NGF receptor. J Neurosci 1994; 14: 127189.
Hefti F, Hartikka J, Salvatierra A, Weiner WJ, Mash DC. Localization
of nerve growth factor receptors in cholinergic neurons of the
human basal forebrain. Neurosci Lett 1986; 69: 3741.
Huerta PT, Lisman JE. Heightened synaptic plasticity of
hippocampal CA1 neurons during a cholinergically induced rhythmic
state. Nature 1993; 364: 7235.
Kitt CA, Mitchell SJ, DeLong MR, Wainer BH, Price DL. Fiber
pathways of basal forebrain cholinergic neurons in monkeys. Brain
Res 1987; 406: 192206.
Kitt CA, Levey AI, Friedman DP, Walker LC, Koliatsos VE, Raskin
LS, et al. Immunocytochemical visualization of cholinergic pathways
in monkey neocortex using silver nitrate [abstract]. Soc Neurosci
Abstr 1988; 14: 631.
Kordower JH, Bartus RT, Marciano FF, Gash DM. Telencephalic
cholinergic system of the New World monkey (Cebus apella):
morphological and cytoarchitectonic assessment and analysis of the
projection to the amygdala. J Comp Neurol 1989; 279: 52845.
Luiten PG, Gaykema RP, Traber J, Spencer DGJr. Cortical projection
patterns of magnocellular basal nucleus subdivisions as revealed by
anterogradely transported Phaseolus vulgaris leucoagglutinin. Brain
Res 1987; 413: 22950.
Marano N, Dietzschold B, Early JJ Jr, Schatteman G, Thompson S,
Grob P, et al. Purication and amino terminal sequence of human
melanoma nerve growth factor receptor. J Neurochem 1987; 48:
22532.
McCormick DA. Cellular mechanisms of cholinergic control of
neocortical and thalamic neuronal excitability. In: Steriade M,
Biesold D, editors. Brain cholinergic systems. Oxford: Oxford
University Press; 1990. p. 23664.
Mesulam M-M. The systems-level organization of cholinergic
innervation in the cerebral cortex and its alterations in Alzheimers
disease. [Review]. Prog Brain Res 1996; 109: 28598.
Mesulam M-M, Geula C. Nucleus basalis (Ch4) and cortical
cholinergic innervation in the human brain: observations based on
the distribution of acetylcholinesterase and choline acetyltransferase.
J Comp Neurol 1988; 275: 21640.
Mesulam M-M, Mufson EJ, Levey AI, Wainer BH. Cholinergic
innervation of cortex by the basal forebrain: cytochemistry and
cortical connections of the septal area, diagonal band nuclei, nucleus
basalis (substantia innominata), and hypothalamus in the rhesus
monkey. J Comp Neurol 1983; 214: 17097.
Mesulam M-M, Volicer L, Marquis JK, Mufson EJ, Green RC.
Systematic regional differences in the cholinergic innervation of the
primate cerebral cortex: distribution of enzyme activities and some
behavioral implications. Ann Neurol 1986; 19: 14451.
Cholinergic bre pathways 2257
Mesulam M-M, Geula C, Moran MA. Anatomy of cholinesterase
inhibition in Alzheimers disease: effect of physostigmine and
tetrahydroaminoacridine on plaques and tangles. Ann Neurol 1987;
22: 68391.
Mesulam M-M, Geula C, Cosgrove R, Mash D, Brimijoin S.
Immunocytochemical demonstration of axonal and perikaryal
acetylcholinesterase in human cerebral cortex. Brain Res 1991; 539:
2338.
Mesulam M-M, Hersh LB, Mash DC, Geula C. Differential
cholinergic innervation within functional subdivisions of the human
cerebral cortex: a choline acetyltransferase study. J Comp Neurol
1992a; 318: 31628.
Mesulam M-M, Mash D, Hersh L, Bothwell M, Geula C. Cholinergic
innervation of the human striatum, globus pallidus, subthalamic
nucleus, substantia nigra, and red nucleus. J Comp Neurol 1992b;
323: 25268.
Mirra SS, Heyman A, McKeel D, Sumi SM, Crain BJ, Brownlee
LM, et al. The Consortium to Establish a Registry for Alzheimers
Disease (CERAD). Part III. Standardization of the neuropathologic
assessment of Alzheimers disease. Neurology 1991; 41: 47986.
Morris JS, Friston KJ, Dolan RJ. Neural responses to salient visual
stimuli. Proc R Soc (Lond) B Biol Sci 1997; 264: 76975.
Sato H, Hata V, Hagihara K, Tsumoto T. Effects of cholinergic
depletion on neuron activities in the cat visual cortex. J Neurophysiol
1987; 58: 78194.
Satoh K, Fibiger HC. Distribution of central cholinergic neurons in
the baboon (Papio papio). I. General morphology. J Comp Neurol
1985; 236: 197214.
Tago H, Kimura H, Maeda T. Visualization of detailed
acetylcholinesterase ber and neuron staining in rat brain by a
sensitive histochemical procedure. J Histochem Cytochem 1986;
34: 14318.
Wainer BH, Mesulam M-M. Ascending cholinergic pathways in the
rat brain. In: Steriade M, Biesold D, editors. Brain cholinergic
systems. Oxford: Oxford University Press; 1990. p. 65119.
Wilson FA, Rolls ET. Neuronal responses related to the novelty and
familiarity of visual stimuli in the substantia innominata, diagonal
band of Broca and periventricular region of the primate basal
forebrain. Exp Brain Res 1990; 80: 10420.
Received 5 June, 1998. Revised July 27, 1998.
Accepted July 28, 1998
Vous aimerez peut-être aussi
- Cellular Dynamics of the Neuron: Symposia of the International Society for Cell Biology, Vol. 8D'EverandCellular Dynamics of the Neuron: Symposia of the International Society for Cell Biology, Vol. 8Pas encore d'évaluation
- Cookson 1996Document16 pagesCookson 1996M4shroomPas encore d'évaluation
- Chat 5HT Brain Stem Mouse AnatomyDocument35 pagesChat 5HT Brain Stem Mouse AnatomyRam KPas encore d'évaluation
- Hornung 2010Document14 pagesHornung 2010Leandy MartinezPas encore d'évaluation
- Carr Central ProjectionsDocument13 pagesCarr Central ProjectionsMengda ZhangPas encore d'évaluation
- Rewiew FINAL 1Document13 pagesRewiew FINAL 1achalbob18Pas encore d'évaluation
- Mesopontine Organization of Cholinergic and Catecholaminergic Cell Groups in The Normal and Narcoleptic DogDocument13 pagesMesopontine Organization of Cholinergic and Catecholaminergic Cell Groups in The Normal and Narcoleptic DogAndjela RoganovicPas encore d'évaluation
- Mufson 1989Document12 pagesMufson 1989Majd HusseinPas encore d'évaluation
- Agenesis of The Corpus Callosum - Genetic, Developmental and Functional Aspects of ConnectivityDocument13 pagesAgenesis of The Corpus Callosum - Genetic, Developmental and Functional Aspects of ConnectivityLoreto Opazo RojasPas encore d'évaluation
- Lateralization of Olfactory ProcessesDocument16 pagesLateralization of Olfactory Processesmiskica miskicaPas encore d'évaluation
- Artigo Human Cingulate SulciDocument8 pagesArtigo Human Cingulate SulciandremvcPas encore d'évaluation
- Miller DiekerDocument8 pagesMiller DiekerJucsy JuxyPas encore d'évaluation
- Lavdas Et Al. - J.neurosc. - 1999 - The MGE Gives Rise To A Population of Early Neurons in The Developing Cerebral CortexDocument8 pagesLavdas Et Al. - J.neurosc. - 1999 - The MGE Gives Rise To A Population of Early Neurons in The Developing Cerebral CortexCris AdFPas encore d'évaluation
- Human Cingulate and Paracingulate Sulci: Pattern, Variability, Asymmetry, and Probabilistic MapDocument8 pagesHuman Cingulate and Paracingulate Sulci: Pattern, Variability, Asymmetry, and Probabilistic MapFrontiersPas encore d'évaluation
- Anatomy of The Mental and Neurobehavior DisordersDocument57 pagesAnatomy of The Mental and Neurobehavior DisordersiqiqiqiqiqPas encore d'évaluation
- Nafisa HDocument17 pagesNafisa Hiromini uthmanPas encore d'évaluation
- Neuroanatomic Connectivity of The Human Ascending ArousalDocument16 pagesNeuroanatomic Connectivity of The Human Ascending ArousalBritany SantanaPas encore d'évaluation
- 2012 Cerebellar Ataxia - Pathophysiology and RehabilitationDocument22 pages2012 Cerebellar Ataxia - Pathophysiology and RehabilitationJefe de Escuela PsicologiaPas encore d'évaluation
- Matsuyama 2004Document11 pagesMatsuyama 2004Rafael BarataPas encore d'évaluation
- Thoracic and Prothoracic Leg Neuromuscular System of The Praying Mantid, Sphodromantis Lineola (Burmeister)Document14 pagesThoracic and Prothoracic Leg Neuromuscular System of The Praying Mantid, Sphodromantis Lineola (Burmeister)JoPas encore d'évaluation
- Petrides, Projections To The Frontal Cortex From The Posterior Parietal Region in The Rhesus Monkey, 1984Document12 pagesPetrides, Projections To The Frontal Cortex From The Posterior Parietal Region in The Rhesus Monkey, 1984Vitor ParentePas encore d'évaluation
- Cereb. Cortex 2003 Duan 950 61Document12 pagesCereb. Cortex 2003 Duan 950 61AlfonsoVillarroelCalistoPas encore d'évaluation
- Alcantara Et Al. - Cerebral Cortex - 2005 - BDNF-modulated Spatial Organization of CR Cells and GABAergic Neurons in The Marginal Zone Plays A Role in The Development of Cortical OrganizationDocument13 pagesAlcantara Et Al. - Cerebral Cortex - 2005 - BDNF-modulated Spatial Organization of CR Cells and GABAergic Neurons in The Marginal Zone Plays A Role in The Development of Cortical OrganizationCris AdFPas encore d'évaluation
- Olfactory Bulbectomy Induces Neuronal Rearrangement in The Entorhinal Cortex in The RatDocument7 pagesOlfactory Bulbectomy Induces Neuronal Rearrangement in The Entorhinal Cortex in The RatJesusPas encore d'évaluation
- Rūta Jurgaitienė Et Al - Morphometric Study of Age-Related Changes in The Human Intracardiac GangliaDocument8 pagesRūta Jurgaitienė Et Al - Morphometric Study of Age-Related Changes in The Human Intracardiac GangliaLuammmPas encore d'évaluation
- Cfos, Jun y ExploracionDocument12 pagesCfos, Jun y ExploracionLuisa Fernanda Acevedo ReyesPas encore d'évaluation
- Broadman 47 Dan AutisDocument4 pagesBroadman 47 Dan AutisSf AkhadiyatiPas encore d'évaluation
- Altmann 2001Document36 pagesAltmann 2001zsmm1993Pas encore d'évaluation
- Cerebelo y AlzheimerDocument11 pagesCerebelo y AlzheimersantiagoPas encore d'évaluation
- Human Brainstem: Cytoarchitecture, Chemoarchitecture, MyeloarchitectureD'EverandHuman Brainstem: Cytoarchitecture, Chemoarchitecture, MyeloarchitecturePas encore d'évaluation
- Cholinesterases: A Histochemical Contribution to the Solution of Some Functional ProblemsD'EverandCholinesterases: A Histochemical Contribution to the Solution of Some Functional ProblemsPas encore d'évaluation
- Ivry1988 - Article - DissociationOfTheLateralAndMed N PDFDocument14 pagesIvry1988 - Article - DissociationOfTheLateralAndMed N PDFFábio SagginPas encore d'évaluation
- Oorschot 1996Document20 pagesOorschot 1996M4shroomPas encore d'évaluation
- Hearing Research: Hisayuki Ojima, Kunio MurakamiDocument8 pagesHearing Research: Hisayuki Ojima, Kunio MurakamiFrontiersPas encore d'évaluation
- Neuropsych Studies of The Frontal LobesDocument26 pagesNeuropsych Studies of The Frontal Lobesстас кшижевскийPas encore d'évaluation
- Neural Network Model of The Cerebellum: Temporal Discrimination and The Timing of Motor ResponsesDocument18 pagesNeural Network Model of The Cerebellum: Temporal Discrimination and The Timing of Motor ResponsespharaohshiPas encore d'évaluation
- Review: Evolution of Vertebrates As Viewed From The CrestDocument9 pagesReview: Evolution of Vertebrates As Viewed From The Crestrocambolescas perthPas encore d'évaluation
- International Standards For Neurological and Functional Classification of Spinal Cord InjuryDocument14 pagesInternational Standards For Neurological and Functional Classification of Spinal Cord InjuryVijay IyerPas encore d'évaluation
- Neuroanatomy of The Zebrafish Brain - A Topological Atlas 1996Document141 pagesNeuroanatomy of The Zebrafish Brain - A Topological Atlas 1996Daniel RuizPas encore d'évaluation
- 2003 Structure and Innervation of The CochleaDocument26 pages2003 Structure and Innervation of The CochleaCaio LeônidasPas encore d'évaluation
- Schmidt 1996Document8 pagesSchmidt 1996M4shroomPas encore d'évaluation
- How Do You Feel?: An Interoceptive Moment with Your Neurobiological SelfD'EverandHow Do You Feel?: An Interoceptive Moment with Your Neurobiological SelfÉvaluation : 4.5 sur 5 étoiles4.5/5 (3)
- Oral Session I Which Theory Do The Structural Brain Changes Support?Document2 pagesOral Session I Which Theory Do The Structural Brain Changes Support?antonioopPas encore d'évaluation
- Central Tegmental TractDocument46 pagesCentral Tegmental TractjovanaPas encore d'évaluation
- Anatomy of Neuropsychiatry: The New Anatomy of the Basal Forebrain and Its Implications for Neuropsychiatric IllnessD'EverandAnatomy of Neuropsychiatry: The New Anatomy of the Basal Forebrain and Its Implications for Neuropsychiatric IllnessPas encore d'évaluation
- Cerebellar Output Motor and Cognitive Channels - TiDocument7 pagesCerebellar Output Motor and Cognitive Channels - Tivikhen0712Pas encore d'évaluation
- PNAS2011 Petanjek NEOTsynaptsDocument6 pagesPNAS2011 Petanjek NEOTsynaptsAlejandraPas encore d'évaluation
- CerebellumDocument13 pagesCerebellumNejla HPas encore d'évaluation
- Atrophy of The Parietal Lobe in Preclinical DementiaDocument10 pagesAtrophy of The Parietal Lobe in Preclinical DementiaNícolas CavalcantePas encore d'évaluation
- Thehumancerebellum: A Review of Physiologic NeuroanatomyDocument11 pagesThehumancerebellum: A Review of Physiologic NeuroanatomyCan EkerPas encore d'évaluation
- Articulo Neuro 1Document12 pagesArticulo Neuro 1Victor CuellarPas encore d'évaluation
- Aumann 1996Document12 pagesAumann 1996M4shroomPas encore d'évaluation
- This Content Downloaded From 200.24.17.12 On Mon, 15 Feb 2021 17:04:11 UTCDocument7 pagesThis Content Downloaded From 200.24.17.12 On Mon, 15 Feb 2021 17:04:11 UTCIsabella Urrea CardonaPas encore d'évaluation
- Bruner 2021Document19 pagesBruner 2021Manuel CuminiPas encore d'évaluation
- Sonic HedgehogDocument11 pagesSonic HedgehogPaola Rivera ÁvilaPas encore d'évaluation
- Ni Hms 393042Document19 pagesNi Hms 393042Mutiara Tri JunisaPas encore d'évaluation
- Serotonin Neurons, Neuroplasticity, and Homeostasis of Neural TissueDocument13 pagesSerotonin Neurons, Neuroplasticity, and Homeostasis of Neural TissueFuyumi RiaPas encore d'évaluation
- HHS Public Access: Spatial and Memory Circuits in The Medial Entorhinal CortexDocument14 pagesHHS Public Access: Spatial and Memory Circuits in The Medial Entorhinal CortexVictor CarrenoPas encore d'évaluation
- Mishkin 1983 PDFDocument4 pagesMishkin 1983 PDFamandaPas encore d'évaluation
- Nureal Control and CoordinationDocument16 pagesNureal Control and CoordinationthushyanthPas encore d'évaluation
- Summative Test in Science 6Document2 pagesSummative Test in Science 6Rox Francisco100% (3)
- CBR PHD Courses 2022Document8 pagesCBR PHD Courses 2022ShwePas encore d'évaluation
- Kerala University of Health Sciences: Bachelor Degree Course in PHARMACY (B.Pharm)Document65 pagesKerala University of Health Sciences: Bachelor Degree Course in PHARMACY (B.Pharm)Mukesh TiwariPas encore d'évaluation
- Macroeconomics 9th Edition Boyes Test BankDocument25 pagesMacroeconomics 9th Edition Boyes Test BankMadisonKirbyeqko100% (54)
- Chapter 2Document35 pagesChapter 2Amiey SehokPas encore d'évaluation
- Week 11 Content List AND ActivityDocument3 pagesWeek 11 Content List AND ActivityKaitlyn RobisonPas encore d'évaluation
- REMEDIAL - Grade 5Document2 pagesREMEDIAL - Grade 5shamshad0% (1)
- Brain CancerDocument74 pagesBrain CancerDan EvanzPas encore d'évaluation
- B.pharma SyllabusDocument100 pagesB.pharma SyllabusRupali GulyaniPas encore d'évaluation
- Approach To The Patient With Difficult in Walking (Hemiplegia)Document78 pagesApproach To The Patient With Difficult in Walking (Hemiplegia)Ruby BhattyPas encore d'évaluation
- Neuro Lesson 8Document11 pagesNeuro Lesson 8api-287575009Pas encore d'évaluation
- Hydrocephalus in Adult PresentDocument25 pagesHydrocephalus in Adult PresentmarthintoriPas encore d'évaluation
- Nervous System LecDocument26 pagesNervous System LecLuningning Nobleza100% (3)
- Bioethics SeminarDocument1 pageBioethics Seminarapi-24500288Pas encore d'évaluation
- MRCP 1 NeurologyDocument16 pagesMRCP 1 NeurologyKarrar A. AlShammaryPas encore d'évaluation
- T V NG TACN1Document18 pagesT V NG TACN1Thanh ToànPas encore d'évaluation
- CSF RhinorrheaDocument34 pagesCSF RhinorrheaAnoop SinghPas encore d'évaluation
- SSC CGL 2 PDFDocument56 pagesSSC CGL 2 PDFKiranPas encore d'évaluation
- ANP1106 ASyllabus 2020Document5 pagesANP1106 ASyllabus 2020유우솔Pas encore d'évaluation
- TRANSCALLOSAL TRANSCHOROIDAL APPROACH TO Third VentriclDocument6 pagesTRANSCALLOSAL TRANSCHOROIDAL APPROACH TO Third VentriclNikolay PeevPas encore d'évaluation
- BIO 3350 - Lec 1 History of NeurosciencesDocument36 pagesBIO 3350 - Lec 1 History of NeurosciencesSimon HagosPas encore d'évaluation
- BIOS5130 Week 10 Lecture Part 1 SlidesDocument37 pagesBIOS5130 Week 10 Lecture Part 1 SlidesOkikiola JohnsonPas encore d'évaluation
- Nervous System WorksheetDocument2 pagesNervous System WorksheetRoda Aranquez RabinoPas encore d'évaluation
- Perbedaan Lesi Korteks Dan SubkortekDocument5 pagesPerbedaan Lesi Korteks Dan SubkortekRisa SahiraPas encore d'évaluation
- Radio - Child Abuse - TdcedenoDocument28 pagesRadio - Child Abuse - TdcedenoDenise CedeñoPas encore d'évaluation
- System: Gigi Luk, Christos Pliatsikas, Eleonora RossiDocument12 pagesSystem: Gigi Luk, Christos Pliatsikas, Eleonora RossiSebatian ospinaPas encore d'évaluation
- CAPITIN - AnSci1.1 Anatomy and Physiology of Farm AnimalsDocument6 pagesCAPITIN - AnSci1.1 Anatomy and Physiology of Farm AnimalsJuan CapitinPas encore d'évaluation