Académique Documents
Professionnel Documents
Culture Documents
Anti-HIV Therapeutic Research - Institute of Human Virology
Transféré par
Man LorTitre original
Copyright
Formats disponibles
Partager ce document
Partager ou intégrer le document
Avez-vous trouvé ce document utile ?
Ce contenu est-il inapproprié ?
Signaler ce documentDroits d'auteur :
Formats disponibles
Anti-HIV Therapeutic Research - Institute of Human Virology
Transféré par
Man LorDroits d'auteur :
Formats disponibles
Anti-HIV Therapeutic Research: Institute of Human Virology
CLINCAL CARE AND
RESEARCH
Division Overview
Site
Directory
Faculty
Search the Site
IHV > Research:Anti-HIV Therapeutic Research
ANTI-HIV THERAPEUTIC RESEARCH
Clinical Trials
Associated Faculty
Anti-HIV Therapeutic Research
Lab Head
Assistant Professor Olga Latinovic, PhD
Clinics
Assistant Professor Alonso Heredia, PhD
Social Entrepreneurship (PDF)
JACQUES Initiative
Focus of our work is the CCR5 chemokine receptor that plays a crucial
role in HIV-1 infection and as such offers an important potential
therapeutic target [Fig.1]. We have established methods to quantify
CCR5 density and to evaluate its impact on virus infectivity in
spreading/single cycle infection and direct virus-cell fusion assays.
Using low doses of the drug Rapamycin, a CCR5 suppressor, we
demonstrate decreased R5 HIV-1 infectivity and enhanced potency of
entry inhibitors. Our data show that Rapamycin reduction of CCR5
density restores sensitivity of drug-resistant R5 HIV-1 to fusion inhibitor
Enfuvirtide,T-20, (Antimicrobial Reagents and Chemotherapy, 2007) and
to the new generation of entry inhibitors-CCR5 antagonists (Antiviral
Research, 2009; Clinical Medicine: Therapeutics, 2009).
Robert Redfield, MD
We found that Rapamycin induced reduction of CCR5 density in
lymphocytes increased sensitivity to Vicriviroc (VCV) in VCV-resistant strains, inhibiting production by ~ 90% (PNAS,
2008). Novel Beta-lactamase (BlaM) entry assay revealed the differences in the activity between CCR5 antagonist
sensitive and CCR5 antagonist resistant virus. In the case of CCR5 antagonist sensitive virus, we observed complete
inhibition in cell lines with high and physiological CCR5 levels. In the case of the resistant virus, there is no inhibition in
higher CCR5 expressive cells, but in cells with physiological expression of CCR5, we do observe susceptibility to
CCR5 antagonist resistant virus.
As an alternative anti-viral therapy approach, we currently employ the CCR5 antibodies in order to determine the affinity
of resistant virus Envelope in cases when it is free versus occupied CCR5 site by the CCR5 antagonists. Looking for
the mechanism of resistant to CCR5 antagonists viruses [Fig. 2], we found out that interaction between the antagonists
occupied CCR5 and the CCR5 antibody is more potent than inhibition provided by CCR5 antibody per se. HIV-1 strains
resistant to the only clinically approved CCR5 antagonist Maraviroc (MVC, Fig. 3) generally remain CCR5 tropic, but
gain the ability to use drug-bound CCR5. We demonstrate that MVCresistant HIV-1 loses the ability to use MVC-bound
CCR5 at low surface CCR5 densities, suggesting a lower affinity for antagonist-bound CCR5 compared to free CCR5.
In accordance, antibodies directed against the second extracellular loop (ECL2) of CCR5 had greater antiviral activity
in MVC-bound than in MVC-free CCR5 infection of cell lines. However, in primary peripheral blood lymphocytes
(PBLs), a dichotomy in antibody efficacy became evident. ECL2 CCR5 mAbs HGS004 and HGS101, inhibited PBL
infection by MVC resistant HIV-1 more potently with MVC-bound than with free CCR5. In addition, HGS004 and
HGS101, but not the other mAbs, restored the antiviral activity of MVC against resistant virus in PBLs. Both, HGS004
and HGS101, currently in clinical development, could help overcome MVC Resistance (Virology, 2011).
In addition, we demonstrate that the CCR5 antibody HGS004 and the CCR5 antagonist Maraviroc have potent antiviral
synergy against R5 HIV-1, translating into dose reductions of >10-fold for Maraviroc and >150-fold for HGS004 (AIDS,
2011). These data, together with the high barrier of resistance to HGS004, suggest that combinations of Maraviroc and
HGS004 could provide effective preventive and therapeutic strategies against R5 HIV-1.
Figure 1
Therapeutic opportunities for inhibition of HIV-1 entry
http://www.ihv.org/research/anti_hiv.html[28/10/2557 3:36:38]
Anti-HIV Therapeutic Research: Institute of Human Virology
HIV-1 entry is mediated by the viral Env protein, which comprises the glycoproteins gp120 and gp41 arranged in
trimeric spikes on the viral surface. Entry encompasses three steps: CD4 binding, coreceptor binding and fusion. The
viral gp120 first binds to CD4, causing a repositioning of the variable loops V1/V2 and V3 and thereby exposing the
bridging sheet and forming a coreceptor binding site. Upon coreceptor binding, conformational changes in gp120 and
gp41 lead to the insertion of gp41 fusion peptide into the cell membrane. Subsequent conformational changes result in
the formation of a six-helix bundle, with the HR2 domains folding back and packing into grooves on the outside of the
triple-stranded HR1 domains. This brings the fusion peptide and transmembrane region of gp41 in close proximity,
forming a fusion pore that allows transfer of the viral core into the cell. Each step on HIV-1 entry can be targeted by
inhibitors currently approved or in clinical development. CD4 binding is targeted by ibalizumab (formerly TNX-355), a
humanized monoclonal antibody that binds to CD4. Coreceptor binding is blocked by small-molecule CCR5 antagonists
(Maraviroc and Vicriviroc) and by CCR5 antibodies (PRO-140 and HGS-004). Finally, the formation of a six-helix
bundle, and thereby fusion, is prevented by enfuvirtide. Figure 1. adapted from Melikyan GB, Retrovirology, 2008.
Figure 2
Model for Maraviroc mechanism of resistance
Maraviroc binds to the transmembrane region of CCR5, thereby inducing confomational changes that cannot be
recognized by R5 HIV-1 gp120. One mechanism of resistance involves changes in HIV-1 Env that permit recognition of
Maraviroc-bound CCR5. As such, resistant viruses are not blocked by increasing Maraviroc doses. Figure 2. adapted
from M. Westby, Curr Opin HIV AIDS, 2007.
http://www.ihv.org/research/anti_hiv.html[28/10/2557 3:36:38]
Anti-HIV Therapeutic Research: Institute of Human Virology
Figure 3
Model for maraviroc mechanism of action
Binding of HIV-1 gp120 to CD4 exposes the bridging sheet and creates a coreceptor binding site. In the absence of
Maraviroc, the bridging sheet and the base of V3 interact with the N-terminus of CCR5, while more distal regions of V3
interact with extracellular loops (mainly ECL2). Binding of Maraviroc to the transmembrane region of CCR5 locks CCR5
in a conformation that does not recognize the distal regions of V3. Figure 3. adapted from Soriano V. et al, AIDS, 2008.
Hotline
The Institute of Human Virology, 725 West Lombard Street, Baltimore MD 21201
http://www.ihv.org/research/anti_hiv.html[28/10/2557 3:36:38]
Vous aimerez peut-être aussi
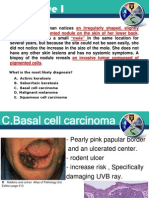
- Benign Skin TumoursDocument7 pagesBenign Skin TumoursMan LorPas encore d'évaluation
- Calendar September 2016 LandscapeDocument1 pageCalendar September 2016 LandscapeMan LorPas encore d'évaluation
- Bowen's Disease: Understanding The SkinDocument6 pagesBowen's Disease: Understanding The SkinMan LorPas encore d'évaluation
- Calendar July 2016 LandscapeDocument1 pageCalendar July 2016 LandscapeMan LorPas encore d'évaluation
- Frizzell - Handbook of PathophysiologyDocument973 pagesFrizzell - Handbook of PathophysiologyididgamaPas encore d'évaluation
- LCE-24C100F (K) LCE-24C100F (N) LCE-24C100F (R) LCE-24C100F (S) : Instruction ManualDocument20 pagesLCE-24C100F (K) LCE-24C100F (N) LCE-24C100F (R) LCE-24C100F (S) : Instruction ManualMan LorPas encore d'évaluation
- History: A 65-Year-Old Man Presented To TheDocument3 pagesHistory: A 65-Year-Old Man Presented To TheMan LorPas encore d'évaluation
- Case Hematology + Genetics I: Β Thalassemia/Hbe DiseaseDocument1 pageCase Hematology + Genetics I: Β Thalassemia/Hbe DiseaseMan LorPas encore d'évaluation
- Skin-All For FriendDocument10 pagesSkin-All For FriendMan LorPas encore d'évaluation
- Skin CutDocument9 pagesSkin CutMan LorPas encore d'évaluation
- Grit: The Power of Passion and PerseveranceD'EverandGrit: The Power of Passion and PerseveranceÉvaluation : 4 sur 5 étoiles4/5 (588)
- The Yellow House: A Memoir (2019 National Book Award Winner)D'EverandThe Yellow House: A Memoir (2019 National Book Award Winner)Évaluation : 4 sur 5 étoiles4/5 (98)
- The Subtle Art of Not Giving a F*ck: A Counterintuitive Approach to Living a Good LifeD'EverandThe Subtle Art of Not Giving a F*ck: A Counterintuitive Approach to Living a Good LifeÉvaluation : 4 sur 5 étoiles4/5 (5795)
- Never Split the Difference: Negotiating As If Your Life Depended On ItD'EverandNever Split the Difference: Negotiating As If Your Life Depended On ItÉvaluation : 4.5 sur 5 étoiles4.5/5 (838)
- Hidden Figures: The American Dream and the Untold Story of the Black Women Mathematicians Who Helped Win the Space RaceD'EverandHidden Figures: The American Dream and the Untold Story of the Black Women Mathematicians Who Helped Win the Space RaceÉvaluation : 4 sur 5 étoiles4/5 (895)
- The Hard Thing About Hard Things: Building a Business When There Are No Easy AnswersD'EverandThe Hard Thing About Hard Things: Building a Business When There Are No Easy AnswersÉvaluation : 4.5 sur 5 étoiles4.5/5 (345)
- Shoe Dog: A Memoir by the Creator of NikeD'EverandShoe Dog: A Memoir by the Creator of NikeÉvaluation : 4.5 sur 5 étoiles4.5/5 (537)
- The Little Book of Hygge: Danish Secrets to Happy LivingD'EverandThe Little Book of Hygge: Danish Secrets to Happy LivingÉvaluation : 3.5 sur 5 étoiles3.5/5 (400)
- Elon Musk: Tesla, SpaceX, and the Quest for a Fantastic FutureD'EverandElon Musk: Tesla, SpaceX, and the Quest for a Fantastic FutureÉvaluation : 4.5 sur 5 étoiles4.5/5 (474)
- A Heartbreaking Work Of Staggering Genius: A Memoir Based on a True StoryD'EverandA Heartbreaking Work Of Staggering Genius: A Memoir Based on a True StoryÉvaluation : 3.5 sur 5 étoiles3.5/5 (231)
- On Fire: The (Burning) Case for a Green New DealD'EverandOn Fire: The (Burning) Case for a Green New DealÉvaluation : 4 sur 5 étoiles4/5 (74)
- The Emperor of All Maladies: A Biography of CancerD'EverandThe Emperor of All Maladies: A Biography of CancerÉvaluation : 4.5 sur 5 étoiles4.5/5 (271)
- Devil in the Grove: Thurgood Marshall, the Groveland Boys, and the Dawn of a New AmericaD'EverandDevil in the Grove: Thurgood Marshall, the Groveland Boys, and the Dawn of a New AmericaÉvaluation : 4.5 sur 5 étoiles4.5/5 (266)
- The Unwinding: An Inner History of the New AmericaD'EverandThe Unwinding: An Inner History of the New AmericaÉvaluation : 4 sur 5 étoiles4/5 (45)
- Team of Rivals: The Political Genius of Abraham LincolnD'EverandTeam of Rivals: The Political Genius of Abraham LincolnÉvaluation : 4.5 sur 5 étoiles4.5/5 (234)
- The World Is Flat 3.0: A Brief History of the Twenty-first CenturyD'EverandThe World Is Flat 3.0: A Brief History of the Twenty-first CenturyÉvaluation : 3.5 sur 5 étoiles3.5/5 (2259)
- The Gifts of Imperfection: Let Go of Who You Think You're Supposed to Be and Embrace Who You AreD'EverandThe Gifts of Imperfection: Let Go of Who You Think You're Supposed to Be and Embrace Who You AreÉvaluation : 4 sur 5 étoiles4/5 (1091)
- The Sympathizer: A Novel (Pulitzer Prize for Fiction)D'EverandThe Sympathizer: A Novel (Pulitzer Prize for Fiction)Évaluation : 4.5 sur 5 étoiles4.5/5 (121)
- Her Body and Other Parties: StoriesD'EverandHer Body and Other Parties: StoriesÉvaluation : 4 sur 5 étoiles4/5 (821)
- OD2e L4 Reading Comprehension AKDocument5 pagesOD2e L4 Reading Comprehension AKNadeen NabilPas encore d'évaluation
- 10 Ideas For ConversationDocument116 pages10 Ideas For ConversationGreenLake36100% (1)
- MAELLT Brochure 2006-1Document2 pagesMAELLT Brochure 2006-1Ryan Glenn Cedan CondaPas encore d'évaluation
- Shadow World in Search of Vurkanan TyesDocument10 pagesShadow World in Search of Vurkanan Tyesbob100% (1)
- Christian Borch & Gernot Bohme & Olafur Eliasson & Juhani Pallasmaa - Architectural Atmospheres-BirkhauserDocument112 pagesChristian Borch & Gernot Bohme & Olafur Eliasson & Juhani Pallasmaa - Architectural Atmospheres-BirkhauserAja100% (1)
- Babylon NaturalDocument31 pagesBabylon NaturalJoana CaeiroPas encore d'évaluation
- CHC40221 Workplace LogbookDocument14 pagesCHC40221 Workplace LogbookKarla GiaPas encore d'évaluation
- JBL Tune 115TWS HeadphoneDocument2 pagesJBL Tune 115TWS HeadphoneTimiPas encore d'évaluation
- AWSCertifiedBigDataSlides PDFDocument414 pagesAWSCertifiedBigDataSlides PDFUtsav PatelPas encore d'évaluation
- Climate Change Forests and Forest Management An O-Wageningen University and Research 481068Document145 pagesClimate Change Forests and Forest Management An O-Wageningen University and Research 481068gulnuromar034Pas encore d'évaluation
- Dragon Is The Fifth in The 12Document3 pagesDragon Is The Fifth in The 12Waylon CahiligPas encore d'évaluation
- Some FactsDocument36 pagesSome Factsapi-281629019Pas encore d'évaluation
- Security Operating Procedures and StandardsDocument5 pagesSecurity Operating Procedures and StandardsQuy Tranxuan100% (2)
- QuizDocument15 pagesQuizGracie ChongPas encore d'évaluation
- Siy Cong Bien Vs HSBCDocument2 pagesSiy Cong Bien Vs HSBCMJ Decolongon100% (1)
- Feature Fusion Based On Convolutional Neural Netwo PDFDocument8 pagesFeature Fusion Based On Convolutional Neural Netwo PDFNguyễn Thành TânPas encore d'évaluation
- Bakery Interview QuestionsDocument15 pagesBakery Interview QuestionsKrishna Chaudhary100% (2)
- 6.marketing ManagementDocument11 pages6.marketing ManagementadararaPas encore d'évaluation
- ECAT STD 2 Sample Question PaperDocument7 pagesECAT STD 2 Sample Question PaperVinay Jindal0% (1)
- 1, Philippine ConstitutionDocument2 pages1, Philippine ConstitutionJasmin KumarPas encore d'évaluation
- Financial Fitness ChecklistDocument4 pagesFinancial Fitness Checklistcoach_22Pas encore d'évaluation
- Surimi Technology: Submitted To: Dr.A.K.Singh (Sr. Scientist) Submitted By: Rahul Kumar (M.Tech, DT, 1 Year)Document13 pagesSurimi Technology: Submitted To: Dr.A.K.Singh (Sr. Scientist) Submitted By: Rahul Kumar (M.Tech, DT, 1 Year)rahuldtc100% (2)
- Tolkien Essay-TreesDocument10 pagesTolkien Essay-Treesapi-657753727Pas encore d'évaluation
- đề thi TADocument15 pagesđề thi TAĐào Nguyễn Duy TùngPas encore d'évaluation
- Toba Tek SinghDocument4 pagesToba Tek Singhrupal aroraPas encore d'évaluation
- Anthony D. Slonim, Murray M. Pollack Pediatric Critical Care Medicine PDFDocument950 pagesAnthony D. Slonim, Murray M. Pollack Pediatric Critical Care Medicine PDFAnca DumitruPas encore d'évaluation
- Math9 - Q2 - Mod2 - WK 3 - JointAndCombinedVariations - Version2Document37 pagesMath9 - Q2 - Mod2 - WK 3 - JointAndCombinedVariations - Version2Precious Arni100% (7)
- Creed Article 4 PPSXDocument17 pagesCreed Article 4 PPSXOremor RemerbPas encore d'évaluation
- AIPT 2021 GuidelineDocument4 pagesAIPT 2021 GuidelineThsavi WijayasinghePas encore d'évaluation
- The Wilderness - Chennai TimesDocument1 pageThe Wilderness - Chennai TimesNaveenPas encore d'évaluation