Académique Documents
Professionnel Documents
Culture Documents
Viewing of Complex Molecules
Transféré par
Mariano PerezCopyright
Formats disponibles
Partager ce document
Partager ou intégrer le document
Avez-vous trouvé ce document utile ?
Ce contenu est-il inapproprié ?
Signaler ce documentDroits d'auteur :
Formats disponibles
Viewing of Complex Molecules
Transféré par
Mariano PerezDroits d'auteur :
Formats disponibles
April 2001
Chem. Pharm. Bull. 49(4) 413417 (2001)
413
Viewing of Complex Molecules of Ethidium Bromide and Plasmid DNA in
Solution by Atomic Force Microscopy
Kuniharu UTSUNO, Masamichi TSUBOI,* Shunji KATSUMATA, and Toschitake IWAMOTO
High-tech Research Center, College of Science and Engineering, Iwaki Meisei University, Iwaki, Fukushima 9708551,
Japan. Received October 16, 2000; accepted December 25, 2000
Tertiary structure changes in plasmid DNA, induced by ethidium bromide intercalation, have been observed
in aqueous solutions by the use of an atomic force microscope. A relaxed closed circular pBR322 molecule became a positively supercoiled complex on the drug binding. The supercoiling always resulted in an interwound
(or a plectonemic) form, but never a solenoidal (or a toroidal) form. A quantitative analysis of the compactness of
such supercoiled complexes has been carried out.
Key words atomic force microscopy; plasmid DNA; ethidium bromide; interwound form of DNA duplex; plectonemic form of
DNA duplex
In a continuation of our series of studies on drugDNA interaction,13) we have examined topographic images of such
interacting molecules, using an atomic force microscope.
Atomic force microscopy (AFM) is an emerging technique
for direct observation of biological macromolecules and their
assemblies.49) One of the advantages of AFM, over other
high-resolution microscopies (electron microscopy, for example), is that sample preparation is relatively simple. It does
not involve negative staining or shadow casting with a metal
coating. In addition, the sample does not need to be kept in
vacuum. Through this technique, we have succeeded in viewing the tertiary structure of plasmid pBR322 DNA, kept in a
proper solution, in single molecule resolution. How and to
what extent is such an image of the tertiary structure altered
by the binding of various drugs? This question has been the
subject of our recent investigation.
Quite recently, Pope et al.10) published their AFM studies
of changes in the pBR322 DNA tertiary structure induced by
the binding of ethidium bromide. The results of their observation are different in some respects from what we have observed of the same drugDNA system. It would be significant, therefore, to publish our results on the ethidium
pBR322 DNA interaction here, before publishing our results
on other drugDNA interaction.
One of the main differences between the Pope et al. investigation and ours is in the sample morphology. In the Pope et
al. experiments, the plasmid DNA solution was spotted on
mica, and the mica was rinsed with H2O, and blown dry with
compressed N2. In our experiments, the DNA solution was
placed in a liquid cell, which was placed on mica, and the
DNA sample, adsorbed on mica, was kept in the solution
during the measurement without drying. It is another advantage of AFM that the imaging is permitted in buffer solution
without drying the sample. This is certainly advantageous,
not only because biological samples can be kept intact, but
also because the results of imaging can be correlated with
other experimental results in solution. On the interaction of
ethidium bromide and pBR322 DNA in solution, various
pieces of information were established by our previous
study.1) One molecule of ethidium bromide, when it is intercalated into a double-helical DNA, is known to unwind the
DNA duplex so that the base pair to base pair angle (viewed
along the helix axis) is reduced. Therefore, a relaxed closed
circular pBR322 DNA molecule is predicted to change into a
To whom correspondence should be addressed.
positively supercoiled form on an ethidium bromide binding.
We can also predict an approximate number of supercoils
(which is nearly equal to writhing number t ) in the plasmid
molecules kept at a given concentration in an aqueous solution which contains a given amount of ethidium bromide.
How do the individual molecules look in such a solution?
The results of our observation regarding this question will be
given below.
Experimental
Materials The sample of plasmid pBR322 DNA was purchased from
Takara Shuzo Co. Topoisomerase I from calf thymus was also purchased
from Takara Shuzo Co., and used without further purification. Ethidium bromide was purchased from Sigma Co.
AFM Plasmid pBR322 DNA was treated with topoisomerase I, and
then subjected to an agarose gel electrophoresis test. When it was confirmed
that the molecules were in a completely relaxed conformation, plasmid was
dissolved into 10 mM HEPES buffer (pH 7.0) containing 1 mM NiCl2.11) The
DNA concentration was always adjusted to 0.05 m g/ml. The ethidium bromide concentration was adjusted to 0, 0.25, 0.50, 0.75, 0.80, 0.90, 1.0, 2.0 or
10 m g/ml. The instrument used was Nanoscope III, Digital Instruments.
Freshly cleaved mica was placed on the instrument, and then the liquid cell
(an attachment provided by Digital Instruments) was set over the mica. The
sample solution was placed in this liquid cell (see Fig. 1). The instrument
was operated using tapping mode AFM. A cantilever equipped with an
oxide-sharpened silicon nitride probe was used. The scan rate was 5.09 Hz,
and tapping frequency was 9 to 10 kHz.
Electrophoresis Plasmid pBR322 DNA was dissolved up to 10 m g/ml
in buffer-T (35 mM TrisHCl (pH 8.0), 72 mM KCl, 5 mM MgCl2, 5 mM
dithiothreitol (DTT), 5 mM spermidine, and 0.01% bovine serum albumine).
The ethidium bromide concentration was adjusted to 0, 0.25, 0.50, 0.75,
1.00, 1.25, or 1.50 m g/ml. The mixture solution was incubated at 37 C for
10 min to complete the binding reaction. Next, the proper amount (6 units
for 200 m l of the mixture solution) of topoisomerase I was added, and the solution was incubated for 2 h, to cause a complete relaxation of the supercoiled plasmid (with the drug bound). Then, the drug and enzyme were re-
Fig. 1.
e-mail: tsuboima@iwakimu.ac.jp
Section of a Liquid Sample Cell for Atomic Force Microscope
2001 Pharmaceutical Society of Japan
414
Vol. 49, No. 4
Table 1. Writhing Number (t ) of the Complex Molecule of pBR322 DNA
and Ethidium Bromide, Estimated from Electrophoresis and Spectroscopic
Experiments
Experiment
Fig. 2. Electrophoresis Analysis of Topoisomers of pBR322 DNA, Produced by an Ethidium Bromide Binding and the Topoisomerase I Relaxation, Followed by the Removal of the Drug and Enzyme
1. Native intact pBR322 DNA; 2. Native pBR322 DNA treated with incubation, phenol extract, and ethanol precipitation in buffer-T containing no topoisomerase I and no
ethidium bromide; 39. Ethidium bromide had been added before topoisomerase was
introduced. DNA concentration510 m g/ml. Drug concentrations (m g/ml): 3, 0; 4, 0.25;
5, 0.50; 6, 0.75; 7, 1.00; 8, 1.25; 9, 1.50.
moved through phenol extraction, and the DNA was isolated by ethanol precipitation. The isolated DNA was then subjected to 1% agarose gel electrophoresis. The buffer used here was TBE (90 mM Trisborate and 2 mM
EDTA). The experiment was done by imposing 40 V, at room temperature
(25 C), for 20 h. After that, the gel was stained with ethidium bromide and
subjected to photography.
Results and Discussions
Configurations Predictable from Electrophoresis In
our previous study,1) the binding of ethidium bromide was
found to unwind DNA duplex. Let us start from a relaxed
closed circular plasmid molecule with 4362 base pairs. Suppose that the number of the bound drug molecules increases,
so that the total unwinding angle reaches 360. Then, the
number (b ) of helical turns must be reduced by one, so that b
becomes 436 to 435. Because the linking number (a ) of this
plasmid should remain unchanged, writhing number t must
increase by one, if the local molecular conformation is kept
unchanged, so that a law of conservation, a 5b 1t , is realized.
The number of ethidium bromide molecules bound to one
pBR322 DNA molecule in a solution must be a function of
the drug concentration in the solution. Therefore, we first examined the average t value of pBR322 DNA in a solution
with the same amount of ethidium bromide as that in the solution subjected to the AFM study. The t value can be estimated by using topoisomerase I relaxation followed by gel
electrophoresis.1) The result of this examination is shown in
Fig. 2. As shown in the figure, the ethidium bromide concentration 0.25 m g/ml caused an average linking number change
D a 52 for plasmid, 0.50 m g/ml caused D a 55, 0.75 m g/ml
caused D a 57, 1.00 m g/ml caused D a 510. Such observed
D a values were assumed to be entirely attributed to the
amounts of unwinding (D b ) caused by the drug binding.
Therefore, these must be equal to the t values caused by the
drug binding. The t values in this assumption are listed in
the second column of Table 1.
In correlating such results of observation with a prediction
for the sample solutions of our present AFM study, we must
take the difference in the solvents into account. The above t
values were determined in buffer-T, which was proper for the
topoisomerase I reaction, whereas present AFM sample solutions were prepared in 10 mM HEPES buffer containing 1 mM
Electrophoresis (see Fig. 2)
Absorption spectroscopy
Solvent
Buffer-T
HEPES11 mM NiCl2
DNA
concentration
10 m g/ml
0.05 m g/ml
t a)
(from D a )
Ethidium bromide concentration (m g/ml)
0
062
0.25
262
0.50
562
0.75
763
0.80
(ca. 8)b)
0.90
(ca. 9)b)
1.00
1063
1.25
1565
1.50
.15
2.00
(.15)
10.0
(.
.15)
m c)
t d)
(f 515)
t d)
(f 53)
0
421
622
745
765
800
831
895
945
1020
1332
0
18
26
31
32
33
35
37
39
43
56
0
4
5
6
6
7
7
7
8
9
11
a) t is assumed to be equal to the change of linking number, D a , in the electrophoresis experiment. b) Estimated by an interpolation. c) The number of drug
molecules bound to one pBR322 DNA molecule. d) t is assumed to be equal to
(m3f /360), where f is the angle of unwinding in the DNA duplex caused by one drug
molecule bound to it.
NiCl2. The change from Tris to HEPES was made because
DNA in HEPES was found to be adsorbed on mica better
than in Tris.12) Also, bovine serum albumin involved in
buffer-T may cause a disturbing image in AFM examination,
and should be avoided. A transition metal ion, such as Ni21
is known to be indispensable in a solvent for solution AFM
observation.11) Thus, it was found to be impossible to do the
electrophoresis and AFM experiments in the same solvent in
our present stage of experimental conditions. Therefore, we
did not attempt any direct correlation. Instead, we used the t
derived from the elctrophoresis experiment as a tentative reference.
Configurations Predictable from Absorption Spectroscopic Measurement Ethidium bromide shows an absorption peak at 480 nm, whereas ethidium bromide/DNA complex is at 520 nm. By the use of this optical property, we can
estimate the equilibrium constant (K) of this drugDNA system in various solvents, as was detailed previously.1) In our
AFM solvent, i.e. 10 mM HEPES buffer (pH 7.0)11 mM
NiCl2, it was found that K52.53105 M21 and n52.6, through
a Scatchard plot13) and a McGheevon Hippel analysis.14)
Here, n is the number of the base pairs occupied by one drug
molecule. From these K and n values, the number (m) of
drug molecules bound to one pBR322 DNA molecule is calculated for every pair of DNA concentration (0.05 m g/ml)
and drug concentration. The result is listed in the third column of Table 1. If the angle (f ) of unwinding of the DNA
duplex is assumed to the 15,1) t is given by t 5f 3m/360.
This is given in the forth column of Table 1.
Conformation Predictable for Each t Value For a
given value of t , two kinds of supercoiled conformations are
predictable. One is an interwound form (or a plectonemically
April 2001
415
Fig. 3. Diagrammatic Illustrations of Possible Tertiary Structures of Plasmid pBR322 DNA Molecules
The line here represents the axis of double-helical DNA chain. Nt is the number of
apparent intersection points, or the number of apparent nodes. This is considered to be
nearly equal to the writhing number t , which can be determined by electrophoresis experiment. a) Completely relaxed form. b) Interwound form where the number (Nt ) of
supercoil is 1. c) Toroidally supercoiled form with Nt 52. d) Plectonemically supercoiled form with Nt 52. e) Toroidally supercoiled form with Nt 56. f) Plectonemically
supercoiled form with Nt 56. g) Plectonemically supercoiled form with Nt 510. h)
Plectonemically supercoiled form with Nt 518.
supercoiled form). The other is a solenoidal form (or a
toroidally supercoiled form), in which the axis of the DNA
duplex lies on an imaginary torus. Some of these kinds of superhelical forms are illustrated in Fig. 3. Each of these eight
figures shows a possible projection of a plasmid molecule on
a two-dimensional plane. The line here represents an axis of
a double-helical DNA chain. The number (Nt ) of the intersection points of the line within each figure is supposed to be
the number of supercoilings of the plasmid molecule. This
number Nt can be correlated with the writhing number t , but
these two are not exactly equal to each other. In general t is
not an integer, whereas Nt is an integer. It would be reasonable, however, to assume that Nt is nearly equal to t with an
ambiguity of 1.0.
AFM Images Figure 4 is a collection of topographic images recorded for plasmid pBR322 DNA molecules kept in
solutions of 0.05 m g/ml. Here, the plasmid was completely
relaxed by the topoisomerase I reaction., and this was confirmed by electrophoresis. Without ethidium bromide, some
of each of the DNA molecules certainly looked circular with
no supercoil (Fig. 4a). Many of the chains, however, had 1
3 intersection points. This is partly attributable to the solvent
(10 mM HEPES11 mM NiCl2) and temperature (25 C), in
which the AFM experiment was made. These are different
from the conditions (buffer-T at 37 C) in which the plasmid
was relaxed. When ethidium bromide was added to the solution, apparently interwound forms were found to appear (Fig.
4b). In the range of ethidium concentration from 0.25 to
Fig. 4. AFM Images of Plasmid pBR322 DNA in Aqueous Solutions,
Containing Ethidium Bromide
DNA concentration: 0.05 m g/ml. Drug concentrations (m g/ml): a) 0, b) 0.75, c) 0.80 ,
d) 1.0, e) 2.0, f) 10. Magnification: every square here has a dimension of 1 m m31 m m.
0.75 m g/ml, however, the observed Nt values were found to
be mostly 2 or 3 only, except for unresolvable images. When
the concentration of ethidium bromide reached 0.80 m g/ml,
plectonemically supercoiled forms with 6 or 7 were observed. However, many of the loops in the supercoiled forms
could not be found resolved; they were observed as thick
rods. Such a rod is interpreted as a condensed plectonemic
supercoil (see Fig. 3g, for example). In the solution with
1.0 m g/ml ethidium bromide, more of such thick rods appeared (Figs. 4d, e). Still, one or two small loops were found
in each molecule. In the solution where the ethidium bromide
concentration was as high as 10 m g/ml, every plasmid molecule appeared as a thick rod. Some molecules were found to
be branched rods (Fig. 4f).
In surveying the observed AFM images, a general trend of
increasing of the t value with the ethidium bromide concentration was certainly confirmed. The upward gradient of t ,
however, is much smaller than what is expected from the
416
Vol. 49, No. 4
Table 2.
Extent of Image of Plasmid DNA as a Function of the Amount of Ethidium Bromide in the Solution
DNA extent
(3104 nm2)
15
610
1115
1620
2125
2630
31
Total
Ethidium bromide concentration (m g/ml)
0
0.25
0.50
0.75
0.80
0.90
1.0
2.0
10
2 (2)
4 (3)
33 (25)
43 (33)
20 (15)
16 (12)
13 (10)
131 (100)
1 (1)
12 (8)
39 (27)
50 (35)
26 (18)
7 (5)
7 (5)
142 (99)
0 (0)
9 (6)
41 (26)
61 (39)
28 (18)
11 (7)
8 (5)
158 (101)
1 (1)
10 (7)
41 (30)
44 (32)
23 (17)
9 (7)
9 (7)
137 (101)
3 (2)
22 (23)
56 (40)
26 (19)
9 (6)
7 (5)
7 (5)
140 (100)
6 (4)
35 (24)
47 (33)
29 (20)
16 (11)
4 (3)
7 (5)
144 (100)
11 (8)
55 (40)
44 (32)
16 (12)
1 (1)
5 (4)
6 (4)
138 (101)
35 (22)
96 (61)
18 (11)
5 (3)
1 (1)
1 (1)
1 (1)
157 (100)
39 (31)
63 (50)
14 (11)
3 (2)
0 (0)
1 (1)
6 (5)
126 (100)
Percentage is shown in parenthesis.
equilibrium constant (K52.53105 M21) and unwinding angle
(f 515) (see forth column of Table 1). At an ethidium bromide concentration of 0.80 m g/ml, for example, Nt was found
to be only 6 or 7 in AFM, whereas t 531 was expected from
the assumption of K52.53105 M21 and f 515. For such an
inconsistency, two factors may be taken into consideration.
First, the effective f value may be smaller than 15. Because a bending of the DNA duplex would increase its torsional rigidity,15) an effective f may become smaller as the
number of bound drug molecules increases. At the ethidium
bromide concentration50.80 m g/ml, for example, nearly half
of the drug sites on pBR322 DNA (having 4362 bp) are occupied (m5765 bp and 2.63m51989 bp). If f 53, however,
t can be only 6 (576533/360). This notion is not sufficient to explain what we observed in our AFM experiment.
Thus, as shown in the last column of Table 1, the expected t
values are much smaller than what were actually observed.
Secondly, in low ionic strength, the linking number deficit
D a is not totally partitioned to D t , but partly to D b .8,16) The
AFM solvent we used has only 1 mM NiCl2, and probably because of the PO22PO22 repulsion, t may be smaller than
what is expected on the assumption that D a 5D t . Unfortunately, the amount of this difference cannot be estimated
using our present knowledge.
Compactness of the Complex Molecule A survey of
the images shown in Figs. 4a to 4e gave us a feeling that the
complex molecule became more and more compact as the
amount of the bound drug molecules increased. To fix this
notion on a more firm basis, let us define an extent of
image as the product of its length along the long axis and
the length along the axis perpendicular to the long axis. We
measured such an extent of image for every recorded
image of plasmid DNA, and the results are summarized in
Table 2. It became clear that the extent was lowered as the
drug concentration in the solution was elevated. A slightly
more detailed survey, however, indicates that the lowering of
the extent is small when going from an ethidium concentration of 0 to 0.75 m g/ml, and that more prominent lowering
starts at about 0.80 m g/ml and continues to 10 m g/ml. An interpretation of this phenomenon is yet to be done.
Particular Shapes In a microscopic study, it is important not only to deduce a general trend from a survey of
many images, but also to observe individual images in detail.
A special shape of a particular molecule could possibly be
significant.
The image in the left-most frame of Fig. 4c is for a plasmid pBR322 DNA molecule observed at an ethidium bro-
mide concentration50.80 m g/ml. This is a plectonemically
and positively supercoiled molecule with the number of supercoilings at 6 (compare with the illustration given in Fig.
3f). The image in the central frame of Fig. 4c has probably
the same number of ethidium bromide molecules and the
same number of coilings. Its shape, however, is quite different; its two interwound small loops are crowded in the central
portion, and its two large loops are placed outside.
What is shown in the left-most frame of Fig. 4d is an
image of a plasmid molecule observed at an ethidium bromide concentration51.0 m g/ml. This is a plectonemically supercoiled with a t value of about 10. Its interpretation is
given by an illustration in Fig. 3g. In the right-most frame of
Fig. 4f is an image for a branched plectoneme. Here, the
number of supercoils must be greater than 15. Its duplex
chain axis is considered to be folded as shown in Fig. 3h.
It is interesting that, contrary to the Pope et al. observation, we could not find any image that was assignable to a
toroidally supercoiled form.
A Comparison with the Results of a Previous Study
In the above mentioned AFM study, Pope et al.10) recorded
topographic images from DNA complexes of various ethidium bromide/base pair stoichiometries, ranging from 1/10 to
8/1. They observed a toroidally supercoiled form in a very
dilute ethidium bromide concentration, for a drug/base pair
stoichiometry of 1/5 to 1/1. On the other hand, we did not observe this form in the whole concentration range examined
(010 m g/ml).
In explaining such a difference between the images observed by Pope et al. and by us, three factors may be taken
into consideration. First, Pope et al. used a natural, heavily
supercoiled pBR322 DNA, whereas we used a relaxed closed
circular pBR322 DNA. Thus, in the Pope et al. experiment,
pBR322 DNA had a high D a value even with a very small
number of drug molecules bound. Secondly, the effective
equilibrium constant in the Pope et al. system is extraordinarily high. Unfortunately, Pope et al. ignored our detailed
study1) on the ethidium bromide1pBR322 system, which estimated the equilibrium constant values in six different solvents. Instead, they estimated the equilibrium constant by the
method described by Coury et al.17) The estimated value was
1.43106 M21. This is just what is expected in a solvent with
no salt,1) and is probably higher than that in our system with
1 mM NiCl2 by one order of magnitude. Third, some unknown reactions may occur in the process of spotted on 3aminopropyltriethoxy silanized mica (AP-mica), gently
rinsed with 20 ml H2O, and immediately blown dry with
April 2001
compressed N2, which was involved in the Pope et al. sampling, and which was not involved in our sampling.
Acknowledgements This work was supported by a High-tech Research
Center project. Kuniharu Utsuno was supported by a High-tech Research
Center postdoctoral fellowship. We thank Ms. Maki Ueno for her help in our
AFM experiment.
References
1) Utsuno K., Tsuboi M., Chem. Pharm. Bull., 45, 15511557 (1997).
2) Utsuno K., Kojima K., Maeda Y., Tsuboi M., Chem. Pharm. Bull., 46,
16671671 (1998).
3) Utsuno K., Maeda M., Tsuboi M., Chem. Pharm. Bull., 47, 1363
1368 (1999).
4) Rippe K., Mcke N., Langowski J., Nucleic Acids Res., 25, 1736
1744 (1997).
5) Shlyakhtenko L. S., Gall A. A., Weimer J. J., Hawn D. D., Lyubchenko
Y. L., Biophys. J., 77, 568576 (1999).
6) Bustamante C., Vesenka J., Tang C. L., Rees W., Guthold M., Keller
R., Biochemistry, 31, 2226 (1992).
417
7)
8)
9)
10)
11)
12)
13)
14)
15)
16)
17)
Shlyakhtenko L. S., Potaman V. N., Sinden R. R., Lyubchenko Y. L., J.
Mol. Biol., 280, 6172 (1998).
Lyubchenko Y. L., Shlyakhtenko L. S., Proc. Natl. Acad. Sci. U.S.A.,
94, 496501 (1997).
Tsuboi M., Katsumata S., Gendai Kagaku (Chemistry Today), No.
353, pp. 1825 (August 2000).
Pope L. H., Davies M. C., Laughton C. A., Roberts C. J., Tendler S. J.
B., Williams P. M., J. Microsc., 199, 6878 (2000).
Hansma H. G., Laney D. E., Biophys. J., 70, 19331039 (1996).
Bezanilla M., Drake B., Nudler E., Kashlev M., Hansma P. K.,
Hansma H. G., Biophys. J., 67, 24542459 (1994).
Scatchard G., Ann. N.Y. Acad. Sci., 51, 660672 (1949).
McGhee J. D., von Hippel P. H., J. Mol. Biol., 86, 469489 (1974).
Heath P. J., Clendenning J. B., Fujimoto B. S., Schurr J. M., J. Mol.
Biol., 260, 718730 (1996).
Bednar J., Furrer P., Stasiak A., Dubochet J., Egelman E. H., Bate A.
D., J. Mol. Biol., 235, 825847 (1994).
Coury J. E., McFail-Isom L., Williams L. D., Bottomley L. A., Proc.
Natl. Acad. Sci. U.S.A., 93, 1228312286 (1996).
Vous aimerez peut-être aussi
- Gyrase Inhibitors Induce An Oxidative Damage Cellular Death Pathway in Escherichia ColiDocument15 pagesGyrase Inhibitors Induce An Oxidative Damage Cellular Death Pathway in Escherichia ColiMariano PerezPas encore d'évaluation
- Protein Dna RecognitionDocument31 pagesProtein Dna RecognitionMariano PerezPas encore d'évaluation
- Peroxiredoxin Systems Structures and FunctionsDocument409 pagesPeroxiredoxin Systems Structures and FunctionsMariano PerezPas encore d'évaluation
- Disulfide Bond Formation and Cysteine Exclusion inDocument10 pagesDisulfide Bond Formation and Cysteine Exclusion inMariano PerezPas encore d'évaluation
- CHOP Induces Death by Promoting Protein Synthesis and Oxidation in The Stressed Endoplasmic ReticulumDocument13 pagesCHOP Induces Death by Promoting Protein Synthesis and Oxidation in The Stressed Endoplasmic ReticulumMariano PerezPas encore d'évaluation
- The Ubiquitin-Proteasome Proteolytic System From Classical Biochemistry To Human DiseasesDocument242 pagesThe Ubiquitin-Proteasome Proteolytic System From Classical Biochemistry To Human DiseasesMariano PerezPas encore d'évaluation
- A Pathway For Disulfide Bond Formation in VivoDocument5 pagesA Pathway For Disulfide Bond Formation in VivoMariano PerezPas encore d'évaluation
- Microplate-Based Kinetic Method For Assay of Mitochondrial NADH - and Succinate - Cytochrome C Reductase ActivitiesDocument3 pagesMicroplate-Based Kinetic Method For Assay of Mitochondrial NADH - and Succinate - Cytochrome C Reductase ActivitiesMariano PerezPas encore d'évaluation
- NAD+ and NADH in Cellular Functions and Cell DeathDocument3 pagesNAD+ and NADH in Cellular Functions and Cell DeathMariano PerezPas encore d'évaluation
- Contribution of Calcium Influx in Mediating Glucose-Stimulated Oxygen Consumption in Pancreatic IsletsDocument11 pagesContribution of Calcium Influx in Mediating Glucose-Stimulated Oxygen Consumption in Pancreatic IsletsMariano PerezPas encore d'évaluation
- Redox Sensing by Escherichia Coli Effects of DithiothreitolDocument4 pagesRedox Sensing by Escherichia Coli Effects of DithiothreitolMariano PerezPas encore d'évaluation
- Mitochondria/Cytosol Fractionation Kit: Sufficient For Analysis of 50 SamplesDocument8 pagesMitochondria/Cytosol Fractionation Kit: Sufficient For Analysis of 50 SamplesMariano PerezPas encore d'évaluation
- Targeting Metabolism To Diagnose and TreatDocument4 pagesTargeting Metabolism To Diagnose and TreatMariano PerezPas encore d'évaluation
- A Molecular Mechanism of Pyruvate Protection Against Cytotoxicity of Reactive Oxygen SpeciesDocument11 pagesA Molecular Mechanism of Pyruvate Protection Against Cytotoxicity of Reactive Oxygen SpeciesMariano PerezPas encore d'évaluation
- Generation of Rho0 Cells Utilizing A Mitochondrially Targeted Restriction Endonuclease and Comparative AnalysesDocument10 pagesGeneration of Rho0 Cells Utilizing A Mitochondrially Targeted Restriction Endonuclease and Comparative AnalysesMariano PerezPas encore d'évaluation
- Reactive Oxygen Species in Cancer Cells Live by The Sword Die by The Sword 2006 Cancer CellDocument2 pagesReactive Oxygen Species in Cancer Cells Live by The Sword Die by The Sword 2006 Cancer CellMariano PerezPas encore d'évaluation
- Selective Removal of Mitochondria Via MitophagyDocument7 pagesSelective Removal of Mitochondria Via MitophagyMariano PerezPas encore d'évaluation
- Phase II Trial of Temozolomide Plus O6-Benzylguanine in Adults With Recurrent, Temozolomide-Resistant Malignant GliomaDocument6 pagesPhase II Trial of Temozolomide Plus O6-Benzylguanine in Adults With Recurrent, Temozolomide-Resistant Malignant GliomaMariano PerezPas encore d'évaluation
- Ratiometric Calcium IndicatorsDocument1 pageRatiometric Calcium IndicatorsMariano PerezPas encore d'évaluation
- Mitochondria/Cytosol Fractionation Kit: Sufficient For Analysis of 50 SamplesDocument8 pagesMitochondria/Cytosol Fractionation Kit: Sufficient For Analysis of 50 SamplesMariano PerezPas encore d'évaluation
- Selective Removal of Mitochondria Via MitophagyDocument7 pagesSelective Removal of Mitochondria Via MitophagyMariano PerezPas encore d'évaluation
- Ratiometric Calcium IndicatorsDocument1 pageRatiometric Calcium IndicatorsMariano PerezPas encore d'évaluation
- The Rate of Oxygen Utilization by CellsDocument32 pagesThe Rate of Oxygen Utilization by CellsMariano PerezPas encore d'évaluation
- Oxidative Protein Folding Is DrivenDocument11 pagesOxidative Protein Folding Is DrivenMariano PerezPas encore d'évaluation
- Protein Folding in The Bacterial PeriplasmDocument7 pagesProtein Folding in The Bacterial PeriplasmMariano PerezPas encore d'évaluation
- Oxidative Protein Folding Is DrivenDocument11 pagesOxidative Protein Folding Is DrivenMariano PerezPas encore d'évaluation
- Fluorescent Dyes Introduction Ratiometric AssaysDocument2 pagesFluorescent Dyes Introduction Ratiometric AssaysMariano PerezPas encore d'évaluation
- An Assessment of Prediction Algorithms For Nucleosome PositioningDocument1 pageAn Assessment of Prediction Algorithms For Nucleosome PositioningMariano PerezPas encore d'évaluation
- An Assessment of Prediction Algorithms For Nucleosome PositioningDocument1 pageAn Assessment of Prediction Algorithms For Nucleosome PositioningMariano PerezPas encore d'évaluation
- Antibiotic Efficacy Is Linked To Bacterial Cellular Respiration.Document8 pagesAntibiotic Efficacy Is Linked To Bacterial Cellular Respiration.Mariano PerezPas encore d'évaluation
- Shoe Dog: A Memoir by the Creator of NikeD'EverandShoe Dog: A Memoir by the Creator of NikeÉvaluation : 4.5 sur 5 étoiles4.5/5 (537)
- The Subtle Art of Not Giving a F*ck: A Counterintuitive Approach to Living a Good LifeD'EverandThe Subtle Art of Not Giving a F*ck: A Counterintuitive Approach to Living a Good LifeÉvaluation : 4 sur 5 étoiles4/5 (5794)
- Hidden Figures: The American Dream and the Untold Story of the Black Women Mathematicians Who Helped Win the Space RaceD'EverandHidden Figures: The American Dream and the Untold Story of the Black Women Mathematicians Who Helped Win the Space RaceÉvaluation : 4 sur 5 étoiles4/5 (890)
- The Yellow House: A Memoir (2019 National Book Award Winner)D'EverandThe Yellow House: A Memoir (2019 National Book Award Winner)Évaluation : 4 sur 5 étoiles4/5 (98)
- The Little Book of Hygge: Danish Secrets to Happy LivingD'EverandThe Little Book of Hygge: Danish Secrets to Happy LivingÉvaluation : 3.5 sur 5 étoiles3.5/5 (399)
- A Heartbreaking Work Of Staggering Genius: A Memoir Based on a True StoryD'EverandA Heartbreaking Work Of Staggering Genius: A Memoir Based on a True StoryÉvaluation : 3.5 sur 5 étoiles3.5/5 (231)
- Never Split the Difference: Negotiating As If Your Life Depended On ItD'EverandNever Split the Difference: Negotiating As If Your Life Depended On ItÉvaluation : 4.5 sur 5 étoiles4.5/5 (838)
- Elon Musk: Tesla, SpaceX, and the Quest for a Fantastic FutureD'EverandElon Musk: Tesla, SpaceX, and the Quest for a Fantastic FutureÉvaluation : 4.5 sur 5 étoiles4.5/5 (474)
- The Hard Thing About Hard Things: Building a Business When There Are No Easy AnswersD'EverandThe Hard Thing About Hard Things: Building a Business When There Are No Easy AnswersÉvaluation : 4.5 sur 5 étoiles4.5/5 (344)
- Grit: The Power of Passion and PerseveranceD'EverandGrit: The Power of Passion and PerseveranceÉvaluation : 4 sur 5 étoiles4/5 (587)
- On Fire: The (Burning) Case for a Green New DealD'EverandOn Fire: The (Burning) Case for a Green New DealÉvaluation : 4 sur 5 étoiles4/5 (73)
- The Emperor of All Maladies: A Biography of CancerD'EverandThe Emperor of All Maladies: A Biography of CancerÉvaluation : 4.5 sur 5 étoiles4.5/5 (271)
- Devil in the Grove: Thurgood Marshall, the Groveland Boys, and the Dawn of a New AmericaD'EverandDevil in the Grove: Thurgood Marshall, the Groveland Boys, and the Dawn of a New AmericaÉvaluation : 4.5 sur 5 étoiles4.5/5 (265)
- The Gifts of Imperfection: Let Go of Who You Think You're Supposed to Be and Embrace Who You AreD'EverandThe Gifts of Imperfection: Let Go of Who You Think You're Supposed to Be and Embrace Who You AreÉvaluation : 4 sur 5 étoiles4/5 (1090)
- Team of Rivals: The Political Genius of Abraham LincolnD'EverandTeam of Rivals: The Political Genius of Abraham LincolnÉvaluation : 4.5 sur 5 étoiles4.5/5 (234)
- The World Is Flat 3.0: A Brief History of the Twenty-first CenturyD'EverandThe World Is Flat 3.0: A Brief History of the Twenty-first CenturyÉvaluation : 3.5 sur 5 étoiles3.5/5 (2219)
- The Unwinding: An Inner History of the New AmericaD'EverandThe Unwinding: An Inner History of the New AmericaÉvaluation : 4 sur 5 étoiles4/5 (45)
- The Sympathizer: A Novel (Pulitzer Prize for Fiction)D'EverandThe Sympathizer: A Novel (Pulitzer Prize for Fiction)Évaluation : 4.5 sur 5 étoiles4.5/5 (119)
- Her Body and Other Parties: StoriesD'EverandHer Body and Other Parties: StoriesÉvaluation : 4 sur 5 étoiles4/5 (821)
- Chemical Waste Disposal of NEUSTDocument2 pagesChemical Waste Disposal of NEUSTRoniel Urbino GamboaPas encore d'évaluation
- The Sthir Karakas: Functions of The Char Karkas AtmakarakDocument2 pagesThe Sthir Karakas: Functions of The Char Karkas AtmakarakDipyaman SahaPas encore d'évaluation
- Eddy CurrentDocument9 pagesEddy Currentkrish_cvr2937Pas encore d'évaluation
- T Tai: U YstDocument7 pagesT Tai: U YstDani Romero GarzònPas encore d'évaluation
- Jantzen Brix P. Caliwliw ME-1 A42 Chapter 2: Atoms, Molecules, and IonsDocument2 pagesJantzen Brix P. Caliwliw ME-1 A42 Chapter 2: Atoms, Molecules, and IonsJantzenCaliwliwPas encore d'évaluation
- English ClassDocument5 pagesEnglish Classmarina55555Pas encore d'évaluation
- Examen IsothermDocument9 pagesExamen IsothermAlonso FloresPas encore d'évaluation
- uploadedBrislingtonCurriculumSchool Closure ResourcesWeek 5specialist CentreYear 9Document13 pagesuploadedBrislingtonCurriculumSchool Closure ResourcesWeek 5specialist CentreYear 9Hitdifferentszaa LovePas encore d'évaluation
- Chinese Cable Details DataDocument131 pagesChinese Cable Details DataJoyanta DebPas encore d'évaluation
- Project Specs - HDPE PipeDocument1 pageProject Specs - HDPE PipeShehbaaz FarzaniPas encore d'évaluation
- A Review On Preparation of Low Cost Adhesive From Waste Materilal Using Citrus FruitsDocument10 pagesA Review On Preparation of Low Cost Adhesive From Waste Materilal Using Citrus FruitsPRASHANT INGOLEPas encore d'évaluation
- IIT KHRGPR - LLB - SampleDocument8 pagesIIT KHRGPR - LLB - Samplepiyush sinhaPas encore d'évaluation
- Essential Words For The TOEFLDocument28 pagesEssential Words For The TOEFLpritam golePas encore d'évaluation
- Rock Deformation: The Study of Stresses and StrainsDocument34 pagesRock Deformation: The Study of Stresses and StrainsStephen CeresPas encore d'évaluation
- 12 Interview Question Related To Non - Destructive TestingDocument4 pages12 Interview Question Related To Non - Destructive TestingHary adiPas encore d'évaluation
- Catalogo Completo Cooper LightingDocument130 pagesCatalogo Completo Cooper LightingWilliam Dante Mescco SairePas encore d'évaluation
- Replacement of Gas Phase With Liquid HexamineDocument6 pagesReplacement of Gas Phase With Liquid HexaminePradhita Ramdani HPas encore d'évaluation
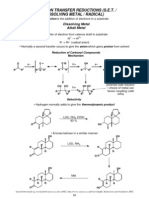
- Metal ReductionDocument11 pagesMetal Reductiondeepthimahanthi sabithaPas encore d'évaluation
- Chemical Reactions and Balancing Chemical Equations Web Quest 1Document5 pagesChemical Reactions and Balancing Chemical Equations Web Quest 1api-2625864460% (1)
- Alloyed: Case-Hardening Steels RM16 - RM16PBDocument3 pagesAlloyed: Case-Hardening Steels RM16 - RM16PBShubhangi ShindePas encore d'évaluation
- Graham Cracker ExperimentDocument12 pagesGraham Cracker Experimentapi-2961255610% (1)
- Chapter 9: TidesDocument40 pagesChapter 9: TidesCarol GirottoPas encore d'évaluation
- 5054 Space Physics Practice QuestionsDocument14 pages5054 Space Physics Practice QuestionsSoya BeansPas encore d'évaluation
- BEHAVIOR OF STRUCTURES UNDER BLAST LOADINGDocument32 pagesBEHAVIOR OF STRUCTURES UNDER BLAST LOADINGramyashri inalaPas encore d'évaluation
- Salt Water and BrineDocument2 pagesSalt Water and BrineleopumpkingPas encore d'évaluation
- BL ExoticaDocument91 pagesBL ExoticaFREDDYMENAPas encore d'évaluation
- Reduction of Phenylalanine To AmphetamineDocument2 pagesReduction of Phenylalanine To AmphetamineFlorian FischerPas encore d'évaluation
- Diathermy Cryo LASER 2023Document81 pagesDiathermy Cryo LASER 2023chakshu chaudhryPas encore d'évaluation
- NQ Panelboards - NQ454L4C PDFDocument2 pagesNQ Panelboards - NQ454L4C PDFdarniel quimbiulcoPas encore d'évaluation
- Fluid Mechanics Hydraulics by Gillesania Chapter 1Document15 pagesFluid Mechanics Hydraulics by Gillesania Chapter 1Mary Grace BitantesPas encore d'évaluation