Académique Documents
Professionnel Documents
Culture Documents
00001
Transféré par
Heaven Singh0 évaluation0% ont trouvé ce document utile (0 vote)
19 vues10 pagespaper
Titre original
00043764-200805000-00001
Copyright
© © All Rights Reserved
Formats disponibles
PDF, TXT ou lisez en ligne sur Scribd
Partager ce document
Partager ou intégrer le document
Avez-vous trouvé ce document utile ?
Ce contenu est-il inapproprié ?
Signaler ce documentpaper
Droits d'auteur :
© All Rights Reserved
Formats disponibles
Téléchargez comme PDF, TXT ou lisez en ligne sur Scribd
0 évaluation0% ont trouvé ce document utile (0 vote)
19 vues10 pages00001
Transféré par
Heaven Singhpaper
Droits d'auteur :
© All Rights Reserved
Formats disponibles
Téléchargez comme PDF, TXT ou lisez en ligne sur Scribd
Vous êtes sur la page 1sur 10
Options for Occupational Health Surveillance
of Workers Potentially Exposed to Engineered
Nanoparticles: State of the Science
Paul A. Schulte, PhD
Douglas Trout, MD
Ralph D. Zumwalde, MS
Eileen Kuempel, PhD
Charles L. Geraci, PhD
Vincent Castranova, PhD
Diane J. Mundt, PhD
Kenneth A. Mundt, PhD
William E. Halperin, MD
Objective: Health authorities, employers, and worker representatives
are increasingly faced with making decisions about occupational health
surveillance of workers potentially exposed to engineered nanoparticles.
This article was developed to identify options that can be considered.
Methods: The published scientific literature on health effects from
engineered and incidental nanoparticles and the principles of occupa-
tional health surveillance were reviewed to describe possible options and
the evidence base for them. Results: Various options for occupational
health surveillance were identified. The options ranged from no action
targeted to nanotechnology workers to an approach that includes
documentation of the presence of engineered nanoparticles, identifica-
tion of potentially exposed workers, and general and targeted medical
testing. Conclusions: Although the first priority should be to implement
appropriate primary preventive measures, additional efforts to monitor
employee health may be warranted. Continued research is needed, and
the collection of such information for exposure registries may be useful
for future epidemiologic studies. (J Occup Environ Med. 2008;50:
517526)
T
he question of what type of occupa-
tional health surveillance is appropri-
ate for workers potentially exposed
to engineered nanoparticles is of in-
creasing importance. This is because
of a growing, but not definitive, body
of evidence for potential health haz-
ards of some types of engineered nano-
particles and the fact that increasing
numbers of workers are currently han-
dling them.
19
These exposures can
occur in research laboratories, and in
operations where engineered nano-
particles are manufactured, used, and
disposed.
4
The development and pro-
liferation of new engineered nano-
particles can be expected to continue
to outpace health researchers ability
to evaluate the potential health con-
sequences. Consequently, health au-
thorities, employers, occupational
health professionals, and workers are
faced with determining appropriate
strategies for occupational health
surveillance in the face of uncer-
tainty about the exposure and hazard
from handling engineered nanopar-
ticles. The current dearth of regula-
tory clarity further contributes to the
ambiguity surrounding what devel-
opers and producers of engineered
nanoparticles should consider doing
to assure that their employees are
protected. Although implementing
appropriate engineering controls, us-
ing effective personal protective
equipment, and establishing safe
handling procedures are paramount,
additional efforts to monitor em-
ployee health may be warranted in
the near future. In this article, we
characterize the range of action options
From the National Institute for Occupational Safety and Health (Drs Schulte, Trout, Kuempel,
Geraci, and Castranova, and Mr Zumwalde), Centers for Disease Control and Prevention, Cincinnati,
Ohio; ENVIRON International Corporation (Dr Mundt), Amherst, Mass; and University of Medicine
and Dentistry of New Jersey (Dr Halperin), NJ.
The findings and conclusions in this report are those of the authors and do not necessarily represent
the views of the National Institute for Occupational Safety and Health.
Address correspondence to: Paul A. Schulte, PhD, National Institute for Occupational Safety and
Health, Centers for Disease Control and Prevention, 4676 Columbia Parkway, MS-C14, Cincinnati, OH
45226-1998; E-mail: pas4@cdc.gov.
Copyright 2008 by American College of Occupational and Environmental Medicine
DOI: 10.1097/JOM.0b013e31816515f7
JOEM
Volume 50, Number 5, May 2008 517
on a continuum of hazard and medical
surveillance. For each action, we de-
scribe the scientific evidence base or
rationale and implications. Because
the body of evidence for potential
hazard related to occupational expo-
sure to engineered nanoparticles is
continually changing, decisions
about the need for and type of occu-
pational health surveillance are
likely to evolve over time.
Occupational Health Surveillance
Occupational health surveillance
is the ongoing systematic collection,
analysis, and dissemination of expo-
sure and health data on groups of
workers for the purpose of early
detection of disease and injury as
well as trends or patterns of occurrence
presumably leading to prevention of
subsequent disease. Occupational
health surveillance can help define
the magnitude and scope of occu-
pational health issues among
groups of workers and guide efforts
to improve worker safety and
health and monitor progress. The
general term occupational health
surveillance includes hazard and
medical surveillance; integration of
hazard and medical surveillance is
key to an effective occupational
health surveillance program.
1015
A substance may be defined as a
hazard if it has the potential to cause
harm under some set of circum-
stances. Hazard surveillance consists
of the periodic characterization of
chemical/physical hazards in the
workplace. Medical surveillance tar-
gets actual health events or a change
in a biologic function in an exposed
person or persons. Medical screening
(monitoring) is one form of medical
surveillance that is designed to detect
early signs of work-related illness by
administering tests to apparently
healthy persons to detect those with
early stages of disease or risk of
disease.
16
Occupational health surveillance
is also conducted for the purpose of
determining whether the hierarchy of
techniques for injury and illness pre-
vention are effective. These tech-
niques can be ordered by primacy
into primary, secondary, and tertiary
prevention.
4,14
Within this hierarchy
are a range of activities from premar-
ket testing for potential toxicity to
preclinical evaluation of exposed
workers; all of which are intended to
prevent long-term impairment and
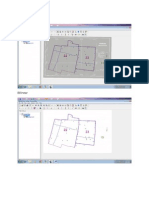
disability (see Fig. 1). Occupational
health surveillance is the process by
which information resulting from
any one of these activities is col-
lected and used to support or alter
what is done at a step higher in the
hierarchy.
Critical in the discussion of occu-
pational health surveillance for
workers potentially exposed to engi-
neered nanoparticles is what triggers
the need or requirement for surveil-
lance. The standard approach to oc-
cupational health surveillance with
known hazards, that is, ones with a
documented evidence base and occu-
pational exposure limits (OELs), is
to utilize the concept of an action
level. The action level represents
some fraction of the OEL, usually
50%, which if exceeded will trigger
various preventive actions including,
in some cases, medical monitoring.
This situation generally does not oc-
cur with engineered nanoparticles
since there is no government or pro-
fessional association OELs specifi-
cally for engineered nanoparticles.
However, workers with airborne ex-
posures to the nanoscale forms of
bulk substances that have OELs will
be subject to the same medical mon-
itoring requirements as those ex-
posed to the bulk form. This is in
contrast to engineered nanoparticles
that either have no established haz-
ard database, OEL, or surveillance
guidelines.
In the absence of OELs and atten-
dant action levels for engineered
nanoparticles, occupational health
surveillance may be considered in
terms of qualitative job hazard expo-
sure analyses. This type of health
surveillance approach has been pio-
neered in the pharmaceutical indus-
try and in the United Kingdom.
17,18
Generally, these approaches are based
on some knowledge of the degree of
hazard. When this is not known, as
with many engineered nanoparticles,
various other approaches may need to
be utilized, for example, by determin-
ing whether toxicity information exists
Fig. 1. The hierarchy of occupational health prevention with examples of surveillance
feedback (Adapted from Am J Ind Med. 1996;29:321323).
518 Health Surveillance and Nanotechnology
Schulte et al
for a similar type of nanoparticle that
can be used as a surrogate for trigger-
ing action.
19
Hazard Potential of
Engineered Nanoparticles
Various factors must be consid-
ered to obtain perspective on the
hazard potential of engineered nano-
particles. First, the types and physi-
cochemical properties of engineered
nanoparticles are vast. The broad
range and combination of parameters
that can influence toxicity, such as
composition, size, shape, surface
coating, charge, attached functional
groups, solubility, contaminants, and
degree of agglomeration, may result
in nanoparticles with large differ-
ences in hazard potential. Second,
much of the scientific evidence on
the potential toxicity of engineered
nanoparticles is based on toxicologi-
cal and epidemiological studies of
certain types of microscale (microme-
ter-scale) and nanoscale (nanometer-
scale) particles, including metal ox-
ides, fumes, and combustion-derived
nanoparticles.
2,8,2022
The term ul-
trafine is frequently used in the
context of nanometer-diameter parti-
cles that have not been intentionally
produced but are the incidental prod-
ucts of processes involving combus-
tion or hot processes. The extent to
which these studies are predictive of
the potential hazard of engineered
nanoparticles will depend on the spe-
cific physicochemical properties of
the engineered particles and on the
exposure conditions.
Potential occupational health risks
associated with manufacturing and
using nanomaterials are not yet well
defined. Many engineered nanomate-
rials and devices are formed from
nanoparticles that are initially pro-
duced as aerosols or colloidal sus-
pensions. Minimal information is
currently available on the potential
for exposure, dominant exposure
routes, and material toxicity. Other
exposures in the workplace where
engineered nanoparticles are handled
(including ambient particulates) may
complicate the measurement of engi-
neered nanoparticles and confound
the association between exposures
and health outcomes that might be
reported. The epidemiological and
toxicological literature on ultrafine
or fine particles and fibers provides
information on the organ systems
and effects that may also be relevant
to clinical evaluations or medical
surveillance of workers exposed to
engineered nanoparticles. However,
engineered nanoparticles may differ
in their physicochemical properties
and may also exhibit enhanced or
unique biological effects related to
increased reactive surface area and
quantum effects.
9,22
Inhalation is generally considered
the primary route of occupational
exposure to nanoparticles. Once in-
haled, a substantial proportion of
nanoparticles are likely to deposit in
the human respiratory tract (30%
90%, depending on breathing rate
and particle size).
23
Nanoparticles
readily reach the deep lung, and the
alveolar region is the primary site of
deposition for nanoparticle sizes in
the 10 to 100 nm range, whereas
head and thoracic deposition pre-
dominate for nanoparticles less than
10 nm. Deposition increases with
exercise and in individuals with
chronic obstructive pulmonary dis-
ease.
2325
Other potential routes of
exposure include passing through the
skin and ingestion. Size and charge
of particles appear to influence both
dermal penetration and gastrointesti-
nal absorption.
2631
Exposures to some types of
nanoscale particles (eg, generated
from combustion or hot processes)
have been associated with adverse
health effects in humans. For ex-
ample, diesel exhaust has been as-
sociated with eye and respiratory
irritation,
32
endothelial dysfunction
(impaired vasodilation) with mild
systemic inflammation,
33
and lung
cancer,
34
while welding fume ex-
posure has been associated with
metal fume fever, susceptibility to
pulmonary infection, obstructive
lung disease, and possible neurologic
changes.
35,36
Cardiovascular system
effects, including markers of blood
coagulation and systemic inflamma-
tion, have been observed in experi-
mental studies of human volunteers
inhaling a low concentration of car-
bon nanoparticles.
24,25,37,38
In the
general population, particulate air
pollution (a mixture that includes
combustion-derived ultrafine parti-
cles) has been associated with in-
creased morbidity and mortality
from adverse respiratory and car-
diovascular effects, primarily
among susceptible populations
such as the elderly or infirm.
3943
No epidemiological studies of oc-
cupational cohorts exposed to engi-
neered nanoparticles have been
published.
In experimental animal studies with
ultrafine and nanoscale particles, the
large surface area of the particles has
been shown to play a key role in
adverse lung effects, including inflam-
mation and tumorigenesis, as does the
surface reactivity, crystallinity, and
biopersistence of the particles in the
lung.
4449
Adverse lung responses ob-
served in humans occupationally ex-
posed to fine and ultrafine particles are
similar to those observed in experi-
mental studies with rodents.
34,50
Translocation is considered more
likely for nanosized particles than for
fine particles. In rodents, nanopar-
ticles have been found to translocate
from the respiratory tract to other
organs, including brain, liver, kidney,
and spleen,
5153
and the rate appears to
depend on the physicochemical prop-
erties of the nanoparticles (eg, size,
chemical composition, shape, and
charge).
5457
In experimental studies
of human volunteers, low-dose,
short-term inhalation of radiolabeled
carbon nanoparticles (Technegas)
showed that most of the deposited
nanoparticles remained in the lungs
up to 2 days after exposure.
58,59
These studies did not find evidence
for the rapid translocation of nano-
particles to the blood circulation or
accumulation in the liver, as had
been reported earlier (possibly due to
JOEM
Volume 50, Number 5, May 2008 519
instability of the radiolabel-nanopar-
ticle complex).
60
Studies of carbon nanotubes in
rodents have shown early-onset of
pulmonary fibrosis, granulomas, and
oxidative stress in the lungs and car-
diovascular system.
5,61 67
Metal
nanoparticles, including cobalt and
nickel caused greater lung inflamma-
tion and injury in rats than microme-
ter-sized particles.
68,69
Liver, kidney,
and spleen were target organs of
copper nanoparticles in mice ex-
posed by oral gavage, causing acute
toxicity, while micrometer-sized
copper did not.
70
As observed in
animal studies with various types of
nanoparticles, contaminants associ-
ated with specific nanoparticles (eg,
metals used as catalysts) may pose a
health risk to workers.
66,67
Continuum of Occupational
Health Surveillance
Like the techniques of prevention
shown in Figure 1, occupational
health surveillance efforts can also
be ordered in a continuum. Because
there is a lack of definitive evi-
dence regarding specific health ef-
fects associated with nanoparticle
exposures, and because engineered
nanoparticles are highly heteroge-
neous, it is difficult to identify an
appropriate evidence-based occupa-
tional health surveillance strategy for
workers handling nanomaterials. It
may be that hazard surveillance
rather than medical surveillance is
more appropriate to initiate at this
time, given the lack of knowledge
regarding specific health outcomes
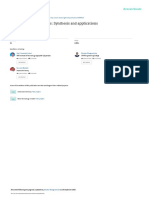
in humans. A continuum of hazard
and medical surveillance is shown in
Figure 2, ranging from no targeted
hazard or medical surveillance action
to targeted medical surveillance of
workers handling nanoparticles. Tar-
geted medical surveillance refers to
medical surveillance for specific bi-
ologic changes known to be linked to
exposure to specific engineered
nanoparticles. Generally, each action
in the continuum builds on the con-
duct of the previous actions.
No Targeted Action (ie, No
Specific Occupational
Surveillance Related to
Engineered Nanoparticles)
A decision to take no targeted
action for workers potentially ex-
posed to engineered nanoparticles
may be influenced by whether or not
there are other medical surveillance
programs in place for these workers.
These programs are established
because of existing hazardous sub-
stances in the workplace rather than
exposure to engineered nanopar-
ticles. Clearly, there is extensive
medical surveillance conducted in
various workplaces where known
hazardous materials are manufac-
tured or used. Over 30 Occupational
Safety and Health Administration
(OSHA) standards and hundreds of
National Institute for Occupational
Safety and Health (NIOSH) guid-
ance documents recommend medical
surveillance for various occupational
hazards. Additionally, three types of
medical surveillance that might also
be occurring in a workplace pertain
to the assessment of the workers abil-
ity to wear or use required respiratory
or other personal protective equip-
ment, and medical examination per-
taining to job placement and medical
examination as part of emergency
medical care after a work-related ex-
posure or incident. So the option at
one end of the continuum is no new
action in addition to what already is
occurring, or, if there is no other
medical surveillance occurring in a
given workplace, then nothing new
related to medical surveillance
would be implemented.
Hazard surveillance may follow
a similar logic as medical surveil-
lance.
13
Employers, as part of their
due diligence and in adherence to
occupational safety and health and
environmental laws, are obligated to
make a determination as to whether
regulated materials are present in the
workplace. This is a rudimentary
form of hazard surveillance. The no
action option involves doing noth-
ing beyond this rudimentary effort.
Whether pertaining to hazard or
health surveillance, a no action
determination should be the product
of active evaluation that is well doc-
umented, and should not be equated
with inaction. In most cases no
action will pertain to no additional
medical or hazard surveillance for
workers potentially exposed to engi-
neered nanoparticles.
Document the Presence of
Engineered Nanomaterials
The presence of free (unbound)
engineered nanomaterials in work-
places where they are produced or
purchased should be obvious to em-
ployers since they are the result of
intentional efforts. This second option
for action on the continuum includes
identifying and recording where in the
Fig. 2. Continuum of hazard and medical surveillance and related research approaches for
nanoparticle workers.
520 Health Surveillance and Nanotechnology
Schulte et al
workplace and production process
engineered nanomaterials are han-
dled (eg, producing nanomaterials as
powders or slurries/suspensions/
solutions), including storage and dis-
posal.
71
The information that would
be useful to record can be gathered
based on existing management
knowledge of processes and job
tasks, including
What nanomaterials are being
manufactured or handled
Where in the workplace exposure
might occur
When these nanomaterials are
made or used
How the nanomaterials are pro-
duced or used.
Special consideration should be
given to processing equipment break-
down and the need for and frequency
of major routine maintenance.
Less clear are situations where
engineered nanoparticles are aggre-
gated, agglomerated or even embed-
ded in materials, such as composites,
and hence are not free or unbound.
71
The question is whether these engi-
neered nanoparticles can be released
and inhaled or come into contact
with the skin. The answer may be
yes if the nanomaterial is subject
to manipulation through kinetic pro-
cesses that result in generating air-
borne nanomaterial or free unbound
engineered nanoparticles. There is
limited information currently avail-
able to evaluate the extent to which
such release actually occurs.
Identify Workers Potentially
Exposed to Nanoparticles
Identification of workers poten-
tially exposed to engineered nano-
particles is the next higher action
option on the continuum. Given the
uncertainty about the hazards of en-
gineered nanoparticles it has been
suggested that a broad definition ini-
tially be used to identify workers
potentially exposed to nanoparticles
and that such a definition be refined
as more reliable information on ex-
posures and health effects become
available.
72
The broad definition
will, by intent, be overly broad in
terms of identification of a group that
is at risk of exposure. The Depart-
ment of Energy, for example, defines
a worker potentially exposed to en-
gineered nanoparticles as one who
handles engineered nanoscale par-
ticles that have the potential to
become dispersed in air;
is routinely exposed (spends a sig-
nificant amount of time in an area
in which engineered nanoparticles
have the potential to be dispersed
in the air);
works on equipment that might be
contaminated with materials that
could foreseeably release engi-
neered nanoparticles during ser-
vicing or maintenance.
72
The purpose for identifying and
recording such information in a sys-
tematic mannersuch as in a data-
baseis 3-fold. First, such records
can be used to determine whether or
not workers with potential exposure
will need to have appropriate train-
ing in the safe handling and disposal
of materials, proper maintenance of
equipment, and use of personal pro-
tective equipment. Second, such
records will be available if there is a
need to establish exposure regis-
tries or participate in epidemiologic
or health surveillance studies.
73,74
Third, should new information about
health effects trigger the need for
medical monitoring, a roster of po-
tentially at risk workers will be
available.
As more information becomes
available about appropriate metrics
and instrumentation for airborne-
engineered nanoparticles, there will
be a need for quantitative rather than
qualitative assessments of exposure
to engineered nanoparticles for indi-
vidual workers. Such information
will allow for assessment of the ef-
fectiveness of controls, adherence
with exposure recommendations and
standards, and could serve to help set
action levels to trigger medical sur-
veillance. At present it is not known
what exposure metrics and particle
characteristics (eg, mass, surface
area, particle count, shape, aerody-
namic diameters) are best for expo-
sure assessment; however, particle
count, surface area and mass are pres-
ently considered to be most useful.
19
Whether monitoring biological
markers of exposure will be useful to
supplement exposure assessment is
not known. Before such action would
be warranted more information is
needed on biotransformation of par-
ticles and toxicokinetics, given the
potential invasiveness and some-
times complicated logistical issues
surrounding biomonitoring.
General Medical Monitoring
Baseline and periodic routine non-
specific medical monitoring is the
next action in the occupational health
continuum. It may take place apart
from or in conjunction with one of
the previous actions. If such medical
monitoring is provided it would
likely either be provided to all work-
ers in a facility or to some subset of
workers potentially exposed to engi-
neered nanoparticles. The utility of
nonspecific medical monitoring is
limited because the health endpoints
that may be linked to engineered
nanoparticle are not well known or
confirmed at this time. Nonetheless,
general medical monitoring may
serve as an early warning system for
possible, yet to be determined, health
effects linked to exposure. This de-
termination will require that the data
be continually analyzed on a group
basis and if possible, linked to expo-
sure and compared with appropriate
comparison population rates. The
limitation of this monitoring ap-
proach is that it may identify health
effects unrelated to exposure to en-
gineered nanoparticles and in some
cases false positives, all of which
will require follow-up and diagnostic
evaluation. It also may give moni-
tored employees a false sense that
such procedures would be sensitive
to any health risk associated with
exposure to engineered nanopar-
ticles. This needs to be balanced
against a reasonable assurance that
periodic examination affords that no
JOEM
Volume 50, Number 5, May 2008 521
obvious or easily detected health ef-
fects are present.
In some worksites, workers will be
monitored through existing programs
if they work in areas with both reg-
ulated hazards (or hazards which
may not be regulated but for which
well-accepted medical monitoring
procedures exist) and engineered
nanoparticles. This medical monitor-
ing, which is conducted due to the
exposure to the regulated hazards,
may have the effect of collecting
baseline health information for
workers. If medical data were in-
cluded, medical history and lifestyle
information (eg, tobacco and alcohol
use, exercise frequency) would also
be useful to record in such baseline
data.
Targeted Medical Testing for
Workers Exposed to
Engineered Nanoparticles
If specific hazards of engineered
nanoparticles are identified and
characterized then it could be pos-
sible to detect preclinical changes
through medical testing and/or
modify exposure potential through
risk management interventions.
Medical monitoring is not meant as
the primary means for protecting
workers but rather as a secondary
prevention effort, after risk manage-
ment efforts (eg, engineering con-
trols and use of personal protection
equipment) have been implement-
ed.
75
The rationale for specific med-
ical surveillance of workers exposed
to nanoparticles is based on the re-
sults of a needs assessment.
10
A
primary determination from a needs
assessment involves some assess-
ment of risk, which is the potential or
probability of a hazardous material
under certain exposure scenarios to
cause harm.
Risk may be thought of as a com-
bination of hazard and exposure. For
example, workers producing a sub-
stance with known toxicity but never
exposed might be judged as not at
risk from that substance. A compo-
nent of determining this risk, how-
ever, may involve the presence or
absence of controls (engineering, ad-
ministrative, personal protection) in
the workplace and whether the expo-
sure is being controlled successfully.
Further, even if low, the probability
of a process failure, operator error, or
industrial accident could result in
some potential risk. If the potential
exists for residual riskthat is, the
workers are at some risk of harm
based on the hazard and exposure
concentration, medical surveillance
may be warranted. Targeted medical
testing data, analyzed on a group
basis, can assist in detecting patterns
and trends, and linking with expo-
sure.
14,16
If known or anticipated
biological changes linked to expo-
sure to engineered nanoparticles can
be identified, then medical testing
can serve to identify sentinel
events.
76,77
On the basis of the toxicological
studies summarized above, prelimi-
nary findings of research on exposure
to engineered nanoparticles and previ-
ous research of ultrafine particles, the
most likely target organ systems will
include the respiratory and circula-
tory systems, although skin exposure
may also occur.
1,79,20,22
Medical
tests that include an evaluation of the
respiratory system could help to in-
dicate changes in pulmonary func-
tion or early indicators of fibrosis.
Although not widely validated, ex-
haled nitrous oxide or isoprostanes
have been used as noninvasive mark-
ers of pulmonary inflammation.
7880
Additionally, the National Institute
of Occupational Health in South Af-
rica is conducting a biomarker study
of gold miners.
81
They are monitor-
ing blood levels of several antioxi-
dant enzymes, cytokines, and total
antioxidant levels. They report that
decreases in red cell glutathione
and serum Clara cell proteins were
associated with silica exposure.
Also, tests that are indicative of
inflammatory response in pulmo-
nary and circulatory systems might
be useful.
5,62,65
None of these tests
are validated for use in asymptom-
atic workers. Beyond the respiratory
and circulatory systems, the impact
of engineered nanoparticles is poorly
understood. Nanoparticles in the
blood circulatory system could reach
many other organs and specific tests
related to early changes in organ
function may be warranted.
3
Trans-
location to the brain by nanoparticles
has been demonstrated but the sever-
ity and clinical relevance is not
known, nor is there enough informa-
tion to suggest what type of testing
could be useful to assess such ef-
fects. Criteria for medical testing for
workers have been published and
would need to be met before imple-
mentation of specific medical
screening of workers potentially ex-
posed to engineered nanopar-
ticles.
10,12,16,8284
At the present
time, it does not appear that there is
scientific and medical evidence suf-
ficient to recommend medical
screening of workers potentially ex-
posed to engineered nanoparticles.
A medical surveillance plan, like all
targeted medical surveillance plans, in-
volves adherence to recognized basic
elements that include interpretation of
the findings for workers and manage-
ment, implementation of follow-up ac-
tion, and program evaluation.
10,82
Data Collection and
Use Responsibilities
Regardless of the level of occupa-
tional health surveillance action that
may be selected; the gathering, stor-
age, and use of individual-level or
individually identifiable data carry
certain legal and ethical obligations.
Various issues are outside the scope
of this article; they include confiden-
tiality, Health Insurance Portability
and Accountability Act (HIPPA) re-
quirements, understanding of origi-
nal and intended use of surveillance
data, informed consent, and report-
ing test results, among others.
12,83,84
Less well appreciated are possible
ethical responsibilities to properly
analyze and report findings from
constructed databases.
522 Health Surveillance and Nanotechnology
Schulte et al
Future Research
In Vitro and In Vivo Studies
The basis for selecting among the
options for occupational health sur-
veillance of workers potentially ex-
posed to engineered nanoparticles is
based on knowledge of potential haz-
ards. The results from in vivo studies
involving various animal models and
in vitro studies using various testing
systems are frequently used as the
basis for that determination.
1,9
Al-
though such research has provided
preliminary indications of hazards
from some types of engineered nano-
particles, the heterogeneity of these
particles is large. Also, whether and
the degree to which effects seen in
various animal testing systems are
relevant to humans (especially at lev-
els to which humans are exposed)
may not be known. Nevertheless,
animal (rat) models of adverse lung
effects from inhaled poorly soluble
particles have been shown to be
consistent with the findings of
some epidemiological studies in-
cluding occupational exposures to
asbestos, silica, and diesel exhaust
particulate.
8588
Ultimately for guidance and regu-
latory purposes, it may be necessary
to determine whether broad catego-
ries of engineered nanoparticles can
be identified that are based on simi-
lar physicochemical parameters. It is
not known if particle size will be the
preeminent factor or various combi-
nations of size and other physico-
chemical parameters will be needed
to help establish risk-based catego-
ries on which to base future surveil-
lance decisions.
Epidemiologic Studies
It will be important to know
whether workers who have handled
and been exposed to engineered
nanoparticles develop adverse health
effects and disease.
19
Chronic ef-
fects, such as pulmonary malignant
and nonmalignant disease, and circu-
latory disease may take decades to
assess in worker populations. This is
because of the inherent nature of
such effects as well as the fact that
exposures, if they occur, are likely to
be quite low given the initial societal
concerns and precautions for control-
ling workplace exposures to engi-
neered nanoparticles.
1,79,75
Acute
effects may be more easily assessed
but the frequency also may be low
given the wide prevalence of work-
place exposure controls.
Epidemiologic studies of worker
populations potentially exposed to
engineered nanoparticles will be dif-
ficult to conduct for various reasons
including
1. Heterogeneity of engineered nano-
particles making it difficult to find a
sufficiently large group of workers
exposed to the same particles;
2. Dispersion of the contemporary
workforce resulting from generally
small numbers of potentially ex-
posed workers in any given facility;
3. Difficulty in identifying and re-
cruiting potentially exposed work-
ers and employers;
4. Insufficient exposure to cause
and/or latency to detect chronic
diseases;
5. Lack of exposure data and stan-
dardized exposure metrics needed
to develop comparable exposure
histories.
Despite these limitations, the es-
tablishment of formal national, or
even an international, exposure and
employee tracking registries might
be warranted. Exposure registries are
useful tools for surveillance of new
or perceived hazards since they pro-
vide documentation of who is work-
ing with which materials, when, and
where in the facility. A registry pro-
vides a structured and orderly ap-
proach to handling the problem of
identifying and maintaining commu-
nication with workers exposed to
hazardous substances,
72
utilizing a
common set of variables.
74
An expo-
sure registry is the enrollment of
persons exposed or likely to have
been exposed to occupational or en-
vironmental hazards that can serve as
a means of identifying persons for
primary or secondary preventive ef-
forts. In occupational situations,
company employee rosters are de
facto registries; however, they may
not address employees who leave a
company. Moreover, for new tech-
nologies, such as nanotechnology,
registries could be developed and
maintained by government entities;
however, there are examples of pri-
vate sector registries related to expo-
sure to commercial products.
Whether such registries would foster
potential discriminatory actions or
legal liabilities would need to be
addressed.
If sufficient numbers of workers
can be recruited with exposure to the
same engineered nanoparticles using
similar exposure metrics, it may be
useful to conduct studies to link ex-
posures with various biological
markers or other biological changes
that may be precursors to more seri-
ous health effects.
89
In the meantime,
results from in vivo and in vitro
research will continue to be the basis
for decision-making concerning oc-
cupational health surveillance.
Companies currently involved
with nanotechnology are faced with
the dilemma of balancing a desire to
expand a potentially bountiful tech-
nology with limited knowledge
about the potential hazards.
90
The
potential health risks associated with
nanotechnology are not well estab-
lished, and the perceived risks are
undetermined. As one commentator
noted: Even if studies showed every
commercially relevant nanoparticle
to be harmless in every real world
scenario, public skepticism about the
safety of nanoparticles could still
build and sharply limit their use in
products.
91
One of the earliest areas
where exposures to nanoparticles is
occurring is in the workplace
including universities and other non-
manufacturing settings.
71
In the face
of uncertainty about the hazards of
nanoparticles, a corporate or societal
response (such as implementing se-
lected exposure registries in poten-
tially high exposure sectors) may
assure the public that appropriate
efforts are being taken to identify
JOEM
Volume 50, Number 5, May 2008 523
and control exposures in a timely and
responsible manner.
References
1. Department for the Environment, Food,
and Rural Affairs (DEFRA). Character-
izing the Potential Risks Posed by En-
gineered Nanoparticles: A First UK
Government Research Report. London:
HM Government; 2006:157. Available
at: http://ec.europa.eu/european_group_
ethics/docs/uk_report_nanoparticles_
en.pdf. Accessed October 23, 2007.
2. Maynard AD, Kuempel ED. Airborne
nanostructural particles and occupational
health. J Nanoparticles Res. 2005;7:587
614.
3. Oberdorster G, Oberdorster E, Oberdor-
ster J. Nanotoxicologyan emerging
discipline involving studies of ultrafine
particles. Environ Health Perspect. 2005;
113:823839.
4. Borm PJA, Robbins D, Haubold S, et al.
The potential risks of nanoparticles: a
review carried out for ECETOC. Particle
Fibre Toxicol. 2006;3:11.
5. Li Z, Hulderman T, Salmen R, et al.
Cardiovascular effects of pulmonary ex-
posure to single-wall carbon nanotubes.
Environ Health Perspect. 2007;115:377
382.
6. Duffin R, Tran L, Brown D, et al. Proin-
flammogenic effects of low-toxicity and
metal nanoparticles in vivo and in vitro:
highlighting the role of particle surface
area and surface reactivity. Inhal Toxicol.
2007;19:849856.
7. Australian Safety and Compensation Coun-
cil (ASCC). A review of the potential
occupational safety and health implications
of nanotechnology. 2000. Available at:
http://www.ascc.gov.au/ascc/healthsafety/
emergingissues/nanotechnology.
8. Aitken RJ, Creely KS, Tran CL. Nanopar-
ticles: an Occupational Hygiene Review.
Health and Safety Executive. Research Re-
port 274. Norwich UK; HSE Books; 2004.
9. Institut de Recherche Robert-Sauve en
Sante du Travail (IRSST). Nanopar-
ticles: Actual Knowledge about Occupa-
tional Health and Safety Risks and
Prevention Measures. Studies and Re-
search Projects R-470. Montreal, Canada:
IRSST;2006:100 pages. Available at:
http://www.irsst.qc.ca/files/documents/
PubIRSST/R-470.pdf. Accessed October
23, 2007.
10. Harber P, Conlon C, McCunney RJ. Oc-
cupational medical surveillance. In: Mc-
Cunney RJ, ed. A Practical Approach to
Occupational and Environmental Medi-
cine. Philadelphia, PA: Lippincott, Wil-
liams, and Wilkins; 2003;582599.
11. Baker EL, Matte TP. Occupational
health surveillance. In: Rosenstock L,
Cullen E, Brodkin R, et al, eds. Textbook
of Clinical Occupational and Environ-
mental Medicine. 2nd ed. Philadelphia,
PA: Elsevier Saunders Company; 2005:
7682.
12. Ashford NA, Spadafor CJ, Hattis DB, et
al. Monitoring the Worker for Exposure
and Disease. Baltimore, MD: The Johns
Hopkins University Press; 1990.
13. Wegman DH. Hazard surveillance. In
Halperin W, Baker EL, Monson RR, eds.
Public Health Surveillance. New York:
VanNostrand Reinhold; 1992:4275.
14. Halperin WE. The role of surveillance in
the hierarchy of prevention. Am J Ind
Med. 1996;29:321323.
15. Wagner GR, Fine LJ. Surveillance and
health screening in occupational health.
In Wallace RB, ed. Maxcy-Rosenau-Last
Public Health and Preventive Medicine.
15th ed. New York: McGraw-Hill Med-
ical Publishing; 2008:759793.
16. Halperin WE, Ratcliffe JM, Frazier JM,
et al. Medical screening in the workplace:
proposed principles. J Occup Med. 1986;
28:522547.
17. Naumann BD, Sargent EV. Setting occu-
pational exposure limits for pharmaceu-
ticals. Occup Med. 1997;12:6780.
18. Health and Safety Executive. Foundry
Control Guidance Sheet FD02: Engineer-
ing Control for Foundry Fumes from
Melting. London: HSE; 2006.
19. Kuempel ED, Geraci CL, Schulte PA.
Risk assessment and research needs for
nanomaterials: an examination of data
and information from current studies. In:
Simeonova PP, et al, eds. Nanotechnol-
ogyToxicological Issues and Environ-
mental Safety. Proceedings of the NATO
Advanced Research Workshop on Nano-
technologyToxicological Issues and
Environmental Safety, Varna, Bulgaria,
1217 August, 2006. New York: Spring-
er; 2007:119145.
20. Donaldson K, Tran L, Jimenez LA, et al.
Combustion-derived nanoparticles: a re-
view of their toxicology following inha-
lation exposure. Part Fibre Toxicol.
2005;2:1014.
21. Oberdorster G, Maynard A, Donaldson
K, et al. Principles for characterizing the
potential human health effects from ex-
posure to nanomaterials: elements of a
screening strategy. Part Fibre Toxicol.
2005;35:28.
22. Gwinn MR, Vallyathan V. Nanoparticles:
health effects-pro and cons. Env Health
Perspect. 2006;114:18181825.
23. International Commission on Radiologi-
cal Protection. Human Respiratory Tract
Model for Radiological Protection. An-
nals of the ICRP Publ. 66. New York:
Pergamon; 1994.
24. Jaques PA, Kim CS. Measurement of
total lung deposition of inhaled ultrafine
particles in healthy men and women.
Inhal Toxicol. 2000;12:715731.
25. Brown JS, Zeman KL, Bennett WD. Ul-
trafine particle deposition and clearance
in the healthy and obstructed lung. Am J
Respir Crit Care Med. 2002;166:1240
1247.
26. Tinkle SS, Antonini JM, Rich BA, et al.
Skin as a route of exposure and sensiti-
zation in chronic beryllium disease.
Environ Health Perspect. 2003;111:
12021208.
27. Kohli AK, Alpar HO. Potential use of
nanoparticles for transcutaneous vaccine
delivery: effect of particle size and
charge. Int J Pharm. 2004;275:1317.
28. Ryman-Rasmussen JP, Riviere JE, Mon-
teiro-Riviere NA. Penetration of intact
skin by quantum dots with diverse phys-
icochemical properties. Toxicol Sci.
2006;91:159165.
29. Jani P, Halbert GW, Langridge J, et al.
Nanoparticle uptake by the rat gastroin-
testinal mucosa: quantitation and particle
size dependency. J Pharm Pharmacol.
1990;42:821826.
30. Desai MP, Labhasetwar V, Amidon GL,
et al. Gastrointestinal uptake of biode-
gradable microparticles: effect of particle
size. Pharm Res. 1996;13:18381845.
31. Florence AT. The oral absorption of mi-
cro- and nanoparticulates: neither excep-
tional nor unusual. Pharm Res. 1997;14:
259266.
32. Sydbom A, Blomberg A, Parnia S, Sten-
fors N, Sandstrom T, Dahlen SE. Health
effects of diesel exhaust emissions. Eur
Resp J. 2001;17:733746.
33. Tornqvist H, Mills NL, Gonzalez M, et
al. Persistent endothelial dysfunction in
humans after diesel exhaust inhalation.
Am J Respir Crit Care Med. 2007;176:
395400.
34. Stayner L, Dankovic D, Smith R, et al.
Predicted lung cancer risk among miners
exposed to diesel exhaust particles. Am J
Ind Med. 1998;34:207219.
35. Ambroise D, Wild P, Moulin JJ. Update
of a meta-analysis on lung cancer and
welding. Scand J Work Environ Health.
2006;32:2231.
36. Antonini JM, Santamaria AB, Jenkins
NT, et al. Fate of manganese associated
with the inhalation of welding fumes:
potential neurological effects. Neuro
Toxicol. 2006;27:304310.
37. Chalupa DC, Morrow PE, Oberdorster G,
et al. Ultrafine particle deposition in sub-
jects with asthma. Environ Health Per-
spect. 2004;112:879882.
524 Health Surveillance and Nanotechnology
Schulte et al
38. Pietropaoli AP, Frampton MW, Oberdor-
ster G, et al. Blood markers of coagula-
tion and inflammation in healthy subjects
exposed to carbon ultrafine particles. In:
Heinrich U, ed. Effects of Air Contami-
nant on the Respiratory TractInterpre-
tations from Molecular to Meta Analysis.
Stuttgart, Germany: INIS Monographs,
Frauinhofer IRB Verlag; 2004;181194.
39. Wichmann H-E, Spix C, Tuch T, et al.
Daily mortality and fine and ultrafine
particles in Erfurt, Germany. Part I: role
of particle number and particle mass. Res
Rep Health Eff Inst. 2000;98:586.
40. Dockery DW, Pope CA, Xu X, et al. An
association between air pollution and
mortality in six U.S. cities. N Engl J Med.
1993;329:17531759.
41. Ibald-Mulli A, Wichmann HE, Kreyling
W, et al. Epidemiological evidence on
health effects of ultrafine particles.
J Aerosol Med Depos. 2002;15:189201.
42. Pope CA, Burnett RT, Thun MJ, et al.
Lung cancer, cardiopulmonary mortality
and long term exposure to fine particulate
air pollution. JAMA. 2002;287:1132
1141.
43. Pope CA, Burnett RT, Thurston GD, et
al. Cardiovascular mortality and long-
term exposure to particulate air pollution:
epidemiological evidence of general
pathophysiological pathways of disease.
Circulation. 2004;109:7174.
44. Oberdorster G, Yu CP. The carcinogenic
potential of inhaled diesel exhaust: a
particle effect? J Aerosol Sci. 1991;21:
S397S401.
45. Driscoll KE. Role of inflammation in the
development of rat lung tumors in re-
sponse to chronic particle exposure. In:
Mauderly JL, McCunney RJ, eds. Parti-
cle Overload in the Rat Lung and Lung
Cancer: Implications for Human Risk
Assessment. Philadelphia, PA: Taylor &
Francis; 1996;139152.
46. Tran CL, Buchanan D, Cullen RT, et al.
Inhalation of poorly soluble particles. II.
Influence of particle surface area on in-
flammation and clearance. Inhal Toxicol.
2000;12:11131126.
47. Hohr D, Steinfartz Y, Schins RP, et al.
The surface area rather than the surface
coating determines the acute inflamma-
tory response after instillation of fine and
ultrafine TiO2 in the rat. Int J Hyg
Environ Health. 2002;205:239244.
48. Warheit DB, Webb TR, Reed KL, et al.
Pulmonary toxicity study in rats with
three forms of ultrafine-TiO2 particles:
differential response related to surface
properties. Toxicology. 2007;230:90
104.
49. Elder A, Gelein R, Finkelstein JN, et al.
Effects of Subchronically Inhaled Carbon
Black in Three Species. I. Retention Ki-
netics, Lung Inflammation, and Histopa-
thology. Toxicol Sci. 2005;88:614629.
50. Castranova V. From coal mine dust to
quartz: mechanisms of pulmonary patho-
genicity. Inhal Toxicol. 2000;(Suppl 3):
714.
51. Elder A, Gelein R, Silva V, et al. Trans-
location of inhaled ultrafine manganese
oxide particles to the central nervous
system. Environ Health Perspect. 2006;
114:11721178.
52. Geiser M, Rothen-Rutishauser B, Kapp
N, et al. Ultrafine particles cross cellular
membranes by nonphagocytic mecha-
nisms in lungs and in cultured cells.
Environ Health Perspect. 2005;113:
15551560.
53. Muhlfeld C, Geiser M, Kapp N, et al.
Re-evaluation of pulmonary titanium di-
oxide nanoparticle distribution using the
relative deposition index: evidence for
clearance through microvasculature. Part
Fibre Toxicol 2007;29:47. Available at:
http://www.particleandfibretoxicology.
com/content/pdf/1743-8977-4-7.pdf. Ac-
cessed October 23, 2007.
54. Oberdorster G, Sharp Z, Atudorei V, et al.
Extrapulmonary translocation of ultrafine
carbon particles following whole-body in-
halation exposure of rats. J Toxicol Environ
Health Part A. 2002;65:15311543.
55. Kreyling WG, Semmler M, Erbe F, et al.
Translocation of ultrafine insoluble irid-
ium particles from lung epithelium to
extrapulmonary organs is size dependent
but very low. J Toxicol Environ Health
Part A. 2002;65:15131530.
56. Kreyling WG, Semmler-Behnke M,
Moller W. Ultrafine particle-lung interac-
tions: does size matter? J Aerosol Med.
2006;19:7483.
57. Hagens WI, Oomen AG, de Jong WH, et
al. What do we (need to) know about the
kinetic properties of nanoparticles in the
body? Regul Toxicol Pharmacol. 2007;
49:217229.
58. Mills NL, Amin N, Robinson SD, et al.
Do inhaled carbon nanoparticles translo-
cate directly into the circulation in hu-
mans? Am J Respir Crit Care Med. 2006;
173:425431.
59. Wiebert P, Sanchez-Crespo A, Seitze J,
et al. Negligible clearance of ultrafine
particles retained in healthy and affected
human lungs. Eur Respir J. 2006;28:
286290.
60. Nemmar A, Hoet PHM, Vanquicken-
borne B, et al. Passage of inhaled parti-
cles into the blood circulation in humans.
Circulation. 2002;105:411414.
61. Lam CW, James JT, McCluskey R, et al.
Pulmonary toxicity of single-wall carbon
nanotubes in mice 7 and 90 days after
intratracheal instillation. Toxicol Sci.
2004;77:126134.
62. Shvedova AA, Kisin ER, Mercer R, et al.
Unusual inflammatory and fibrogenic
pulmonary responses to single-walled
carbon nanotubes in mice. Am J Physiol
Lung Cell Mol Physiol. 2005;289:L698
L708.
63. Donaldson K, Aitken R, Tran L, et al.
Carbon nanotubes: a review of their prop-
erties in relation to pulmonary toxicology
and workplace safety. Toxicol Sci. 2006;
92:522.
64. Helland A, Wick P, Koehler A, et al.
Reviewing the environmental and human
health knowledge base of carbon nano-
tubes. Environ Health Perspect. 2007;
115:11251131.
65. Radomski A, Juraz P, Alonso-Escolano
D, et al. Nanoparticle-induced platelet
aggregation and vascular thrombosis.
Br J Pharmacol. 2005;146:882893.
66. Lam C-W, James JT, McCluskey R, et al.
A review of carbon nanotube toxicity and
assessment of potential occupational and
environmental health risks. Crit Rev
Toxicol. 2006;36:189217.
67. Mossman BT, Borm PJ, Castranova V, et
al. Mechanisms of action of inhaled fi-
bers, particles and nanoparticles in lung
and cardiovascular diseases. Part Fibre
Toxicol. 2007; 4:4. Available at: http://
www.particleandfibretoxicology.com/
content/pdf/1743-8977-4-4.pdf. Ac-
cessed October 23, 2007.
68. Zhang Q, Kusaka Y, Donaldson K. Com-
parative pulmonary responses caused by
exposure to standard cobalt and ultrafine
cobalt. J Occup Health. 2000;42:179
184.
69. Zhang Q, Kusaka Y, Zhu X, et al.
Comparative toxicity of standard nickel
and ultrafine nickel in lung after intra-
tracheal instillation. J Occup Health.
2003;45:2330.
70. Chen Z, Meng H, Xing G, et al. Acute
toxicological effects of copper nanopar-
ticles in vivo. Toxicol Lett. 2006;163:
109120.
71. Schulte PA, Geraci C, Zumwalde R,
Hoover M, Kuempel E. Occupational risk
management of engineered nanoparticles. J
Occup Environ Health. 2008;5:239249.
72. Department of Energy (DOE). Approach
to Nanomaterial E S & H. Nanoscale
Science Research Centers. Revision 2
June, 2007. Available at: http://www.sc.
doe.gov/bes/DOE_NSRC_Approach_
to_Nanomaterial_ESH.pdf.
73. Nasterlack M, Zober A, Oberlinner C.
Considerations on occupational medical
surveillance in employees handling nano-
particles. Int Arch Occup Environ Health.
2008;81:721726.
JOEM
Volume 50, Number 5, May 2008 525
74. Schulte PA, Kaye WE. Exposure regis-
tries. Arch Environ Health. 1988;43:155
161.
75. National Institute for Occupational
Safety and Health (NIOSH). Approaches
to safe nanotechnology: an information
exchange with NIOSH. Available at:
www.cdc.gov/niosh/topics/nanotech/
nano_exchange.html.
76. Mullan RJ, Murthy LI. Occupational senti-
nel health events: an updated list for phy-
sician recognition and public health surveil-
lance. Am J Ind Med. 1999;19:775799.
77. Rutstein D, Mullan R, Frazier T, et al.
Sentinel health events (occupational): a
basis for physician recognition and public
health surveillance. Am J Public Health.
1983;73:10541062.
78. Birrell MA, McCluskie K, Hardaker E,
Knowles R, Belvisi MG. Utility of
exhaled nitric oxide as a noninvasive
biomarker of lung inflammation in a
disease model. Eur Respir J. 2006;28:
12361244.
79. Beilman G. Exhaled nitric oxide in
pathophysiologic states: the substance
behind the gas. Chest. 2004;125:1113.
80. Makris D, Paraskakis E, Korakas P, et al.
Exhaled breath condensate 8-isoprostane,
clinical parameters, radiological indices
and airway inflammation in COPD. Res-
piration. 2008;75:138144.
81. Murray J, Girdler-Brown B. Validation
of biomarkers for improved assessment
of exposure and early effect from expo-
sure to crystalline silica. Final report
SIOM. 03-08-03 Phase 2. Johannesburg,
South Africa: South African Institute of
Occupational Medicine; 2007.
82. National Institute for Occupational
Safety and Health (NIOSH). Criteria for
a Recommended Standard: Occupational
Exposure to Refractory Ceramic Fibers.
Cincinnati, Ohio: DHHS, Centers for
Disease Control and Prevention (NIOSH)
Publication No. 2006-125; 2006.
83. International Labour Office. Technical and
Ethical Guidelines for Workers Health
Surveillance (Occupational Safety &
Health Series No. 72). Geneva: Interna-
tional Labour Office;1998:54. Available at:
http://www.ilo.org/public/english/protection/
safework/cops/english/download/e000017.
pdf. Accessed October 23, 2007.
84. American College of Occupational Envi-
ronmental Medicine. Position statement:
medical surveillance in the workplace.
2000. Available at: http://www.acoem.
org/paprquid/papers/msurv.htm.
85. Mauderly JL. Relevance of particle-
induced rat lung tumors for assessing
lung carcinogenic hazard and human lung
cancer risk. Environ Health Perspect.
1997;105(Suppl 5):13371346.
86. Stayner L, Dankovic D, Smith R, et al.
Predicted lung cancer risk among miners
exposed to diesel exhaust particles. Am J
Ind Med. 1998;34:207219.
87. Kuempel ED, Smith RJ, Dankovic DA, et
al. Concordance of rat and human based
risk estimates for particle-related lung
cancer. Ann Occup Hyg. 2002;46(Suppl
1):6266.
88. Kuempel ED, Tran CL, Bailer AJ, et al.
Biological and statistical approaches to
predicting human lung cancer risk from
silica. J Environ Pathol Toxicol Oncol.
2001;20(Suppl 1):1532.
89. Schulte PA. The use of biomarkers in
surveillance, medical screening and inter-
vention. Mutat Res. 2005;592:155163.
90. Schulte PA, Salamanca-Buentello F. Eth-
ical and scientific issues of nanotechnol-
ogy in the workplace. Environ Health
Perspect. 2007;115:512.
91. Holman MW. Nanotech environmental,
health, and safety risks: action needed.
In: Health and Nanotechnology: Eco-
nomic, Societal, and Institutional Impact.
Perspectives on the Future of Science and
Technology. Report from conference
convened with the cooperation of the
United States Department of State and
the European Commission. Varenna, It-
aly, May 2123. 2006:7887.
526 Health Surveillance and Nanotechnology
Schulte et al
Vous aimerez peut-être aussi
- Grit: The Power of Passion and PerseveranceD'EverandGrit: The Power of Passion and PerseveranceÉvaluation : 4 sur 5 étoiles4/5 (588)
- The Yellow House: A Memoir (2019 National Book Award Winner)D'EverandThe Yellow House: A Memoir (2019 National Book Award Winner)Évaluation : 4 sur 5 étoiles4/5 (98)
- Tool Used: Q2 of MST CLO 1: Calculate Deformation of Statically Determinate Structures Using Geometric and Energy MethodsDocument59 pagesTool Used: Q2 of MST CLO 1: Calculate Deformation of Statically Determinate Structures Using Geometric and Energy MethodsHeaven SinghPas encore d'évaluation
- Format For LabsDocument2 pagesFormat For LabsHeaven SinghPas encore d'évaluation
- List of ELC GroupsDocument4 pagesList of ELC GroupsHeaven SinghPas encore d'évaluation
- Unit 2 Influence Lines Statically Determinate Trusses: StructureDocument24 pagesUnit 2 Influence Lines Statically Determinate Trusses: StructureHeaven SinghPas encore d'évaluation
- Format For LabsDocument2 pagesFormat For LabsHeaven SinghPas encore d'évaluation
- Department: CIVIL ENGINEERING (CED) ODD SemesterDocument2 pagesDepartment: CIVIL ENGINEERING (CED) ODD SemesterHeaven SinghPas encore d'évaluation
- 2.6.1 Details of PO's 1-12 - BE CivilDocument2 pages2.6.1 Details of PO's 1-12 - BE CivilHeaven SinghPas encore d'évaluation
- 10.1007@978 3 319 91137 3 - 14Document6 pages10.1007@978 3 319 91137 3 - 14Heaven SinghPas encore d'évaluation
- Weinstock 2016Document30 pagesWeinstock 2016Heaven SinghPas encore d'évaluation
- Letter of RecommendationDocument2 pagesLetter of RecommendationHeaven SinghPas encore d'évaluation
- Stress-Strain Modelfor Grade275 Reinforcingsteel With Cyclic LoadingDocument9 pagesStress-Strain Modelfor Grade275 Reinforcingsteel With Cyclic LoadingHeaven SinghPas encore d'évaluation
- NearestDocument1 pageNearestHeaven SinghPas encore d'évaluation
- PROOF OF INTERACTION WITH OUTSIDE WORLD - AbetDocument10 pagesPROOF OF INTERACTION WITH OUTSIDE WORLD - AbetHeaven SinghPas encore d'évaluation
- Tut Test 1 MM: 10, TIME: 50 MinsDocument1 pageTut Test 1 MM: 10, TIME: 50 MinsHeaven SinghPas encore d'évaluation
- Design and Cost Analysis of Multistoreyed Building Using Composite SectionDocument2 pagesDesign and Cost Analysis of Multistoreyed Building Using Composite SectionHeaven SinghPas encore d'évaluation
- The Subtle Art of Not Giving a F*ck: A Counterintuitive Approach to Living a Good LifeD'EverandThe Subtle Art of Not Giving a F*ck: A Counterintuitive Approach to Living a Good LifeÉvaluation : 4 sur 5 étoiles4/5 (5795)
- Never Split the Difference: Negotiating As If Your Life Depended On ItD'EverandNever Split the Difference: Negotiating As If Your Life Depended On ItÉvaluation : 4.5 sur 5 étoiles4.5/5 (838)
- Hidden Figures: The American Dream and the Untold Story of the Black Women Mathematicians Who Helped Win the Space RaceD'EverandHidden Figures: The American Dream and the Untold Story of the Black Women Mathematicians Who Helped Win the Space RaceÉvaluation : 4 sur 5 étoiles4/5 (895)
- The Hard Thing About Hard Things: Building a Business When There Are No Easy AnswersD'EverandThe Hard Thing About Hard Things: Building a Business When There Are No Easy AnswersÉvaluation : 4.5 sur 5 étoiles4.5/5 (345)
- Shoe Dog: A Memoir by the Creator of NikeD'EverandShoe Dog: A Memoir by the Creator of NikeÉvaluation : 4.5 sur 5 étoiles4.5/5 (537)
- The Little Book of Hygge: Danish Secrets to Happy LivingD'EverandThe Little Book of Hygge: Danish Secrets to Happy LivingÉvaluation : 3.5 sur 5 étoiles3.5/5 (400)
- Elon Musk: Tesla, SpaceX, and the Quest for a Fantastic FutureD'EverandElon Musk: Tesla, SpaceX, and the Quest for a Fantastic FutureÉvaluation : 4.5 sur 5 étoiles4.5/5 (474)
- A Heartbreaking Work Of Staggering Genius: A Memoir Based on a True StoryD'EverandA Heartbreaking Work Of Staggering Genius: A Memoir Based on a True StoryÉvaluation : 3.5 sur 5 étoiles3.5/5 (231)
- On Fire: The (Burning) Case for a Green New DealD'EverandOn Fire: The (Burning) Case for a Green New DealÉvaluation : 4 sur 5 étoiles4/5 (74)
- The Emperor of All Maladies: A Biography of CancerD'EverandThe Emperor of All Maladies: A Biography of CancerÉvaluation : 4.5 sur 5 étoiles4.5/5 (271)
- Devil in the Grove: Thurgood Marshall, the Groveland Boys, and the Dawn of a New AmericaD'EverandDevil in the Grove: Thurgood Marshall, the Groveland Boys, and the Dawn of a New AmericaÉvaluation : 4.5 sur 5 étoiles4.5/5 (266)
- The Unwinding: An Inner History of the New AmericaD'EverandThe Unwinding: An Inner History of the New AmericaÉvaluation : 4 sur 5 étoiles4/5 (45)
- Team of Rivals: The Political Genius of Abraham LincolnD'EverandTeam of Rivals: The Political Genius of Abraham LincolnÉvaluation : 4.5 sur 5 étoiles4.5/5 (234)
- The World Is Flat 3.0: A Brief History of the Twenty-first CenturyD'EverandThe World Is Flat 3.0: A Brief History of the Twenty-first CenturyÉvaluation : 3.5 sur 5 étoiles3.5/5 (2259)
- The Gifts of Imperfection: Let Go of Who You Think You're Supposed to Be and Embrace Who You AreD'EverandThe Gifts of Imperfection: Let Go of Who You Think You're Supposed to Be and Embrace Who You AreÉvaluation : 4 sur 5 étoiles4/5 (1091)
- The Sympathizer: A Novel (Pulitzer Prize for Fiction)D'EverandThe Sympathizer: A Novel (Pulitzer Prize for Fiction)Évaluation : 4.5 sur 5 étoiles4.5/5 (121)
- Her Body and Other Parties: StoriesD'EverandHer Body and Other Parties: StoriesÉvaluation : 4 sur 5 étoiles4/5 (821)
- Applied Catalysis B: EnvironmentalDocument6 pagesApplied Catalysis B: EnvironmentalAmir ChaNdiaPas encore d'évaluation
- Review Article: An Overview of Nanomaterials For Water and Wastewater TreatmentDocument11 pagesReview Article: An Overview of Nanomaterials For Water and Wastewater TreatmentxKrA1tosPas encore d'évaluation
- Programme BPS 2019Document25 pagesProgramme BPS 2019Mohammad Jellur RahmanPas encore d'évaluation
- Biotecnologia DoradaDocument8 pagesBiotecnologia DoradaRomina Jimenez RevillaPas encore d'évaluation
- 50 Years in The Development of Insulating LiquidsDocument13 pages50 Years in The Development of Insulating LiquidsDante FilhoPas encore d'évaluation
- STS Activity Sheet Week 10Document5 pagesSTS Activity Sheet Week 10Kirstine SanchezPas encore d'évaluation
- Book ChaptersDocument5 pagesBook Chaptersapi-359216567Pas encore d'évaluation
- Presentation NanofluidDocument23 pagesPresentation NanofluidnitaichePas encore d'évaluation
- 05 NanoTechnology Session 1 Properties, Approaches, Methods To Produce Nanomaterials PDFDocument7 pages05 NanoTechnology Session 1 Properties, Approaches, Methods To Produce Nanomaterials PDFhimynameismarshallPas encore d'évaluation
- Review On Nanomaterials: Synthesis and Applications: Materials Today: Proceedings August 2019Document10 pagesReview On Nanomaterials: Synthesis and Applications: Materials Today: Proceedings August 2019DANIEL CORTESPas encore d'évaluation
- AQA Chemistry Bonding Structure and The Properties of Matter KnowIT GCSE1Document76 pagesAQA Chemistry Bonding Structure and The Properties of Matter KnowIT GCSE1Stone ZhongPas encore d'évaluation
- Smart Tools For Smart ApplicationsDocument208 pagesSmart Tools For Smart ApplicationsTaha AbdouPas encore d'évaluation
- 18225-Article Text-81558-1-10-20170301 PDFDocument274 pages18225-Article Text-81558-1-10-20170301 PDFMuhammad Tariq RazaPas encore d'évaluation
- Bacterial SynthesisDocument16 pagesBacterial SynthesisDibyajyoti20 Das4001Pas encore d'évaluation
- AP 2 Module 6Document13 pagesAP 2 Module 6Anshuman NandanPas encore d'évaluation
- IB - Biocompatibility and Therapeutic Evaluation of Magnetic Liposomes Designed For Self-Controlled Cancer Hyperthermia and ChemotherapyDocument11 pagesIB - Biocompatibility and Therapeutic Evaluation of Magnetic Liposomes Designed For Self-Controlled Cancer Hyperthermia and ChemotherapyManash GogoiPas encore d'évaluation
- Chapter 1Document39 pagesChapter 1Shaban BukhariPas encore d'évaluation
- Technik Forum 1 2008 Fachzeitschrift enDocument36 pagesTechnik Forum 1 2008 Fachzeitschrift enAnonymous 83NuRqplPas encore d'évaluation
- UG Project Report: Nano - Material Based BiosensorsDocument23 pagesUG Project Report: Nano - Material Based BiosensorsAJay MahichPas encore d'évaluation
- Design For ManufacturingDocument21 pagesDesign For ManufacturingNat Melchor100% (1)
- Green Chemistry by Nanocatalysis PDFDocument12 pagesGreen Chemistry by Nanocatalysis PDFAnonymous rm2rf6Pas encore d'évaluation
- 8th Invention To Innovation SummitDocument49 pages8th Invention To Innovation SummitSyed ZamanPas encore d'évaluation
- Optimization of Au0-Cu0Document12 pagesOptimization of Au0-Cu0juanPas encore d'évaluation
- 2019 04 July Aug IssueDocument15 pages2019 04 July Aug IssueRafael PiresPas encore d'évaluation
- Review ArticleDocument17 pagesReview ArticleArun AchalamPas encore d'évaluation
- BYK AdditivesDocument28 pagesBYK Additivesmanoj973100% (2)
- Synthesis of Zno Nanoparticles ThesisDocument5 pagesSynthesis of Zno Nanoparticles Thesisgladyscepparulonewark100% (1)
- Hassan 2015Document9 pagesHassan 2015Mia karolinaPas encore d'évaluation
- Application of Nanoparticles in AgricultureDocument11 pagesApplication of Nanoparticles in AgricultureShetti Swamy PatelPas encore d'évaluation
- High-Temperature Behavior of Laser ElectrodispersiDocument18 pagesHigh-Temperature Behavior of Laser Electrodispersiateer6727Pas encore d'évaluation