Académique Documents
Professionnel Documents
Culture Documents
Worksheet 30
Transféré par
Vijay BhaskarDescription originale:
Titre original
Copyright
Formats disponibles
Partager ce document
Partager ou intégrer le document
Avez-vous trouvé ce document utile ?
Ce contenu est-il inapproprié ?
Signaler ce documentDroits d'auteur :
Formats disponibles
Worksheet 30
Transféré par
Vijay BhaskarDroits d'auteur :
Formats disponibles
30 Worksheet (A2)
Data needed to answer questions can be found in the Data, formulae and relationships sheet.
1 a Write down Einsteins famous equation relating mass and energy. [1]
b Determine the change in energy equialent to a change in mass!
i of 1." g [#]
ii equal to that of an electron $%.1 1"
&'1
(g). [#]
2 *n nuclear physics, it is common practice to quote the mass of a nuclear particle in terms of
the unified atomic mass unit, u. +he unified atomic mass unit u is defined as one,twelfth of the
mass of an atom of the carbon isotope -
1#
.
.
a Determine the mass of each of the following particles in terms of u!
i an /,particle of mass ...0 1"

(g [1]
ii a carbon,1' atom of mass #.1. 1"
&#.
(g. [1]
b Determine the mass of each of the following particles in (ilograms!
i a proton of mass 1."1 u [1]
ii a uranium,#'0 nucleus of mass #'2.%% u. [1]
3 3tate three quantities consered in all nuclear reactions. [']
4 a E4plain why e4ternal energy is required to 5split a nucleus. [1]
b Define the binding energy of a nucleus. [1]
c +he binding energy of the nuclide 6
1.
7
is 1#7 8e9. -alculate the binding energy
per nucleon. [#]
5 a Define the half-life of a radioactie isotope. [1]
b +he half,life of a particular isotope is #" minutes. : sample initially contains N
"
nuclei
of this isotope. Determine the number of nuclei of the isotope left in the sample after!
i #" minutes [1]
ii 1." hour. [#]
6 +he actiity of an /,source is 02" ;q. +he (inetic energy of each /,particle is 7.. < 1"
&12
=.
+he isotope in the source has a ery long half,life.
a -alculate the number of /,particles emitted by the source in!
i 1 second [1]
ii 1 hour. [1]
b Determine the total energy released by the source in a time of 1 second. [']
c 3tate the rate at which energy is emitted from this /,source. [1]
:3 and : >eel ?hysics 6riginal material @ -ambridge Aniersity ?ress #"1" 1
30 Worksheet (A2)
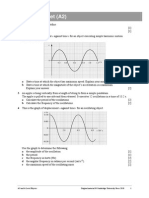
7 +he binding energy per nucleon against nucleon number graph for some common nuclides is
shown below.
a *dentify the most stable nuclide. E4plain your answer. [#]
b Ase the graph to estimate the binding energy for the nucleus of -
1#
.
. [#]
c Ase the graph to estimate the energy released in the following fusion reaction. [2]
Be B B
2
#
#
1
#
1
+
d +he fusion reaction shown in c is one of the many that occur in the interior of stars.
3tate the conditions necessary to initiate such reactions in stars. [#]
8 Ase the data gien below to determine the binding energy and the binding energy per
nucleon of the nuclide A
#'0
%#
. [1]
mass of proton = 1.""1 u
mass of neutron = 1.""% u mass of uranium,#'0 nucleus = #'2.%%# u
:3 and : >eel ?hysics 6riginal material @ -ambridge Aniersity ?ress #"1" 2
30 Worksheet (A2)
9 a Describe the process of induced nuclear fission. [1]
b +he diagram shows the fission of uranium,#'0 in accordance with the nuclear equation!
n # Ce 3r n A
1
"
1'%
02
%0
'7
1
"
#'0
%#
+ + +
i -opy the diagram, adding labels to identify the neutrons, the strontium nuclide and
the 4enon nuclide. [1]
ii E4plain why energy is released in the reaction aboe. [#]
iii Ase the following data to determine the energy released in a single fission reaction
inoling A
#'0
%#
and n
1
"
. [0]
mass of A
#'0
%#
= '.%"# 1"
�
(g
mass of n
1
"
= 1..10 1"

(g
mass of 3r
%0
'7
= 1.010 1"
�
(g
mass of Ce
1'%
02
= #.'". 1"
�
(g
10 6ne of the neutron,induced fission reactions of uranium,#'0 may be represented by the
following nuclear equations.
A
#'0
%#
D n
1
"
A
#'.
%#
A
#'.
%#
>a
12.
01
D ;r
71
'0
D ' n
1
"
+he binding energies per nucleon for these nuclides are!
A,
#'.
%#
1.0% 8e9E >a,
12.
01
7.21 8e9E ;r,
71
'0
7.0% 8e9.
-alculate the energy released in 8e9 when the A
#'.
%#
nucleus undergoes fission. [']
11 +he half,life of the radon isotope Fn
##"
7.
is 0. s.
a Determine the decay constant in s
&1
. [']
b -alculate the actiity of a sample containing .." 1"
1"
nuclei of Fn
##"
7.
. [']
12 +he actiity of a radioactie source containing 7." 1"
12
undecayed nuclei is 0." 1"
%
;q.
a Determine the decay constant in s
&1
. [']
b -alculate the half,life of the nucleus. [']
c Bow many undecayed nuclei will be left after 2" hoursG [']
13 a Define the decay constant of a nucleus. [1]
b +he thorium isotope +h
##1
%"
has a half,life of 17 days.
: particular radioactie source contains 2." 1"
1#
nuclei of the isotope +h
##1
%"
.
i Determine the decay constant for the thorium isotope +h
##1
%"
in s
&1
. [']
ii What is the initial actiity of the sourceG [']
iii -alculate the actiity of the source after '. days. [#]
14 : sample of roc( is (nown to contain 1." Hg of the radioactie radium isotope Fa
##.
77
.
+he half,life of this particular isotope is 1."" years. +he molar mass of radium,##. is ##. g.
a Determine the number of nuclei of the isotope Fa
##.
77
in the roc( sample. [#]
b -alculate the actiity from decay of the radium,##. in the sample. [']
:3 and : >eel ?hysics 6riginal material @ -ambridge Aniersity ?ress #"1" 3
30 Worksheet (A2)
15 *n a process referred to as 5annihilation, a particle interacts with its antiparticle and the
entire mass of the combined particles is transformed into energy in the form of photons.
+he following equation represents the interaction of a proton $p) and its antiparticle, the
antiproton $
p
).
I I p p
1
1
1
1
+ +
+he antiproton has the same mass as a proton J the only difference is that it has a negatie
charge. Determine the waelength of each of the two identical photons emitted in the
reaction aboe. $8ass of a proton = 1.1 1"

(g.) [0]
16 Does fusion or fission produce more energy per (ilogram of fuelG :nswer this question by
considering the fusion reaction in 7 c and the fission reaction in 9 b.
$+he molar masses of hydrogen,# and uranium,#'0 are # g and #'0 g, respectiely.) [1]
17 3ome astronomers beliee that our solar system was formed 0." < 1"
%
years ago.
:ssuming that all uranium,#'7 nuclei were formed before this time, what fraction of the
original uranium,#'7 nuclei remain in the solar system todayG +he isotope of uranium
A
#'7
%#
has a half,life of 2.0 1"
%
y. [2]
+otal!
1""
3core! K
:3 and : >eel ?hysics 6riginal material @ -ambridge Aniersity ?ress #"1" 4
Vous aimerez peut-être aussi
- The Elements from Neutron to Magnesium: Nuclear ReactionsD'EverandThe Elements from Neutron to Magnesium: Nuclear ReactionsPas encore d'évaluation
- Worksheet 30 PDFDocument4 pagesWorksheet 30 PDFVijay Bhaskar100% (3)
- Encircle The Letter That Corresponds To The Best Answer To Each of The Following Questions. You May Use The Back Pages For Scratch WorkDocument3 pagesEncircle The Letter That Corresponds To The Best Answer To Each of The Following Questions. You May Use The Back Pages For Scratch WorkMarc RitzPas encore d'évaluation
- Unit 7 Test MC Answers and ExplanationDocument25 pagesUnit 7 Test MC Answers and ExplanationOnur YavuzcetinPas encore d'évaluation
- Nuclear Chem Notes 14-15Document3 pagesNuclear Chem Notes 14-15api-262718384Pas encore d'évaluation
- Chapter 6 - Jan14 New VersionDocument131 pagesChapter 6 - Jan14 New VersionkumuthaPas encore d'évaluation
- Nuclei: - . - Types of Elements: Isotopes: IsobarsDocument12 pagesNuclei: - . - Types of Elements: Isotopes: IsobarsrgryjhgsdvrtPas encore d'évaluation
- Nuclear Physics: (I) Atomic NumberDocument6 pagesNuclear Physics: (I) Atomic NumberrujintoPas encore d'évaluation
- Science Department: Topic 7.1 - Discrete Energy & Radioactivity Topic 7.2 - Nuclear Reactions 12 Grade (Physics SL)Document8 pagesScience Department: Topic 7.1 - Discrete Energy & Radioactivity Topic 7.2 - Nuclear Reactions 12 Grade (Physics SL)ananPas encore d'évaluation
- Microsoft Word - E2NuclearDocument14 pagesMicrosoft Word - E2NuclearMcKaylinPas encore d'évaluation
- Nuclei: One Mark QuestionsDocument8 pagesNuclei: One Mark QuestionsBrittany GreenPas encore d'évaluation
- Nuclear DataDocument35 pagesNuclear DataOMARPas encore d'évaluation
- Lesson 20-10-2020-Nuclear ChemistryDocument10 pagesLesson 20-10-2020-Nuclear ChemistryAlemkeng BrendaPas encore d'évaluation
- 15 Radio ActivityDocument7 pages15 Radio ActivityHasmukh ShahPas encore d'évaluation
- Nuclear Physics Notes For A-Level by HubbakDocument33 pagesNuclear Physics Notes For A-Level by HubbakHubbak Khan100% (1)
- Nuclei QB XiiDocument23 pagesNuclei QB XiiToshani GuptaPas encore d'évaluation
- Exercise 1 - GR 10 - Physics (Sec 5 Nuclear Physics)Document10 pagesExercise 1 - GR 10 - Physics (Sec 5 Nuclear Physics)oktavianusjordanPas encore d'évaluation
- Nuclei: Atomic Mass Unit (U)Document10 pagesNuclei: Atomic Mass Unit (U)Jatin PanjwaniPas encore d'évaluation
- RadioactivityDocument16 pagesRadioactivityEng BahanzaPas encore d'évaluation
- MODULE 3 Nuclear ChemistryDocument28 pagesMODULE 3 Nuclear ChemistryILIVEFOR MONSTA7Pas encore d'évaluation
- Worksheet 29Document4 pagesWorksheet 29Vijay BhaskarPas encore d'évaluation
- Nuclear AssignmentDocument10 pagesNuclear AssignmentMuhammad Haroon KhanPas encore d'évaluation
- 02 - Atomic Structure CORNELLDocument11 pages02 - Atomic Structure CORNELLGeorge SolomouPas encore d'évaluation
- Introductory Nuclear PhysicsDocument21 pagesIntroductory Nuclear PhysicsDavid BrownPas encore d'évaluation
- Nuclear Physics: Basic Properties of NucleusDocument17 pagesNuclear Physics: Basic Properties of NucleusSK. NAFISH SADIQUE AYON 1903043Pas encore d'évaluation
- Nuclear Physics Sheet TDocument15 pagesNuclear Physics Sheet T06 boymaxxPas encore d'évaluation
- Chapitre 1 1Document15 pagesChapitre 1 1Ri HabPas encore d'évaluation
- Nuclear Chemistry PDFDocument6 pagesNuclear Chemistry PDFlesterPas encore d'évaluation
- CBSE Class 12 Physics: Atoms and Nuclei QuestionsDocument4 pagesCBSE Class 12 Physics: Atoms and Nuclei QuestionsBug LordPas encore d'évaluation
- Nuclear Tutorial SolutionDocument19 pagesNuclear Tutorial SolutionGordon GohPas encore d'évaluation
- Nuclear Energy (Radioactivity, Nuclear Fission and Fusion)Document14 pagesNuclear Energy (Radioactivity, Nuclear Fission and Fusion)Mohammad Irfan YousufPas encore d'évaluation
- Form 5 Physics Chapter 5 - Teacher'sDocument12 pagesForm 5 Physics Chapter 5 - Teacher'sPavithiran100% (5)
- UNIT III Nuclear ChemistryDocument6 pagesUNIT III Nuclear ChemistrySivakumar PonnusamyPas encore d'évaluation
- NucleusDocument57 pagesNucleusSuhail KunjavaPas encore d'évaluation
- Notes On Nuclear PhysicsDocument12 pagesNotes On Nuclear PhysicsBS PhysicsPas encore d'évaluation
- High Energy Physics MCQDocument9 pagesHigh Energy Physics MCQpriyaPas encore d'évaluation
- Nuclear Physics: Decay RateDocument36 pagesNuclear Physics: Decay RateINNOCENT KHULIWAPas encore d'évaluation
- Share CE-112-MODULE-5-NUCLEAR-CHEM-usedDocument30 pagesShare CE-112-MODULE-5-NUCLEAR-CHEM-usedAngelo GranadaPas encore d'évaluation
- Chem Form 6 Lessons 1Document3 pagesChem Form 6 Lessons 1mona lisaPas encore d'évaluation
- 02 Moden Physics 2exercise PC 1668951850787Document34 pages02 Moden Physics 2exercise PC 1668951850787bhimbasuliPas encore d'évaluation
- Important Formula For Numerical:-: 4 Einstein S Mass Energy Equivalence Relation Is E MCDocument28 pagesImportant Formula For Numerical:-: 4 Einstein S Mass Energy Equivalence Relation Is E MCdhirendrasisodiaPas encore d'évaluation
- Modern Physics-06-Subjective Unsolved LevelDocument3 pagesModern Physics-06-Subjective Unsolved LevelRaju SinghPas encore d'évaluation
- 12-Ph 19908Document26 pages12-Ph 19908Surendra SinghPas encore d'évaluation
- Atoms & NucleiDocument15 pagesAtoms & NucleixkryxxzPas encore d'évaluation
- TNOW Homework Assignment 5: Chapter 2 Sections 1 & 2: Atomic Structure and Subatomic Particles: The Nuclear AtomDocument10 pagesTNOW Homework Assignment 5: Chapter 2 Sections 1 & 2: Atomic Structure and Subatomic Particles: The Nuclear Atomabc 123Pas encore d'évaluation
- Fisika DasarDocument5 pagesFisika DasarBulan NiaPas encore d'évaluation
- Atomic StructureDocument30 pagesAtomic Structurenaeem mushtaqPas encore d'évaluation
- SR Inter IPE Question Bank Chapter-XIV (NUCLEI)Document18 pagesSR Inter IPE Question Bank Chapter-XIV (NUCLEI)sojakoj867Pas encore d'évaluation
- STD 8 Chapter 5Document6 pagesSTD 8 Chapter 5ROHIT KADAMPas encore d'évaluation
- 14 - Nucleus 14.1 Nucleus - General InformationDocument12 pages14 - Nucleus 14.1 Nucleus - General InformationHrishabh GuptaPas encore d'évaluation
- 14 15 H2 Nuclear Updated Full NotesDocument22 pages14 15 H2 Nuclear Updated Full NotesAgus LeonardiPas encore d'évaluation
- Bansal Nuclear PhysicsDocument16 pagesBansal Nuclear Physicsbhnprtp90Pas encore d'évaluation
- Chapter - 19 The Atomic NucleusDocument10 pagesChapter - 19 The Atomic Nucleususama113Pas encore d'évaluation
- CBSE TERM2 - NucleiDocument50 pagesCBSE TERM2 - Nucleivenom ePas encore d'évaluation
- NucleiDocument50 pagesNucleiGiridhar MeruvalaPas encore d'évaluation
- A Level - Chemistry, George FacerDocument147 pagesA Level - Chemistry, George FacerHeba Ali100% (4)
- Atom, Atom Element, Compound, Diffusion, Brownian MotionDocument37 pagesAtom, Atom Element, Compound, Diffusion, Brownian MotionMohammad khalidPas encore d'évaluation
- Nuclei 22912Document7 pagesNuclei 22912user 003Pas encore d'évaluation
- Chapter 8: Nucleus: 8.1 Properties of Nucleus 8.2 Binding Energy and Mass DefectDocument70 pagesChapter 8: Nucleus: 8.1 Properties of Nucleus 8.2 Binding Energy and Mass DefectJoanne SohPas encore d'évaluation
- Worksheet 33Document3 pagesWorksheet 33Pathmanathan NadesonPas encore d'évaluation
- Worksheet 32Document2 pagesWorksheet 32Vijay BhaskarPas encore d'évaluation
- Worksheet 32Document2 pagesWorksheet 32Pathmanathan NadesonPas encore d'évaluation
- Transformationsclassifieds PDFDocument35 pagesTransformationsclassifieds PDFVijay BhaskarPas encore d'évaluation
- Worksheet 31Document3 pagesWorksheet 31Vijay BhaskarPas encore d'évaluation
- Worksheet 29Document4 pagesWorksheet 29Vijay BhaskarPas encore d'évaluation
- Worksheet 26Document3 pagesWorksheet 26Vijay BhaskarPas encore d'évaluation
- Worksheet 26 PDFDocument3 pagesWorksheet 26 PDFVijay BhaskarPas encore d'évaluation
- 27 Worksheet (A2) : AS and A Level Physics Original Material © Cambridge University Press 2010Document3 pages27 Worksheet (A2) : AS and A Level Physics Original Material © Cambridge University Press 2010Syed Abdul Rehman ShahPas encore d'évaluation
- Worksheet 20Document3 pagesWorksheet 20Vijay BhaskarPas encore d'évaluation
- Worksheet 29 PDFDocument4 pagesWorksheet 29 PDFVijay BhaskarPas encore d'évaluation
- Worksheet 28 PDFDocument2 pagesWorksheet 28 PDFVijay Bhaskar100% (3)
- Worksheet 25Document3 pagesWorksheet 25Vijay Bhaskar100% (1)
- Worksheet 27Document3 pagesWorksheet 27Vijay Bhaskar0% (1)
- 22 Worksheet (A2) : AS and A Level Physics Original Material © Cambridge University Press 2010Document3 pages22 Worksheet (A2) : AS and A Level Physics Original Material © Cambridge University Press 2010Vijay BhaskarPas encore d'évaluation
- Worksheet 23 PDFDocument2 pagesWorksheet 23 PDFVijay Bhaskar100% (1)
- Worksheet 25 PDFDocument3 pagesWorksheet 25 PDFVijay Bhaskar100% (2)
- Worksheet 24 PDFDocument3 pagesWorksheet 24 PDFVijay BhaskarPas encore d'évaluation
- Worksheet 21 PDFDocument3 pagesWorksheet 21 PDFVijay Bhaskar0% (1)
- Worksheet 23Document2 pagesWorksheet 23Vijay BhaskarPas encore d'évaluation
- Worksheet 24Document3 pagesWorksheet 24Vijay BhaskarPas encore d'évaluation
- Worksheet 17 PDFDocument2 pagesWorksheet 17 PDFVijay Bhaskar0% (1)
- Worksheet 19Document2 pagesWorksheet 19etud3cl100% (1)
- Worksheet 20 PDFDocument3 pagesWorksheet 20 PDFVijay BhaskarPas encore d'évaluation
- Worksheet 19Document2 pagesWorksheet 19Vijay BhaskarPas encore d'évaluation
- Worksheet 17Document2 pagesWorksheet 17Vijay BhaskarPas encore d'évaluation
- Worksheet 18Document4 pagesWorksheet 18Vijay BhaskarPas encore d'évaluation
- Worksheet 18 PDFDocument3 pagesWorksheet 18 PDFVijay BhaskarPas encore d'évaluation
- Worksheet 16 PDFDocument4 pagesWorksheet 16 PDFVijay Bhaskar33% (6)
- 5054 s11 QP 12Document16 pages5054 s11 QP 12mstudy123456Pas encore d'évaluation
- Sed 122Document136 pagesSed 122palmer okiemutePas encore d'évaluation
- Atom g8Document21 pagesAtom g8florie jane macayaPas encore d'évaluation
- Unhh TitledDocument39 pagesUnhh Titledraza anandPas encore d'évaluation
- Physical Science Quarter 1 Module 2Document32 pagesPhysical Science Quarter 1 Module 2Cyra Gen Cose Hingco100% (4)
- Physical Science - Q3 - LAS2 - THE ATOMIC NUMBER AND THE SYNTHESIS OF NEW ELEMENTS - JOSHUA B UDTUJANDocument17 pagesPhysical Science - Q3 - LAS2 - THE ATOMIC NUMBER AND THE SYNTHESIS OF NEW ELEMENTS - JOSHUA B UDTUJANJoshua UdtujanPas encore d'évaluation
- Modern Physics Target QuestionDocument2 pagesModern Physics Target QuestionAnkur GoyalPas encore d'évaluation
- 11 Gen Chemistry PDFDocument12 pages11 Gen Chemistry PDFDonna Mhae RolloquePas encore d'évaluation
- Week 2 Activities GenChem1 AnswerDocument3 pagesWeek 2 Activities GenChem1 AnswerJujie PascualPas encore d'évaluation
- 18 Quantitative Aspects of Chemical ChangeDocument30 pages18 Quantitative Aspects of Chemical Changeapi-235269401Pas encore d'évaluation
- M. A. Preston (Author) - Structure of The Nucleus-CRC Press (1975)Document716 pagesM. A. Preston (Author) - Structure of The Nucleus-CRC Press (1975)DanielStauffenbergPas encore d'évaluation
- Nickel and Its AlloysDocument162 pagesNickel and Its AlloysAnonymous TI2bUT100% (1)
- GEN CHEMISTRY 1 FIDP (Q1 and Q2)Document11 pagesGEN CHEMISTRY 1 FIDP (Q1 and Q2)Crisanta Ganado100% (1)
- Atomic PhysicsDocument301 pagesAtomic PhysicstomoPas encore d'évaluation
- Roger Muncaster - Nuclear Physics and Fundamental Particles-Stanley Thornes (1995)Document145 pagesRoger Muncaster - Nuclear Physics and Fundamental Particles-Stanley Thornes (1995)dastegirPas encore d'évaluation
- Inside Atom MC Review AnswersDocument2 pagesInside Atom MC Review AnswersBelinda LapsitPas encore d'évaluation
- Nova Hunting The Elements Video GuideDocument3 pagesNova Hunting The Elements Video GuideJaclyn Dugger100% (1)
- Atoms Molecules IonsDocument36 pagesAtoms Molecules IonsGraciella Anne LandichoPas encore d'évaluation
- Module 1 - ChemistryDocument48 pagesModule 1 - ChemistryShapnil FinneyPas encore d'évaluation
- 30A Ch4 Atoms Elements PDFDocument48 pages30A Ch4 Atoms Elements PDFAKo Si NikKoPas encore d'évaluation
- 2004 MRSM Without Answer PDFDocument49 pages2004 MRSM Without Answer PDFanisPas encore d'évaluation
- 2.1 (131 Marks) : MarkschemeDocument48 pages2.1 (131 Marks) : MarkschemeSemwezi EnockPas encore d'évaluation
- Chemistry Notes CompleteDocument136 pagesChemistry Notes CompletePrayrit JainPas encore d'évaluation
- Structure of AtomDocument7 pagesStructure of AtomMera FunportalPas encore d'évaluation
- 1 BhattiAcademy - Com Chemistry 3. Azeem Acadmy (Subjective)Document32 pages1 BhattiAcademy - Com Chemistry 3. Azeem Acadmy (Subjective)hba sportsPas encore d'évaluation
- 1.1 Atomic Structure EXAMPRODocument36 pages1.1 Atomic Structure EXAMPROamandasparkle100% (1)
- Atoms: The Building Blocks of Matter: Chapter 3 ReviewDocument8 pagesAtoms: The Building Blocks of Matter: Chapter 3 ReviewAref Dahabrah0% (1)
- Nanyang JC Atoms, Molecules and Stoichiometry PDFDocument26 pagesNanyang JC Atoms, Molecules and Stoichiometry PDFVarshLokPas encore d'évaluation
- Dwnload Full Biological Science 6th Edition Freeman Solutions Manual PDFDocument36 pagesDwnload Full Biological Science 6th Edition Freeman Solutions Manual PDFcraterglebyllcc100% (17)
- 2 How Atoms Differ 2022Document3 pages2 How Atoms Differ 2022alexandraPas encore d'évaluation