Académique Documents
Professionnel Documents
Culture Documents
401 06 VI Antifungal PDF
Transféré par
Liezel CauilanDescription originale:
Titre original
Copyright
Formats disponibles
Partager ce document
Partager ou intégrer le document
Avez-vous trouvé ce document utile ?
Ce contenu est-il inapproprié ?
Signaler ce documentDroits d'auteur :
Formats disponibles
401 06 VI Antifungal PDF
Transféré par
Liezel CauilanDroits d'auteur :
Formats disponibles
Page 1
VI. Antifungal Agents
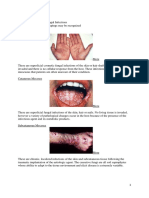
Fungal Infections
Fungal infections are caused by microscopic organisms that can invade the epithelial tissue. The
fungal kingdom includes yeasts, molds, rusts and mushrooms. Fungi, like animals, are
hetrotrophic, that is, they obtain nutrients from the environment, not from endogenous sources
(like plants with photosynthesis). Most fungi are beneficial and are involved in biodegradation,
however, a few can cause opportunistic infections if they are introduced into the skin through
wounds, or into the lungs and nasal passages if inhaled.
Diseases caused by fungi include superficial infections of the skin by dermatophytes in the
Microsporum, Trichophyton or Epidermophyton genera. These dermophytic infections are named
for the site of infection rather than the causative organism.
Dermophytic Infection
Causative Organism
Tinea corporis (ringworm)
Tinea pedis (athletes foot)
Tinea cruis (jock itch)
Tinea capis (scalp)
Tinea barbae (beard/hair)
Tinea unguium (nails)
Microsporum canis, Trichophyton mentagrophytes
T. rubrum, T. mentagrophytes, Epidermophyton floccosum
T. rubrum, T. mentagrophytes, E. floccosum
M. canis T. tonsurans
T. rubrum, T. mentagrophytes
T. rubrum, T. mentagrophytes, E. floccosum
Systemic infections are caused by the inhalation of spores and cause fungal pneumonia. This
pneumonia cannot be transmitted from human to human. These infections can occur in otherwise
healthy individuals. Many of the organisms that cause systemic fungal infections are confined to
specific geographic locations due to favorable climates for their proliferation.
Systemic Infections
Causative Organism
Geographic Location
Coccidioidomycosis
Histoplasmosis
Brazilian Blastomycosis
Blastomycosis
Cocidioides immitis
Histoplasma capsulatum
Paracoccidioides brasiliensis
Blastomyces dermatitidis
Southwestern U.S. and parts of Latin America
Central and Eastern U.S.
South America
Southeastern U.S. and Mississippi River valley
Organisms that cause opportunistic infections will not gain a foothold in healthy individuals, but
in the immunocompromised they can cause serious, sometimes life-threatening infections.
Patients especially susceptible to these infections include individuals with leukemia and other
blood diseases, cancer, HIV and other immunodeficiencies, and diabetes. These organisms can be
found throughout the U.S.
Opportunistic Infections
Causative Organism
Target Organs
Candidaisis, Thrush,
Vulvovaginitis
Cryptococcal meningitis
Candida albicans
Cryptococcus neoformans
GI tract and vagina
Through inhalation, may cause mild lung
infection. Mainly affects CNS
Lung, brain, sinuses and other organs
Sinuses, eyes, blood and brain
Lungs (especially prevalent in HIV patients)
Aspergillosis
Aspergillus sp.
Mucormycosis
Murcor sp.
Pneumocystis carinii pneumonia Pneumocystis carinii
MEDCH 401
Immunizing and Antimicrobial Agents
R.S. Myers
Spring 2006
Page 2
Biochemical Targets for Antifungal Chemotherapy
Fungal cells are complex organisms that share many biochemical targets with other eukaryotic
cells. Therefore, agents that interact with fungal targets not found in eukaryotic cells are needed.
The fungal cell wall is a unique organelle that fulfills the criteria for selective toxicity.
The fungal cell wall differs greatly from the bacterial cell wall and is not affected by antibacterial
cell wall inhibitors such as the -lactams or vancomycin.
Mannoprotein
-Glucan/Chitin
-Glucan
Plasma Membrane
Arrangement of the biomolecular components of the cell wall accounts for the individual identity
of the organism. Although, each organism has a different biochemical composition, their gross
cell wall structure is similar. There are three general mechanisms of action for the antifungal
agents: cell membrane disruption, inhibition of cell division and inhibition of cell wall formation.
Inhibition of Cell Wall Formation
Interference with fungal cell wall biosynthesis has not been as successful and effective as
penicilins and cephalosporins against bacteria. Many chemicals have been discovered that
interfere with various steps in fungal cell wall synthesis with excellent antifungal activity in vitro.
Unfortunately, development of these agents into useful drugs has proven very difficult. Many of
these agents are developed to target -glucan synthesis.
Cell Membrane Disruption
Antifungal agents that disrupt the cell membrane do so by targeting ergosterol, either by binding
to the sterol, forming pores and causing the membrane to become leaky (as with polyene
antifungals), or inhibiting ergosterol biosynthesis (as seen with azole antifungal agents).
Ergosterol is similar to mammalian cholesterol, thus agents binding ergosterol may have a
cytotoxic effect in the host tissue. Ergosterol has two conjugated double bonds that are lacking in
mammalian sterols.
H 3C
CH 3
CH 3
CH 3
CH 3
H
H 3C
CH 3
CH 3
CH 3
H
CH 3
CH 3
HO
HO
Cholesterol
MEDCH 401
Ergosterol
Immunizing and Antimicrobial Agents
R.S. Myers
Spring 2006
Page 3
Inhibition of Cell Division
Nucleoside antifungal agents affect cell division by targeting the microtubule effects in forming
the mitotic spindle:
or by inhibiting DNA transcription:
MEDCH 401
Immunizing and Antimicrobial Agents
R.S. Myers
Spring 2006
Page 4
Polyene Antifungal Agents (Cell Membrane Disruption)
Amphotericin B
PORE
Ergosterol
Phospholipid
Water
HO
H O
O
C
H2
N
HO
OH
O H2
N H
O
C
H
O
Lipid
OH
OH
HO
HO
OH
HO
OH
HO
OH
OH
HO
HO
Lipid
O
H
Water
O
C
H N
H2 O
OH
HO
O
C
N
O H
H 2 OH
Fig. 1: Mechanism of Action of Polyenes
(Amphotericin B as example)
AMPHOTERICIN B
Class of Antifungal:
Polyene
Mechanism of Action: Binds to ergosterol in fungal membrane causing membrane to become
leaky (see Fig. 1)
Indications: Amp. B is indicated for treatment of severe, potentially life threatening fungal
infections. Unfortunately, it must be given IV and is toxic (due to nonselective action on
cholesterol in mammalian cell membranes). Serious fungal infections involve long therapy.
Resistance is due to lower production of membrane sterols or altered sterols, but is relatively rare
at present. Target modification and reduced access to target. Antifungal resistance to date
generally involves emergence of naturally resistant species. No data to suggest that antibiotic
modification in an important antifungal resistance mechanism.
Disposition: Amp. B is not absorbed orally. It is given as a colloidal dispersion by slow IV
infusion. It is highly bound to cholesterol-lipoprotein and has a plasma T1/2 of about 1 day and
1-2 weeks from tissues. It is excreted in urine over a long time. Penetration into the CNS is poor.
However, for fungal infections of the CNS, amphotericin B is mized with cerebrospinal fluid
MEDCH 401
Immunizing and Antimicrobial Agents
R.S. Myers
Spring 2006
Page 5
(CSF) that is obtained from a spinal tap. The solution of amphotericin is then reinjected through
the tap.
Adverse Effects
Headache, fever, chills, anorexia, vomiting, muscle and joint pain.
Pain at site of injection and thrombophlebitis (Drug must never be given intramuscular)
Can give aspirin, meperidine, steroids, antiemetics etc.
Nephrotoxicity - chronic renal tox in up to 80% of patients taking the drug for prolonged periods.
It is reversible but can be irreversible in high doses. Test for kidney function regularly. This is
the most common limiting toxicity of the drug.
Hematologic - hemolytic anemia due to effects on rbc membrane.
Other less common reactions - cardiac, convulsions, neuropathy, hearing loss, allergic, etc.
The use of liposomal preparations will decrease some adverse effects particularly nephrotoxicity.
It is believed the lipid preps decrease nonspecific binding to mammalian membranes.
Products
Fungizone (Bristol Myers Squibb) 50 mg/vial with 41mg of sodium deoxycholate. Reconstitute
with water. Give a test dose and gradually increase dose. Don't exceed 1.5mg/kg/d. Alternate
day therapy is sometimes used. Several months of therapy is usually needed.
Abelcet (Liposome Co.) 1:1 mixture of amphotericin and lipid complex, 100 mg/20 ml.
Rationale for this lipid preparation is that amphotericin B should have a greater affinity for the
lipid vehicle than for cholesterol in cell membranes, thus lower toxicity. Lipid associated
amphotericin B is drawn into the reticuloendothelial system, concentrating in lymphatic tissues,
spleen, liver and lungs where infectious fungi concentrate. Lipases excreted from fungi release
drug from lipid carrier allowing to bind to ergosterol in fungal cell membranes to exert fungistatic
and fungicidal activities.
Aphotec (Sequus Pharmaceuticals) cholesterol colloidal dispersion, 50 or 100 mg/20 ml (not on
UW formulary) Supplied in variety of topical forms including a 3% cream, lotion or ointment
and 100mg/mL oral suspension to treat cutaneous and mucocutaneous mycoses caused by
Candida albicans
AmBiosome (Fujisawa) liposomal, 50mg/vial.
NYSTATIN
Class of Antifungal: Polyene
Mechanism of Action: Binds to ergosterol in fungal membrane causing membrane to become
leaky (see Fig. 1)
MEDCH 401
Immunizing and Antimicrobial Agents
R.S. Myers
Spring 2006
Page 6
Indications: Nystatin was originally isolated from Streptomyces noursei in 1951. A conjugated
tetraene, it was the first clinically useful polyene antifungal antibiotic.
Disposition: Available in oral tablets, powder for suspension, vaginal tablets, pastilles, for local
therapy only (not absorbed). Nystatin will treat gut candidiasis, and is used in a "swish and
swallow" routine for oral candidiasis.
Adverse Effects: No significant adverse effects with these uses, however itching, irritation and
burning may occur. Rarely nystatin can cause diarrhea and nausea Nystatin can be combined with
tetracycline to prevent monilial overgrowth caused by the destruction of bacterial microflora of
the intestine during tetracycline therapy.
Product: Mycostatin and other generic products.
natamycin
NATAMYCIN (Pimaricin, Natacyn)
Class of Antifungal: Polyene
Mechanism of Action: Binds to ergosterol in fungal membrane causing membrane to become
leaky (see Fig. 1)
Indications: Natamycin was first isolated from cultures of Streptomyces natalensis. suspension
intended for the treatment of fungal conjunctivitis, blepharitis and keratitis. Structures consists of
26-membered lactone instead of the 38 for Nystatin and Amphotericin B. The 26-membered
polyenes cause both K leakage and cell lysis at same concentration.
Disposition: Natamycin is supplied as a 5% ophthalmic suspension intended for the treatment of
fungal conjunctivitis, blepharitis and keratitis.
Adverse Effects: Eye irritation, redness and swelling not present prior to use.
Product: Natacyn
MEDCH 401
Immunizing and Antimicrobial Agents
R.S. Myers
Spring 2006
Page 7
Azoles and Triazole Antifungal Agents:
Ergosterol Biosynthesis Inhibitors
Fig. 2: Ergosterol biosynthesis from squalene.
Site of action of azole antifungals (Flu, Itra, Vor) and Terbinafine (Terb) indicated.
Azole antifungal agents are the largest class of synthetic antimycotics. About 20 agents on the
market today. Some used topically to treat superficial dermatophytic and yeast infections. Others
used systemically to treat severe fungal infections. Antifungal activity stems from the presence
of an aromatic five member heterocyclic, either an imidazole or a triazole.
The first members of the class were highly substituted imidazoles (clotrimazole, miconazole)
were not absorbed orally. Ketoconazole introduced in 1984 was the first effective oral therapy for
Candida. Structure activity studies revealed that the imidazole ring can be replaced by a
bioisosteric triazole ring without affecting the antifungal activity but achieving higher selectivity
of the fungal targets vs. host. Itraconazole and Fluconazole are more potent, less toxic and
provide effective oral therapy for many systemic fungal infections. These two are triazoles.
Another triazole has been recently introduced (voriconazole). That said, amphotericin B is
usually the preferred drug for life threatening systemic fungal infections. It is still the gold
standard.
Mechanism of Action: These imidazoles and triazoles inhibit CYP P450 14 - demethylase in
fungi. This enzyme is involved in the conversion of lanosterol to ergosterol. Other P450s in
sterol biosynthesis may be affected. The basic nitrogen of the azole ring forms a tight bond with
MEDCH 401
Immunizing and Antimicrobial Agents
R.S. Myers
Spring 2006
Page 8
the heme iron of the fungal P450 preventing substrate and oxygen binding. Inhibition of the
C14-demethylase results in accumulation of sterols still bearing a C14 methyl group changing
the exact shape and physical properties of the membrane causing permeability changes and
malfunction of membrane imbedded proteins. They have a lower affinity for mammalian P450's.
The effect is fungistatic, but may be fungicidal at higher concentrations.
P450
CH 3
HO
HO
ergosterol
lanosterol
Spectrum and Uses of Azole and Triazole Antifungals
Cl
N
O
N
R
CH
H2C
Miconazole
Cl
Econazole
Cl
Oxiconazole
Cl
Sulconazole
CH2
O
Cl
H 2C
R=
Cl
N
Cl
Cl
O
H2C
S
H2C
O
Tioconazole
H2C
S
Cl
Five azole antifungals, miconazole, econazole, oxiconazole, sulconazole and tioconazole share a
common general structure but differ by the additional structural elements attached to the central
methylene carbon. All five antifungals are used in topical application only.
MICONAZOLE (developed by Janssen Pharmaceutica) is used for skin infections such as tinea
pedis, tinea cruris and vulvovaginitis. It comes in cream, lotion, powder, spray liquid and spray
powder, and also in suppository form for vaginal use. Miconazole is used once or twice a day for
one month for tinea pedis or two weeks for other skin infections. For vaginal infections it is used
once a day at bedtime for three or seven days.
Adverse effects include: increased burning, itching or irritation of the skin or vagina, stomach
pain, fever or foul-smelling vaginal discharge.
Products: Micatin, Monistat-3, Monistat-7, Monistat-Derm, Monistat Dual-Pak
ECONAZOLE (developed by Janssen Pharmaceutica) is a topical cream applied to the skin to
treat fungal infections including: tinea corporis, tinea pedis, tinea cruris, and superficial
candidiasis.
Adverse effects include: Burning, itching, stinging, redness and skin rash.
MEDCH 401
Immunizing and Antimicrobial Agents
R.S. Myers
Spring 2006
Page 9
Products: Spectrazole, Ecostatin
OXICONAZOLE (developed by F. Hoffmann-LaRoche and Siegfried AG) is a cream or lotion
applied to the skin in the treatment of tinea corporis, tinea pedis and tinea cruris.
Adverse effects include: Burning, itching, blistering, crusting, dryness or flaking of the skin,
scaling, severe redness, soreness, swelling and pain in hairy areas with pus at the root of hair.
Products: Oxistat, Oxizole
SULCONAZOLE (developed by Syntex Research) is a topical cream or solution to treat tinea
corporis, tinea pedis and tinea cruris.
Adverse effects include: Burning, stinging, itching and redness of the skin.
Products: Exelderm
TIOCONAZOLE (developed by Pfizer U.K.) is a cream to treat tinea corporis, tinea pedis, tinea
cruris and cutaneous candidiasis.
Adverse effects include: Burning, itching, redness, skin rash and swelling.
Products: Trosyd AF, Trosyd J
N
O
O
N
H 3C
O
Cl
Ketoconazole
Cl
KETOCONAZOLE is supplied as a cream or in shampoos at one- or two-percent, for the
treatment of tinea pedis, tinea corporis, tinea cruris and cutaneous candidiasis.
Adverse effects include: itching, stinging, skin rash, dry skin, and dry or oily scalp.
Products: Nizoral Cream, Nizoral A-D Shampoo (1%), Nizoral Shampoo (2%)
N
N
N
O
O
Cl
Cl
N
N
Itraconazole
ITRACONAZOLE is taken orally in capsule form to treat fungal infections that start in the
lungs and spread throughout the body. Itraconazole can also be used to treat fungal infections of
the nails, although it is important to point out that treatment of nail fungal infections does not
result in healthier looking nails. Normal nail appearance will occur only with new growth, which
can take up to six months for full nail growth. Oral solutions of this antifungal agent can be used
to treat oral candidiasis.
MEDCH 401
Immunizing and Antimicrobial Agents
R.S. Myers
Spring 2006
Page 10
Drug Interactions: Patients on proton pump inhibitors should take itraconazole with a cola soft
drink to aid in bioavailability.
Adverse effects include: diarrhea, constipation, gas, stomach pain, heartburn, sore or bleeding
gums, sores in and around the mouth, headache, dizziness, sweating, muscle pain, decreased
sexual desire or ability, nervousness, depression and runny nose. More severe side effects can
include: excessive tiredness, loss of appetite, upset stomach, vomiting, tingling or numbness in
the extremities, fever, chills, rash, hives and difficulty breathing or swallowing.
HEPATOXICITY: yellowing of the eyes or skin, dark urine or pale stools.
Product: Sporanox
N
N
N
F
N
HO
F
Fluconazole
FLUCONAZOLE is a one-a-day tablet or suspension to treat yeast infections of the vagina,
mouth, throat, esophagus, abdomen, lungs, blood and other organs. Fluconazole is also used to
treat meningitis and can prevent yeast infections in patients who are likely to become infected due
to chemotherapy or radiation therapy before a bone marrow transplant.
Adverse effects include: headache, dizziness, diarrhea, stomach pain, heartburn and changes in
the ability to taste food. More severe side effects can include: excessive tiredness, loss of appetite,
upset stomach, vomiting, tingling or numbness in the extremities, fever, chills, rash, hives and
difficulty breathing or swallowing. HEPATOXICITY: yellowing of the eyes or skin, dark urine
or pale stools.
Product: Diflucan
N
N
N
N HO
Voriconazole F
F
VORICONAZOLE is formulated in an oral suspension, tablets or parenteral injection. It is used
to treat different kinds of serious fungal infections and may be used in patients who have not
responded to other antifungal agents.
Drug interactions: Xanax, Versed, Halcion, Agenernase, Viracept, Invirase, Hismanal,
barbiturates, cyclosporine, ergot alkaloids, HMG CoA reductase inhibitors and warfarin.
Adverse effects include: rash, bloating or swelling of face, arms, hands, lower legs or feet,
stomach pain, blurred vision, chills, convulsions, dizziness, dry mouth, headache and muscle
pain. HEPATOXICITY: dark urine or pale stools.
Product: VFEND
MEDCH 401
Immunizing and Antimicrobial Agents
R.S. Myers
Spring 2006
Page 11
Cl
Cl
S
Cl
Butoconazole
BUTOCONAZOLE is a cream suppository used to treat vulvovaginitis. It is used either once or
in a seven-day regimen at bedtime.
Adverse effects include: burning or irritation in the vagina when cream is inserted, stomach pain,
fever or foul-smelling vaginal discharge.
Product: Gynazole-1
N
Cl
Cl
O
Terconazole
O
N
N
TERCONAZOLE is supplied as a cream or suppository to treat vulvovaginitis. It is usually used
daily at bedtime for either three or seven days.
Adverse effects include: headache, missed menstrual periods, burning or irritation in vagina when
cream or suppository is inserted, stomach pain, fever, or foul-smelling vaginal discharge.
Product: Terazol 3, Terazol 7.
N
N
O
N
OH
O
N
O
N
N
Posaconazole
F
POSACONAZOLE (Schering-Plough) is a novel triazole in Phase II clinical trials to be used as
an oral suspension to treat invasive fungal infections caused by Candida and aspergillus. The
current clinical trial will conclude in October 2006.
MEDCH 401
Immunizing and Antimicrobial Agents
R.S. Myers
Spring 2006
Page 12
Antifungal Agents Targeting Squalene Epoxidase:
Ergosterol Biosynthesis Inhibitors
The allylamines have a more limited spectrum of activity than the azoles and triazoles and are
only effective against dermatophytes. They are employed in the treatment of fungal infections of
the skin and nails.
Mechanism of Action: These antifungal agents are reversible, noncompetitive inhibitors of the
first step in ergosterol biosynthesis (see fig. 2), the conversion of squalene to squalene-2,3epoxide by squalene epoxidase. The buildup of squalene in the cell membrane is toxic to the cell,
causing pH imbalances and malfunction of membrane bound proteins.
Terbinafine
TERBINAFINE comes as a tablet to take orally or as a topical cream It is used to treat fungal
infections of the nails.
Drug interactions: warfarin, antidepressant drugs, beta-blockers, proton pump inhibitors and
drugs to suppress the immune system.
Adverse effects include: headache, dizziness, diarrhea, stomach pain, heartburn and changes in
the ability to taste food. More severe side effects can include: excessive tiredness, loss of appetite,
upset stomach, vomiting, tingling or numbness in the extremities, fever, chills, rash, hives and
difficulty breathing or swallowing. HEPATOXICITY: yellowing of the eyes or skin, dark urine
or pale stools.
Product: Lamisil,
S
O
Tolnatate
TOLNAFTATE is is a topical cream to treat tinea infections of the skin.
Mechanism of Action: The exact mechanism unknown; however, it has been reported to distort
the hyphae and to stunt mycelial growth in susceptible organisms. Inhibition of squalene
epoxidation has also been reported.
Adverse effects are rare. Skin irritation has been reported.
Products:Aftate, Tinactin, Ting, Breezee
MEDCH 401
Immunizing and Antimicrobial Agents
R.S. Myers
Spring 2006
Page 13
Other Antifungals Affecting Cell Membrane Stability
OO
Ciclopirox
CICLOPIROX is a topical solution used to treat fungal infections of the nails and hair.It is a
broad-spectrum antifungal medication that also has antibacterial and anti-inflammatory
properties.
Mechanism of Action: Its main mode of action is thought to be its high affinity for trivalent
cations, which inhibit essential co-factors in enzymes. Ciclopirox exhibits either fungistatic or
fungicidal activity in vitro against a broad spectrum of fungal organisms, such as dermatophytes,
yeasts, dimorphic fungi, eumycetes, and actinomycetes. In addition to its broad spectrum of
action, ciclopirox also exerts antibacterial activity against many Gram-positive and Gramnegative bacteria. Furthermore, the anti-inflammatory effects of ciclopirox have been
demonstrated in human polymorphonuclear cells, where ciclopirox has inhibited the synthesis of
prostaglandin and leukotriene. Ciclopirox can also exhibit its anti-inflammatory effects by
inhibiting the formation of 5-lipoxygenase and cyclooxygenase.
Ciclopirox is thought to act through the chelation of polyvalent metal cations, such as Fe3+ and
Al3+. These cations inhibit many enzymes, including cytochromes, possibly disrupting the
biosynthesis of ergosterol. Ciclopirox also appears to modify the plasma membrane of fungi,
resulting in the disorganization of internal structures.
Adverse effects: redness, irritation, burning, blistering or swelling at the site of application and
discoloration of the nails or surrounding area. Treated nails may become ingrown.
Product: Loprox, Penlac nail lacquer
Inhibitors of Cell Division
NH2
OH
F
N
O
N
H
Cytosine
Deaminase
N
H
FLUCYTOSINE was synthesized in 1957 as an antitumor agent. It was inactive but it was
found to have antifungal activity.
Mechanism of Action: The drug enters the fungal cell through active transport on ATPases that
normally transport pyrimidines. Once inside cells, fungal cytosine deaminase convert the drug to
active 5-fluorouracil (5FU, a very effective antitumor agent, by the way!) which is incorporated
into RNA causing faulty RNA synthesis and also is a strong, non competitive inhibitor of
thymidylate synthesis interrupting the one carbon pool substrate. Mammalian cells do not contain
cytosine deaminase.
Resistance develops rapidly and occurs on many levels e.g. transport into the cell and cytosine
deaminase steps. After a few dosing intervals the drug is essentially useless. To avoid rapid
resistance, combination with Amphotericin B, and the combination is synergistic. It is also
synergistic with itraconazole and fluconazole, and interest in these combinations for treatment of
MEDCH 401
Immunizing and Antimicrobial Agents
R.S. Myers
Spring 2006
Page 14
systemic Candida infections is increasing. Amphotericin B damaged membranes are thought to
allow better entry of flucytosine. Used (with Amp. B) for Cryptococcal meningitis, systemic
Candida infections, and some other systemic fungal infections. This is an oral drug. Good also
for fungal UTI.
Disposition: The drug is well absorbed and well distributed. CSF levels are 65-90% of plasma
levels. Is eliminated unchanged largely in the urine.
Adverse Effects: GI upset, hepatic involvement seen in the increase in transaminases,
Hematologic involvement include anemia, leucopenia. Thrombocytopenia is the major
complication of therapy and may be due to low levels of 5-FU circulating.
Product: Ancobon (Roche)
O
O O
O
O
O
Cl
Griseofulvin
GRISEOFULVIN is an antifungal produced from Penicillium griseofulvin. Therapy must
continue until new tissue replaces old diseased tissue. When given orally, plasma-borne
griseofulvin becomes incorporated into keratin precursor cells and ultimately into
kearatin that cannot support fungal growth.
Mechanism of Action: Griseofulvin inhibits microtubule polymerization thus inhibiting the
formation of the mitotic spindle.
Adverse effects: Headache is a common adverse effect. May cause aplastic anemia. Being
gradually replaced by newer agents.
Products: Fulvicin-U/F, Grifulvin V, Gris-PEG
Cell Wall Inhibitors
H 2N
NH
HO
H 2N
OH
O
O
H
N
N
H
OH
O HN
HO
O
HO
NH O
H
N
O
H 3C
CH 3 CH 3
CH 3
N
OH
OH
HO
CASPOFUNGIN acetate is an parenteral injection used in the treatment of invasive aspergillosis
in patients refractory to or intolerant of other antifungal therapies. Studies have shown
caspofungin to be effective against invasive candidaisis. It is a semisynthetic lipopeptide
(echinocandin) derived from a fermentation product of Glarea lozoyensis.
MEDCH 401
Immunizing and Antimicrobial Agents
R.S. Myers
Spring 2006
Page 15
Mechanism of Action: Caspofungin is a (1,3)-D-glucan synthesis inhibitor, thus disrupting
the formation of -glucan in the cell walls. -glucan is essential to the structural integrity
of the cell wall.
Drug interactions: Reduces AUC, Cmax and concentration of tacrolimus. Cyclosporine increases
AUC of caspofungin.
Adverse effects: thrombophlebitis, vein irritation, histamine-related symptoms, anaphylaxis has
been reported.
Product: Cancidas
ANIDULAFUNGIN (Pfizer) has recently been approved to treat infections by Candida.
Mechanism of Action: It inhibits glucan synthase, disrupting the formation of b-glucan. It is
especially effective against fluconazole-resistant Candida.
Adverse Effects: Diarrhea, elevation of liver enzymes.
Product: Eraxis
O
Undecylenic Acid
OH
UNDECYLENIC ACID is widely used topically as the zinc salt in OTC preparations for topical
treatment of infections by dermatophytes.
Mechanism of Action: This organic acid will interact non-specifically with components in the cell
membrane. It can be used in concentrations up to 10% in solution, powder and emulsions.
Traditionally used for athletes foot (tinea pedis) although cure rates are low.
Adverse effects are rare. Skin irritation has been reported.
Products: Desenex, Cruex, Decylenes Powder, Caldesene, Gordochom Solution
MEDCH 401
Immunizing and Antimicrobial Agents
R.S. Myers
Spring 2006
Page 16
Other Inhibitors of Fungal Cell Wall Synthesis: Under Development
Mannoprotein
-Glucan/Chitin
-Glucan
Plasma Membrane
Other -1,3 glucan synthetase inhibitors: Papulacandins glycolipid antifungal produced by
Papularia sp. Loss of b-glucan results in weakening of the cell wall, thus internal pressures can
cause the cells to lyse
Chitin Synthase inhibitors: Polyoxins and Nikkomycins nucleoside peptides. Chitin is also a
major component to the cell wall and its loss will also weaken the cell walls.
C Mannan binding antifungals: Pradimicins and benanomicins. C-mannan is a cell wall
glycoprotein that combats the host immune response through disruption of cytokine response and
possibly T-lymphocyte action.
MEDCH 401
Immunizing and Antimicrobial Agents
R.S. Myers
Spring 2006
Vous aimerez peut-être aussi
- 2a Embryonic DevelopmentDocument16 pages2a Embryonic DevelopmentLiezel CauilanPas encore d'évaluation
- CHEDROcc PDFDocument21 pagesCHEDROcc PDFLiezel CauilanPas encore d'évaluation
- Chapter 3: Disorders of Water and Electrolytes MetabolismDocument57 pagesChapter 3: Disorders of Water and Electrolytes MetabolismLiezel CauilanPas encore d'évaluation
- 2 Myths and RealitiesDocument3 pages2 Myths and RealitiesLiezel CauilanPas encore d'évaluation
- Unit 1 Chapter 3 Worksheet PDFDocument4 pagesUnit 1 Chapter 3 Worksheet PDFLiezel CauilanPas encore d'évaluation
- Unit 1 Chapter 3 Worksheet PDFDocument4 pagesUnit 1 Chapter 3 Worksheet PDFLiezel CauilanPas encore d'évaluation
- 9-Reproductive HealthDocument32 pages9-Reproductive HealthLiezel CauilanPas encore d'évaluation
- 6 Sudden EmergenciesDocument14 pages6 Sudden EmergenciesLiezel CauilanPas encore d'évaluation
- Basic CPR Lecture (Standard 1)Document3 pagesBasic CPR Lecture (Standard 1)Liezel CauilanPas encore d'évaluation
- Barchartsquickstudymicrobiology 131224153256 Phpapp02 PDFDocument4 pagesBarchartsquickstudymicrobiology 131224153256 Phpapp02 PDFLiezel CauilanPas encore d'évaluation
- 6 Sudden EmergenciesDocument14 pages6 Sudden EmergenciesLiezel CauilanPas encore d'évaluation
- Chapter 5 Outline For Notes PDFDocument2 pagesChapter 5 Outline For Notes PDFLiezel CauilanPas encore d'évaluation
- Principles of Drug AdministrationDocument8 pagesPrinciples of Drug Administrationapi-373991090% (21)
- Communicable Dse ReviewerDocument13 pagesCommunicable Dse ReviewerRichard Ines Valino100% (29)
- Speech Sound Development PDFDocument7 pagesSpeech Sound Development PDFLiezel CauilanPas encore d'évaluation
- Sensory Development Checklist PDFDocument7 pagesSensory Development Checklist PDFLiezel CauilanPas encore d'évaluation
- Stages of Language Developmen1 PDFDocument10 pagesStages of Language Developmen1 PDFLiezel CauilanPas encore d'évaluation
- Syllabus ECCDocument3 pagesSyllabus ECCLiezel CauilanPas encore d'évaluation
- Stages of Language Developmen1 PDFDocument10 pagesStages of Language Developmen1 PDFLiezel CauilanPas encore d'évaluation
- Speech Sound Development Checklist PDFDocument4 pagesSpeech Sound Development Checklist PDFLiezel CauilanPas encore d'évaluation
- Play and Social Development Checklist PDFDocument4 pagesPlay and Social Development Checklist PDFLiezel Cauilan100% (1)
- Sensory Development PDFDocument3 pagesSensory Development PDFLiezel CauilanPas encore d'évaluation
- Self Care and Organisation Development Checklist PDFDocument5 pagesSelf Care and Organisation Development Checklist PDFLiezel CauilanPas encore d'évaluation
- phh04 PDFDocument3 pagesphh04 PDFLiezel CauilanPas encore d'évaluation
- Self Care and Organisation Development PDFDocument5 pagesSelf Care and Organisation Development PDFLiezel CauilanPas encore d'évaluation
- Phonological Awareness Checklist PDFDocument3 pagesPhonological Awareness Checklist PDFLiezel CauilanPas encore d'évaluation
- Phonological Awareness Development PDFDocument2 pagesPhonological Awareness Development PDFLiezel CauilanPas encore d'évaluation
- 6th Central Pay Commission Salary CalculatorDocument15 pages6th Central Pay Commission Salary Calculatorrakhonde100% (436)
- NMAT ID Form-1031507414 PDFDocument1 pageNMAT ID Form-1031507414 PDFLiezel CauilanPas encore d'évaluation
- The Subtle Art of Not Giving a F*ck: A Counterintuitive Approach to Living a Good LifeD'EverandThe Subtle Art of Not Giving a F*ck: A Counterintuitive Approach to Living a Good LifeÉvaluation : 4 sur 5 étoiles4/5 (5794)
- The Little Book of Hygge: Danish Secrets to Happy LivingD'EverandThe Little Book of Hygge: Danish Secrets to Happy LivingÉvaluation : 3.5 sur 5 étoiles3.5/5 (400)
- Shoe Dog: A Memoir by the Creator of NikeD'EverandShoe Dog: A Memoir by the Creator of NikeÉvaluation : 4.5 sur 5 étoiles4.5/5 (537)
- Hidden Figures: The American Dream and the Untold Story of the Black Women Mathematicians Who Helped Win the Space RaceD'EverandHidden Figures: The American Dream and the Untold Story of the Black Women Mathematicians Who Helped Win the Space RaceÉvaluation : 4 sur 5 étoiles4/5 (895)
- The Yellow House: A Memoir (2019 National Book Award Winner)D'EverandThe Yellow House: A Memoir (2019 National Book Award Winner)Évaluation : 4 sur 5 étoiles4/5 (98)
- The Emperor of All Maladies: A Biography of CancerD'EverandThe Emperor of All Maladies: A Biography of CancerÉvaluation : 4.5 sur 5 étoiles4.5/5 (271)
- A Heartbreaking Work Of Staggering Genius: A Memoir Based on a True StoryD'EverandA Heartbreaking Work Of Staggering Genius: A Memoir Based on a True StoryÉvaluation : 3.5 sur 5 étoiles3.5/5 (231)
- Never Split the Difference: Negotiating As If Your Life Depended On ItD'EverandNever Split the Difference: Negotiating As If Your Life Depended On ItÉvaluation : 4.5 sur 5 étoiles4.5/5 (838)
- Grit: The Power of Passion and PerseveranceD'EverandGrit: The Power of Passion and PerseveranceÉvaluation : 4 sur 5 étoiles4/5 (588)
- On Fire: The (Burning) Case for a Green New DealD'EverandOn Fire: The (Burning) Case for a Green New DealÉvaluation : 4 sur 5 étoiles4/5 (74)
- Elon Musk: Tesla, SpaceX, and the Quest for a Fantastic FutureD'EverandElon Musk: Tesla, SpaceX, and the Quest for a Fantastic FutureÉvaluation : 4.5 sur 5 étoiles4.5/5 (474)
- Devil in the Grove: Thurgood Marshall, the Groveland Boys, and the Dawn of a New AmericaD'EverandDevil in the Grove: Thurgood Marshall, the Groveland Boys, and the Dawn of a New AmericaÉvaluation : 4.5 sur 5 étoiles4.5/5 (266)
- The Unwinding: An Inner History of the New AmericaD'EverandThe Unwinding: An Inner History of the New AmericaÉvaluation : 4 sur 5 étoiles4/5 (45)
- Team of Rivals: The Political Genius of Abraham LincolnD'EverandTeam of Rivals: The Political Genius of Abraham LincolnÉvaluation : 4.5 sur 5 étoiles4.5/5 (234)
- The World Is Flat 3.0: A Brief History of the Twenty-first CenturyD'EverandThe World Is Flat 3.0: A Brief History of the Twenty-first CenturyÉvaluation : 3.5 sur 5 étoiles3.5/5 (2259)
- The Gifts of Imperfection: Let Go of Who You Think You're Supposed to Be and Embrace Who You AreD'EverandThe Gifts of Imperfection: Let Go of Who You Think You're Supposed to Be and Embrace Who You AreÉvaluation : 4 sur 5 étoiles4/5 (1090)
- The Hard Thing About Hard Things: Building a Business When There Are No Easy AnswersD'EverandThe Hard Thing About Hard Things: Building a Business When There Are No Easy AnswersÉvaluation : 4.5 sur 5 étoiles4.5/5 (344)
- The Sympathizer: A Novel (Pulitzer Prize for Fiction)D'EverandThe Sympathizer: A Novel (Pulitzer Prize for Fiction)Évaluation : 4.5 sur 5 étoiles4.5/5 (121)
- Her Body and Other Parties: StoriesD'EverandHer Body and Other Parties: StoriesÉvaluation : 4 sur 5 étoiles4/5 (821)
- Mikosis AdelaideDocument69 pagesMikosis AdelaideADIPas encore d'évaluation
- Superficial and Cutaneous Mycoses: 2. Disease CharacteristicsDocument4 pagesSuperficial and Cutaneous Mycoses: 2. Disease CharacteristicsMA. ANGELI DELA CRUZPas encore d'évaluation
- 4 DermatomycosesDocument96 pages4 DermatomycosesShany SchwarzwaldPas encore d'évaluation
- Mycology 2019Document8 pagesMycology 2019Angellie100% (1)
- 4th Shifting Micro Lab ReviewerDocument154 pages4th Shifting Micro Lab ReviewerJade MonrealPas encore d'évaluation
- Superficial CutaneousDocument107 pagesSuperficial CutaneouskarenjoycePas encore d'évaluation
- Dermatomycosis 18th July 2021Document120 pagesDermatomycosis 18th July 2021Sekar OktaviaPas encore d'évaluation
- Review Notes in Diagnostic Mycology Virology CEFI PDFDocument13 pagesReview Notes in Diagnostic Mycology Virology CEFI PDFZhiddah Japzon0% (1)
- Micoses Superficiais - GlobalDocument19 pagesMicoses Superficiais - GlobalJosé Paulo Ribeiro JúniorPas encore d'évaluation
- Fungi of Medical ImportanceDocument11 pagesFungi of Medical ImportanceRi FitrianaPas encore d'évaluation
- #1 &2 Myco & Viro + Definition of TermsDocument5 pages#1 &2 Myco & Viro + Definition of TermsJared SicorPas encore d'évaluation
- Mycology Manual: Mohammed Saleem Ali Shtayeh Rana M. JamousDocument87 pagesMycology Manual: Mohammed Saleem Ali Shtayeh Rana M. JamousAlberto Carlos ContrerasPas encore d'évaluation
- Efficacy and Safety of Terbinafine Hydrochloride 1 PDFDocument4 pagesEfficacy and Safety of Terbinafine Hydrochloride 1 PDFAlexandria Firdaus Al-farisyPas encore d'évaluation
- Quiz IntegumentaryDocument44 pagesQuiz IntegumentaryMedShare100% (5)
- Microscopic Exam of FungiDocument13 pagesMicroscopic Exam of FungijadeyjadePas encore d'évaluation
- Dermatophytes: (Superficial Mycoses)Document41 pagesDermatophytes: (Superficial Mycoses)safemindPas encore d'évaluation
- Atlas de MicologiaDocument851 pagesAtlas de MicologiaPick D Dragón100% (1)
- Microbiology Prax ReviewerDocument4 pagesMicrobiology Prax ReviewerChristine NazarenoPas encore d'évaluation
- Dermatovenerology CS 253 PDFDocument36 pagesDermatovenerology CS 253 PDFIvanes IgorPas encore d'évaluation
- Ciulla - MycoDocument13 pagesCiulla - MycoSalve Rachelle BillenaPas encore d'évaluation
- Muhilan Jegadeesan's Clinico-Etiological Study of Tinea Corporis: Emergence of Trichophyton MentagrophytesDocument5 pagesMuhilan Jegadeesan's Clinico-Etiological Study of Tinea Corporis: Emergence of Trichophyton MentagrophytesjmuhilanPas encore d'évaluation
- Rationale in Microbiology and Parasitology: Prepared By: Ralf LadreraDocument4 pagesRationale in Microbiology and Parasitology: Prepared By: Ralf LadreraralfPas encore d'évaluation
- Kti InternasionalDocument188 pagesKti Internasionalckcy ahaiPas encore d'évaluation
- Luliconazole Topical Cream MonographDocument11 pagesLuliconazole Topical Cream Monographdaizhussain004Pas encore d'évaluation
- Cutaneous Mycoses. Case StudyDocument49 pagesCutaneous Mycoses. Case Studysaii100% (3)
- Eukaryotes and MicroorganismsDocument10 pagesEukaryotes and MicroorganismsRIA MAEVE NGOPas encore d'évaluation
- Mycology McqsDocument6 pagesMycology McqsmryamPas encore d'évaluation
- Classification of Fungal DiseasesDocument8 pagesClassification of Fungal DiseasesLlermi LiborioPas encore d'évaluation
- MycologyyyyyyyyyyyyyyDocument12 pagesMycologyyyyyyyyyyyyyyNyxa AbdullaPas encore d'évaluation
- Classification of Fungal InfectionsDocument26 pagesClassification of Fungal Infectionstev26Pas encore d'évaluation