Académique Documents
Professionnel Documents
Culture Documents
The Biomedical Engineering Handbook: Second Edition
Transféré par
Karthik Rajv0 évaluation0% ont trouvé ce document utile (0 vote)
25 vues10 pagesvirtual instrumentation
Titre original
ch088
Copyright
© © All Rights Reserved
Formats disponibles
PDF, TXT ou lisez en ligne sur Scribd
Partager ce document
Partager ou intégrer le document
Avez-vous trouvé ce document utile ?
Ce contenu est-il inapproprié ?
Signaler ce documentvirtual instrumentation
Droits d'auteur :
© All Rights Reserved
Formats disponibles
Téléchargez comme PDF, TXT ou lisez en ligne sur Scribd
0 évaluation0% ont trouvé ce document utile (0 vote)
25 vues10 pagesThe Biomedical Engineering Handbook: Second Edition
Transféré par
Karthik Rajvvirtual instrumentation
Droits d'auteur :
© All Rights Reserved
Formats disponibles
Téléchargez comme PDF, TXT ou lisez en ligne sur Scribd
Vous êtes sur la page 1sur 10
Rosow, E., Adam . J . Virtual Instrumentation: Applications in Biomedical Engineering.
The Biomedical Engineering Handbook: Second Edition.
Ed. J oseph D. Bronzino
Boca Raton: CRC Press LLC, 2000
2000 by CRC Press LLC
88
Virtual
Instrumentation:
Applications in
Biomedical Engineering
88.1 Overview
A RevolutionGraphical Programming and Virtual
Instrumentation
88.2 Virtual Instrumentation and Biomedical
Engineering
Example 1 Example 2
88.1 Overview
A RevolutionGraphical Programming and Virtual Instrumentation
Over the last decade, the graphical programming revolution has empowered engineers to develop cus-
tomized systems, the same way the spreadsheet has empowered business managers to analyze nancial
data. This software technology has resulted in another type of revolutionthe virtual instrumentation
revolution, which is rapidly changing the instrumentation industry by driving down costs without
sacricing quality.
Virtual Instrumentation can be dened as:
A layer of software and/or hardware added to a general-purpose computer in such a fashion that users
can interact with the computer as though it were their own custom-designed traditional electronic
instrument.
Today, computers can serve as the engine for instrumentation. Virtual instruments utilize the open
architecture of industry-standard computers to provide the processing, memory, and display capabilities;
while the off-the-shelf, inexpensive interface boards plugged into an open bus, standardized communi-
cations bus provides the vehicle for the instruments capabilities. As a result, the open architecture of
PCs and workstations allow the functionality of virtual instruments to be user dened. In addition, the
processing power of virtual instruments is much greater than stand-alone instruments. This advantage
will continue to accelerate due to the rapid technology evolution of PCs and workstations that results
from the huge investments made in this industry.
The major benets of virtual instrumentation include increased performance and reduced costs. In
addition, because the user controls the technology through software, the exibility of virtual instrumentation
Eric Rosow
Hartford Hospital and Premise
Development Corporation
Joseph Adam
Premise Development Corporation
2000 by CRC Press LLC
is unmatched by traditional instrumentation. The modular, hierarchical programming environment of
virtual instrumentation is inherently reusable and recongurable.
88.2 Virtual Instrumentation and Biomedical Engineering
Virtual Instrumentation applications have encompassed nearly every industry including the telecommu-
nications, automotive, semiconductor, and biomedical industries. In the elds of healthcare and biomed-
ical engineering, virtual instrumentation has empowered developers and end-users to conceive of,
develop, and implement a wide variety of research-based biomedical applications and executive infor-
mation tools. These applications fall into several categories including: clinical research, equipment testing
and quality assurance, data management, and performance improvement.
In a collaborative approach, physicians, researchers, and biomedical and software engineers at Hartford
Hospital (Hartford, CT) and Premise Development Corporation (Avon, CT) have developed various data
acquisition and analysis systems that successfully integrate virtual instrumentation principles in a wide
variety of environments. These include:
The EndoTester, a patented quality assurance system for beroptic endoscopes
a Non-Invasive Pulmonary Diffusion and Cardiac Output Measurement System
a Cardiovascular Pressure-Dimension Analysis System
BioBench, a powerful turnkey application for physiological data acquisition and analysis
PIVIT, a Performance Indicator Virtual Instrument Toolkit to manage and forecast nancial
data
a Virtual Intelligence Program to manage the discrete components within the continuum of care
BabySave, an analysis and feedback system for apnea interruption via vibratory stimulation
This chapter will describe several of these applications and describe how they have allowed clinicians
and researchers to gain new insights, discover relationships that may not have been obvious, and test
and model hypotheses based on acquired data sets. In some cases, these applications have been developed
into commercial products to address test and measurement needs at other healthcare facilities throughout
the world.
Example 1: BioBenchA Virtual Instrument Application for Data
Acquisition and Analysis of Physiological Signals
The biomedical industry is an industry that relies heavily on the ability to acquire, analyze, and display
large quantities of data. Whether researching disease mechanisms and treatments by monitoring and
storing physiological signals, researching the effects of various drugs interactions, or teaching students
in labs where students study physiological signs and symptoms, it was clear that there existed a strong
demand for a exible, easy-to-use, and cost-effective tool. In a collaborative approach, biomedical engi-
neers, software engineers and clinicians, and researchers created a suite of virtual instruments called
BioBench.
BioBench (National Instruments, Austin, TX) is a new software application designed for physiolog-
ical data acquisition and analysis. It was built with LabVIEW, the worlds leading software development
environment for data acquisition, analysis, and presentation.
1
Coupled with National Instruments data
acquisition (DAQ) boards, BioBench integrates the PC with data acquisition for the life sciences market.
Many biologists and physiologists have made major investments over time in data acquisition hardware
built before the advent of modern PCs. While these scientists cannot afford to throw out their investment
in this equipment, they recognize that computers and the concept of virtual instrumentation yield
tremendous benets in terms of data analysis, storage, and presentation. In many cases, traditional
1
BioBench was developed for National Instruments (Austin, TX) by Premise Development Corporation (Avon,
CT).
2000 by CRC Press LLC
medical instrumentation may be too expensive to acquire and/or maintain. As a result, researchers and
scientists are opting to create their own PC-based data monitoring systems in the form of virtual
instruments.
Other life scientists, who are just beginning to assemble laboratory equipment, face the daunting task
of selecting hardware and software needed for their application. Many manufacturers for the life sciences
eld focus their efforts on the acquisition of raw signals and converting these signals into measurable
linear voltages. They do not concentrate on digitizing signals or the analysis and display of data on the
PC. BioBench is a low-cost turnkey package that requires no programming. BioBench is compatible
with any isolation amplier or monitoring instrument that provides an analog output signal. The user
can acquire and analyze data immediately because BioBench automatically recognizes and controls the
National Instruments DAQ hardware, minimizing conguration headaches.
Some of the advantages of PC-Based Data Monitoring include:
Easy-to-use-software applications
Large memory and the PCI bus
Powerful processing capabilities
Simplied customization and development
More data storage and faster data transfer
More efcient data analysis
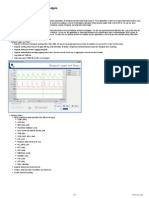
Figure 88.1 illustrates a typical setup of a data acquisition experiment using BioBench. BioBench also
features pull-down menus through which the user can congure devices. Therefore, those who have
made large capital investments can easily migrate their existing equipment into the computer age.
Integrating a combination of old and new physiological instruments from a variety of manufacturers is
an important and straightforward procedure. In fact, within the clinical and research setting, it is a
common requirement to be able to acquire multiple physiological signals from a variety of medical devices
and instruments which do not necessarily communicate with each other. Often times, this situation is
compounded by the fact that end-users would like to be able to view and analyze an entire waveform
and not just an average value. In order to accomplish this, the end-user must acquire multiple channels
of data at a relatively high sampling rate and have the ability to manage many large data les. BioBench
can collect up to 16 channels simultaneously at a sampling rate of 1000 Hz per channel. Files are stored
in an efcient binary format which signicantly reduces the amount of hard disk and memory require-
ments of the PC. During data acquisition, a number of features are available to the end-user. These
features include:
FIGURE 88.1
A typical biomedical application using BioBench (courtesy of National Instruments).
2000 by CRC Press LLC
Data Logging:
Logging can be enabled prior to or during an acquisition. The application will either
prompt the user for a descriptive lename or it can be congured to automatically assign a lename
for each acquisition. Turning the data logging option on and off creates a log data event record
that can be inspected in any of the analysis views of BioBench.
Event Logging:
The capacity to associate and recognize user commands associated with a data le
may be of signicant value. BioBench has been designed to provide this capability by automatically
logging user-dened events, stimulus events, and le logging events. With user-dened events, the
user can easily enter and associate date and time-stamped notes with user actions or specic
subsets of data. Stimulus events are also data and time-stamped and provide the user information
about whether a stimulus has been turned on or off. File logging events note when data has been
logged to disk. All of these types of events are stored with the raw data when logging data to le
and they can be searched for when analyzing data.
Alarming:
To alert the user about specic data values and thresholds, BioBench incorporates user-
dened alarms for each signal which is displayed. Alarms appear on the user interface during data
acquisition and notify the user that an alarm condition has occurred.
Figure 88.2 is an example of the Data Acquisition mode of BioBench. Once data has been acquired,
BioBench can employ a wide array of easy-to-use analysis features. The user has the choice of importing
recently acquired data or opening a data le that had been previously acquired for comparison or teaching
purposes. Once a data set has been selected and opened, BioBench allows the user to simply select and
highlight a region of interest and choose the analysis options to perform a specic routine.
BioBench implements a wide array of scalar and array analyses. For example, scalar analysis tools will
determine the minimum, maximum, mean, integral, and slope of a selected data set, while the array
analysis tools can employ Fast Fourier Transforms (FFTs), peak detection, histograms, and X vs. Y plots.
The ability to compare multiple data les is very important in analysis and BioBench allows the user
to open an unlimited number of data les for simultaneous comparison and analysis. All data les can
be scanned using BioBenchs search tools in which the user can search for particular events that are
FIGURE 88.2
BioBench acquisition mode with alarms enabled.
2000 by CRC Press LLC
associated with areas of interest. In addition, BioBench allows the user to employ lters and transforma-
tions to their data sets and all logged data can be easily exported to a spreadsheet or database for further
analysis. Finally, any signal acquired with BioBench can be played back, thus taking lab experience into
the classroom. Figure 88.3 illustrates the analysis features of BioBench.
Example 2: A Cardiovascular Pressure-Dimension Analysis System
Introduction
The intrinsic contractility of the heart muscle (myocardium) is the single most important determinant
of prognosis in virtually all diseases affecting the heart (e.g., coronary artery disease, valvular heart disease,
and cardiomyopathy). Furthermore, it is clinically important to be able to evaluate and track myocardial
function in other situations, including chemotherapy (where cardiac dysfunction may be a side effect of
treatment) and liver disease (where cardiac dysfunction may complicate the disease).
The most commonly used measure of cardiac performance is the ejection fraction. Although it does
provide some measure of intrinsic myocardial performance, it is also heavily inuenced by other factors
such as heart rate and loading conditions (i.e., the amount of blood returning to the heart and the
pressure against which the heart ejects blood).
Better indices of myocardial function based on the relationship between pressure and volume through-
out the cardiac cycle (pressurevolume loops) exist. However, these methods have been limited because
they require the ability to track ventricular volume continuously during rapidly changing loading con-
ditions. While there are many techniques to measure volume under steady state situations, or at end-
diastole and end-systole (the basis of ejection fraction determinations), few have the potential to record
volume during changing loading conditions.
Echocardiography can provide online images of the heart with high temporal resolution (typically 30
frames per second). Since echocardiography is radiation-free and has no identiable toxicity, it is ideally
suited to pressurevolume analyses. Until recently however, its use for this purpose has been limited by the
need for manual tracing of
the endocardial borders, an extremely tedious and time-consuming endeavor.
FIGURE 88.3
BioBench analysis mode.
2000 by CRC Press LLC
The System
Biomedical and software engineers at Premise Development Corporation (Avon, CT), in collaboration
with physicians and researchers at Hartford Hospital, have developed a sophisticated research application
called the Cardiovascular Pressure-Dimension Analysis (CPDA) System. The CPDA system acquires
echocardiographic volume and area information from the acoustic quantication (AQ) port, in conjunc-
tion with vetricular pressure(s) and ECG signals to rapidly perform pressurevolume and pressure-area
analyses. This fully automated system allows cardiologists and researchers to perform online pressure-
dimension and stroke work analyses during routine cardiac catheterizations and open-heart surgery. The
system has been designed to work with standard computer hardware. Analog signals for ECG, pressure,
and area/volume (AQ) are connected to a standard BNC terminal board. Automated calibration routines
ensure that each signal is properly scaled and allows the user to immediately collect and analyze pressure-
dimension relationships.
The CPDA can acquire up to 16 channels of data simultaneously. Typically, only three physiological
parameters, ECG, pressure, and the AQ signals are collected using standard data acquisition hardware.
In addition, the software is capable of running on multiple operating systems including Macintosh,
Windows 95/98/NT, and Solaris. The CPDA also takes advantage of the latest hardware developments
and form-factors and can be used with either a desktop or a laptop computer.
The development of an automated, online method of tracing endocardial borders (Hewlett Packards
AQ Technology) (Hewlett-Packard Medical Products Group, Andover, MA) has provided a method for
rapid online area and volume determinations. Figure 88.4 illustrates this AQ signal from a Hewlett
Packard Sonos Ultrasound Machine. This signal is available as an analog voltage (1 to +1 volts) through
the Sonos Dataport option (BNC connector).
Data Acquisition and Analysis
Upon launching this application, the user is presented with a dialog box that reviews the license agreement
and limited warranty. Next, the Main Menu is displayed, allowing the user to select from one of six
options as shown in Fig. 88.5.
Clinical Signicance
Several important relationships can be derived from this system. Specically, a parameter called the
End-
Systolic Pressure-Volume Relationship
(
ESPVR
)
describes the line of best t through the peak-ratio (max-
imum pressure with respect to minimum volume) coordinates from a series of pressurevolume loops
FIGURE 88.4
The Acoustic Quantication (AQ) signal (Hewlett Packard).
2000 by CRC Press LLC
generated under varying loading conditions. The slope of this line has been shown to be a sensitive index
of myocardial contractility that is independent of loading conditions. In addition, several other analyses,
including
time varying elastance
(
E
max
)
and
stroke work,
are calculated.
Time-varying elastance is measured
by determining the maximum slope of a regression line through a series of isochronic pressurevolume
coordinates. Stroke work is calculated by quantifying the area of each pressurevolume loop. Statistical
parameters are also calculated and displayed for each set of data. Figure 88.7 illustrates the pressure-
dimension loops and each of the calculated parameters along with the various analysis options. Finally,
the user has the ability to export data sets into spreadsheet and database les and export graphs and
indicators into third-party presentation software packages such as Microsoft PowerPoint.
FIGURE 88.5
Cardiovascular pressure-dimension analysis main menu.
FIGURE 88.6
The data selection front panel.
2000 by CRC Press LLC
Summary
Virtual Instrumentation allows the development and implementation of innovative and cost-effective
biomedical applications and information management solutions. As the healthcare industry continues
to respond to the growing trends of managed care and capitation, it is imperative for clinically useful,
cost-effective technologies to be developed and utilized. As application needs will surely continue to
change, virtual instrumentation systems will continue to offer users exible and powerful solutions
without requiring new equipment or traditional instruments.
References
1. Fisher, J.P., Mikan, J.S., Rosow, E., Nagle, J., Fram, D.B., Maffucci, L.M., McKay, R.G., and Gillam,
L.D., Pressure-Dimension Analysis of Regional Left Ventricular Performance Using Echocardio-
graphic Automatic Boundary Detection: Validation in an Animal Model of Inotropic Modulation,
J. Am. College of Cardiol.,
19(3), 262A, 1992.
2. Fisher, J.P., McKay, R.G., Mikan, J.S., Rosow, E., Nagle, J., Mitchel, J.F., Kiernan, F.J., Hirst, J.A.,
Primiano, C.A., Fram, D.B., Gillam, L.D., Hartford Hospital and University of Connecticut, Hart-
ford, CT,
Human Left Ventricular Pressure-Area and Pressure-Volume Analysis Using Echocardio-
graphic Automatic Boundary Detection.
65th Scientic Session of the American Heart Association
(11/92).
3. Fisher, J.P., Mitchel, J.F., Rosow, E., Mikan, J.S., Nagle, J., Kiernan, F.J., Hirst, J.A., Primiano, Gillam,
L.D., Hartford Hospital and University of Connecticut, Hartford, CT,
Evaluation of Left Ventricular
Diastolic Pressure-Area Relations with Echocardiographic Automatic Boundary Detection,
65th Sci-
entic Session of the American Heart Association (11/92).
4. Fisher, J.P., McKay, R.G., Mikan, J.S., Rosow, E., Nagle, J., Mitchel, J.F., Fram, D.B., Gillam, L.D.,
Hartford Hospital,
A Comparison of Echocardiographic Methods of Evaluating Regional LV Systolic
Function: Fractional Area Change vs. the End-Systolic Pressure-Area Relation.
65th Scientic Session
of the American Heart Association (11/92).
FIGURE 88.7
The cardiac cycle analysis front panel.
2000 by CRC Press LLC
5. Fisher, J.P., McKay, R.G., Rosow, E. Mikan, J. Nagle, J. Hirst, J.A., Fram, D.B., Gillam, L.D.,
On-
Line Derivation of Human Left Ventricular Pressure-Volume Loops and Load Independent Indices of
Contractility Using Echocardiography with Automatic Boundary Detection-A Clinical Reality.
Cir-
culation 88, I-304, 1993.
6. Fisher, J.P., Chen, C., Krupowies, N., Li Li, Kiernan, F.J., Fram, D.B., Rosow, E., Adam, J., Gillam,
L.D.,
Comparison of Mechanical and Pharmacologic Methods of Altering Loading Conditions to
Determine End-Systolic Indices of Left Ventricle Function.
Circulation 90 (II), 1-494, 1994.
7.
Fisher, JP, Martin, J., Day FP, Rosow, E., Adam J, Chen C, Gillam LD. Validation of a Less Invasive
Method for Determining Preload Recruitable Stroke Work Derived with Echocardiographic Auto-
matic Boundary Detection. Circulation 92, 1-278, 1995.
8. Fontes ML. Adam J, Rosow E, Mathew J, DeGraff AC. Non-Invasive Cardiopulmonary Function
Assessment System, J Clin Monit 13, 413, 1997.
9. Johnson, GW, LabVIEW Graphical Programming: Practical Applications in Instrumentation and
Control, 2nd ed., McGraw-Hill, New York, 1997.
10. Mathew JP, Adam J, Rosow E, Fontes ML, Davis L, Barash PG, Gillam L., Cardiovascular Pres-
sure-Dimension Analysis System, J Clin Monit 13, 423, 1997.
11. National Instruments 1999 Measurement and Automation Catalog, National Instruments, Austin,
TX.
12. Rosow, E. Technology and Sensors, Presented at the United States Olympic Committees Sports
Equipment and Technology Conference, Colorado Springs, CO; November 19-23, 1992.
13. Rosow, E. Biomedical Applications using LabVIEW, Presented at the New England Society of
Clinical Engineering, Sturbridge, MA; November 14, 1993.
14. Rosow, E., Adam, J.S., Nagle, J., Fisher, J.P. and Gillam, L.D.; Premise Development Corporation,
Avon, CT and Hartford Hospital, Hartford, CT, A Cardiac Analysis System: LabVIEW and Acoustic
Quantication (AQ), Presented at the Association for the Advancement of Medical Instrumenta-
tion in Washington, D.C. May 21-25, 1994.
Vous aimerez peut-être aussi
- The Subtle Art of Not Giving a F*ck: A Counterintuitive Approach to Living a Good LifeD'EverandThe Subtle Art of Not Giving a F*ck: A Counterintuitive Approach to Living a Good LifeÉvaluation : 4 sur 5 étoiles4/5 (5794)
- The Gifts of Imperfection: Let Go of Who You Think You're Supposed to Be and Embrace Who You AreD'EverandThe Gifts of Imperfection: Let Go of Who You Think You're Supposed to Be and Embrace Who You AreÉvaluation : 4 sur 5 étoiles4/5 (1090)
- Never Split the Difference: Negotiating As If Your Life Depended On ItD'EverandNever Split the Difference: Negotiating As If Your Life Depended On ItÉvaluation : 4.5 sur 5 étoiles4.5/5 (838)
- Hidden Figures: The American Dream and the Untold Story of the Black Women Mathematicians Who Helped Win the Space RaceD'EverandHidden Figures: The American Dream and the Untold Story of the Black Women Mathematicians Who Helped Win the Space RaceÉvaluation : 4 sur 5 étoiles4/5 (895)
- Grit: The Power of Passion and PerseveranceD'EverandGrit: The Power of Passion and PerseveranceÉvaluation : 4 sur 5 étoiles4/5 (588)
- Shoe Dog: A Memoir by the Creator of NikeD'EverandShoe Dog: A Memoir by the Creator of NikeÉvaluation : 4.5 sur 5 étoiles4.5/5 (537)
- The Hard Thing About Hard Things: Building a Business When There Are No Easy AnswersD'EverandThe Hard Thing About Hard Things: Building a Business When There Are No Easy AnswersÉvaluation : 4.5 sur 5 étoiles4.5/5 (345)
- Elon Musk: Tesla, SpaceX, and the Quest for a Fantastic FutureD'EverandElon Musk: Tesla, SpaceX, and the Quest for a Fantastic FutureÉvaluation : 4.5 sur 5 étoiles4.5/5 (474)
- Her Body and Other Parties: StoriesD'EverandHer Body and Other Parties: StoriesÉvaluation : 4 sur 5 étoiles4/5 (821)
- The Emperor of All Maladies: A Biography of CancerD'EverandThe Emperor of All Maladies: A Biography of CancerÉvaluation : 4.5 sur 5 étoiles4.5/5 (271)
- The Sympathizer: A Novel (Pulitzer Prize for Fiction)D'EverandThe Sympathizer: A Novel (Pulitzer Prize for Fiction)Évaluation : 4.5 sur 5 étoiles4.5/5 (121)
- The Little Book of Hygge: Danish Secrets to Happy LivingD'EverandThe Little Book of Hygge: Danish Secrets to Happy LivingÉvaluation : 3.5 sur 5 étoiles3.5/5 (400)
- The World Is Flat 3.0: A Brief History of the Twenty-first CenturyD'EverandThe World Is Flat 3.0: A Brief History of the Twenty-first CenturyÉvaluation : 3.5 sur 5 étoiles3.5/5 (2259)
- The Yellow House: A Memoir (2019 National Book Award Winner)D'EverandThe Yellow House: A Memoir (2019 National Book Award Winner)Évaluation : 4 sur 5 étoiles4/5 (98)
- Devil in the Grove: Thurgood Marshall, the Groveland Boys, and the Dawn of a New AmericaD'EverandDevil in the Grove: Thurgood Marshall, the Groveland Boys, and the Dawn of a New AmericaÉvaluation : 4.5 sur 5 étoiles4.5/5 (266)
- A Heartbreaking Work Of Staggering Genius: A Memoir Based on a True StoryD'EverandA Heartbreaking Work Of Staggering Genius: A Memoir Based on a True StoryÉvaluation : 3.5 sur 5 étoiles3.5/5 (231)
- Team of Rivals: The Political Genius of Abraham LincolnD'EverandTeam of Rivals: The Political Genius of Abraham LincolnÉvaluation : 4.5 sur 5 étoiles4.5/5 (234)
- Canine and Feline Endocrinology PDFDocument685 pagesCanine and Feline Endocrinology PDFSandra Solano100% (8)
- On Fire: The (Burning) Case for a Green New DealD'EverandOn Fire: The (Burning) Case for a Green New DealÉvaluation : 4 sur 5 étoiles4/5 (74)
- The Unwinding: An Inner History of the New AmericaD'EverandThe Unwinding: An Inner History of the New AmericaÉvaluation : 4 sur 5 étoiles4/5 (45)
- Bio Lab ReportDocument7 pagesBio Lab Reportjoylee05Pas encore d'évaluation
- Sensors: Breathing Analysis Using Thermal and Depth Imaging Camera Video RecordsDocument10 pagesSensors: Breathing Analysis Using Thermal and Depth Imaging Camera Video RecordsKarthik RajvPas encore d'évaluation
- Non Contact Measurement Vital ParametersDocument108 pagesNon Contact Measurement Vital ParametersKarthik RajvPas encore d'évaluation
- Sensors: Absorbance Based Light Emitting Diode Optical Sensors and Sensing DevicesDocument27 pagesSensors: Absorbance Based Light Emitting Diode Optical Sensors and Sensing DevicesKarthik RajvPas encore d'évaluation
- Manual ElectrodiagnosticsAndElectrotherapyDocument5 pagesManual ElectrodiagnosticsAndElectrotherapyKarthik RajvPas encore d'évaluation
- Assignment 1Document2 pagesAssignment 1Karthik RajvPas encore d'évaluation
- Very Imp ConceptDocument4 pagesVery Imp ConceptKarthik RajvPas encore d'évaluation
- Section11 AkDocument6 pagesSection11 AkKarthik RajvPas encore d'évaluation
- Ultrasound in Gynecology N ObstetricDocument9 pagesUltrasound in Gynecology N ObstetricKarthik RajvPas encore d'évaluation
- NI Tutorial 9037 enDocument4 pagesNI Tutorial 9037 enKarthik RajvPas encore d'évaluation
- 7.014 Problem Set 4Document9 pages7.014 Problem Set 4Karthik RajvPas encore d'évaluation
- Psych Motivation & EmotionsDocument13 pagesPsych Motivation & EmotionsnasibdinPas encore d'évaluation
- 11 Ch1208 5127 PDFDocument25 pages11 Ch1208 5127 PDFThamarai ElanthirayanPas encore d'évaluation
- EJMC1999Document1 011 pagesEJMC1999Mini MinutePas encore d'évaluation
- Uropati ObstruktifDocument22 pagesUropati ObstruktifEvan Permana100% (1)
- Synechococcus SP On The Growth and Production of OilDocument5 pagesSynechococcus SP On The Growth and Production of OilIrawan DwiPas encore d'évaluation
- Anatomy and Physiology of NeuronsDocument4 pagesAnatomy and Physiology of NeuronsVan Jasper Pimentel BautistaPas encore d'évaluation
- Pathology of Neurodegenerative DiseasesDocument22 pagesPathology of Neurodegenerative DiseasesKarel GuevaraPas encore d'évaluation
- And Um-101Document3 pagesAnd Um-101ver_at_workPas encore d'évaluation
- Edexcel GCSE Science Topic B1.1 Classification, Variation and Inheritance Test 13 - 14Document10 pagesEdexcel GCSE Science Topic B1.1 Classification, Variation and Inheritance Test 13 - 14Paul BurgessPas encore d'évaluation
- Heat Stress Management Training ProgramDocument28 pagesHeat Stress Management Training ProgramDheeraj Dheeru100% (1)
- Antioxidants !! The Great DiscoveryDocument3 pagesAntioxidants !! The Great DiscoverypatPas encore d'évaluation
- Pathoma, Sketchy Pharm & Micro, B&B Video TimesDocument52 pagesPathoma, Sketchy Pharm & Micro, B&B Video TimesAlina PolancoPas encore d'évaluation
- 07 Bibliography FormatDocument14 pages07 Bibliography FormatArjun KaflePas encore d'évaluation
- MCQ Answer KeyDocument46 pagesMCQ Answer KeyDr. RamadanPas encore d'évaluation
- Complex Anorectal Disorders PDFDocument817 pagesComplex Anorectal Disorders PDFGabriela Balan100% (2)
- Vital SignsDocument3 pagesVital SignsNicole NipasPas encore d'évaluation
- Faisalabad Board: Grade 9 Biology 2016 GROUP 1Document11 pagesFaisalabad Board: Grade 9 Biology 2016 GROUP 1DEPUTY DIRECTOR SOCIAL WELFAREPas encore d'évaluation
- Fluids and Transport: Fundamentals of Anatomy & PhysiologyDocument161 pagesFluids and Transport: Fundamentals of Anatomy & Physiologyann aquinoPas encore d'évaluation
- Anatomy AIIMSDocument18 pagesAnatomy AIIMSvkPas encore d'évaluation
- Fudamentals of Biomechanics and Biomechanics of Levelling and Aligning (Includes Biomechanics of Molar Control)Document326 pagesFudamentals of Biomechanics and Biomechanics of Levelling and Aligning (Includes Biomechanics of Molar Control)Margareth Brito100% (4)
- Motivation - BPTDocument7 pagesMotivation - BPTRenukaPas encore d'évaluation
- B1 Quiz 12Document2 pagesB1 Quiz 12giulianotboPas encore d'évaluation
- Endocrine and Metabolic Disorders PDFDocument31 pagesEndocrine and Metabolic Disorders PDFAna-Maria50% (2)
- Cardio CaseDocument21 pagesCardio CaseJan Rae Y. BattungPas encore d'évaluation
- FA 2014 RenalRespDocument118 pagesFA 2014 RenalResprvar839Pas encore d'évaluation
- Acupuncture Final Exam ReviewDocument16 pagesAcupuncture Final Exam Reviewpeter911xPas encore d'évaluation
- Human Body EssayDocument1 pageHuman Body EssayAtlas CruxPas encore d'évaluation
- Fisioterapi JurnalDocument7 pagesFisioterapi JurnalAchenk BarcelonistaPas encore d'évaluation