Académique Documents
Professionnel Documents
Culture Documents
Electrical Communication 45
Transféré par
Anonymous t7MdBjnOCopyright
Formats disponibles
Partager ce document
Partager ou intégrer le document
Avez-vous trouvé ce document utile ?
Ce contenu est-il inapproprié ?
Signaler ce documentDroits d'auteur :
Formats disponibles
Electrical Communication 45
Transféré par
Anonymous t7MdBjnODroits d'auteur :
Formats disponibles
5thSASTech 2011, Khavaran Higher-education Institute, Mashhad, Iran. May 12-14.
Facile synthesis of WS2 inorganic fullerene-like nanomaterials from
WS2 amorphous nanoparticles
S.Salar Meshkat1*, H.R.Gholami1
1Address: Iran, Urmia, Urmia University of Technology, Chemical Engineering Facaulty
E-mail:s.meshkat@che.uut.ac.ir
Paper Reference Number : 0104-701
Name of the Presenter: S.Salar Meshkat
Abstract
The amorphous WS2 nanoparticles have been synthesized by a simple oxidation-reduction
reaction in an aqueous solution. A series of products with different morphologies, such as WS2 nano
spheres, inorganic fullerene-like nanospheres, nanorods and W bended rods, can be obtained by
annealing the amorphous WS2 nanoparticles under N2 atmosphere under 400-1200 0C. These products
have been characterized by X-ray diffraction, field emission scanning electronic microscopy,
transmission electron microscopy and high-resolution transmission electron microscopy in detail. The
possible transformation mechanism for the structure has been discussed based on the experimental
results. In addition, the optical properties of IF-WS2 have also been performed by UV-vis absorption
spectroscopy.
Keywords: Fullerene, Nanostructures, Chemical synthesis, X-ray diffraction, Crystal structure
1.Introduction
Since the discovery of C60 molecule and carbon nanotubes, intense interests have been aroused to
synthesize inorganic materials with fullerene-like (IF) structures and nanotubes from different
inorganic compounds [1,2 .] During the past years, the synthesis of transition metal dichalcognides
MX2 (M = W, Mo; X = S, Se) with IF structure and nanotubes [3-5] has opened a challenging new
field in solid state physics, chemistry and materials science with a wide range of possible
applications, such as electrochemical hydrogen storage [6], cathode material for rechargeable lithium
batteries and solar cell materials [7,8], electric transport [9], useful solid lubricant [10], sulfur removal
catalyst [11] and an intercalation host [12]. Since the IF-MX2 initially synthesized by Tenne and
co-workers, a large variety of forms ranging from nanoparticles, nanotubes, nanosheets,
5thSASTech 2011, Khavaran Higher-education Institute, Mashhad, Iran. May 12-14.
nanofibers, and nanobelts to fullerenes have been obtained [13-15]. In order to elucidate the growth
mechanism and to produce the materials for large-scale commercial applications, many synthetic
routes have been developed from their first formation from respective trioxides in H 2S and N2/H2
atmosphere by gas solid or gas-gas reactions, such as electron-beam irradiation activation and arc
discharge [16,17], hydrothermal or solvothermal synthesis [18,19], sonochemical process [20],
template synthesis [21], atmospheric pressure chemical vapor deposition [22,23] and
electrochemical/chemical synthesis [24]. An economical and effective method for large-scale
synthesis of high quality IF-MoS2 and WS2 is always a challenge for the scientists. Here we report a
simple and low-cost route to synthesize the amorphous WS2 nanoparticles by oxidation-reduction
reaction in aqueous solution. A series of products with different morphologies, such as WS2
nanospheres, IF-nanospheres and nanorods can be obtained by annealing the amorphous WS2 in N2
atmosphere. The interest in WS2 nanostructure is considered a focal at present. In the paper, we
investigate the transformation of nanostructure of WS2 annealed at different temperatures in detail in
order to evaluate the thermal stability of IF structure and to control the morphology of samples. The
synthesis route may offer an opportunity for fundamental studies and technological applications.
2. Experimental
1.2 Preparation
The preparation of amorphous WS2 nanoparticles was carried out by adding (NH4)6W7O24 4H2O (AR,
0.88 g) and Na2S 9H2O (AR, 2.64 g) to 100 mL HCl solution (0.8 mol L -1) in a 250 mL roundbottom flask with vigorous stirring. Heated at 80 8C for 0.5 h, NH2OH HCl (AR, 0.7 g) was added all
at once then with vigorous stirring for 1 h . The black powder finally was obtained by centrifugal
separation and washed with deionized water for several times to remove the residue of the reactants,
then dried in the air to obtain the final product of WS2 powder .The WS2 powder was annealed in a
sealed muffle under N2 atmosphere (0.5-0.8 501Pa) for 2 h at different temperatures (400-1200 8C).
The detailed experimental equipment is described in the reference [25].
2.2 Characterizations
The phase-structures and morphology of the as-prepared samples were characterized by X-ray
diffraction (XRD ,D/max-rA) employing Cu Ka radiation, field emission scanning electronic
microscopy (FESEM, JSM-6700F ,)transmission electron microscopy (TEM, H-800EM), and highresolution transmission electron microscopy (HRTEM ,JEM-3010). Optical characterization of IFMoS2 was performed by UV-vis absorption spectroscopy (UV-vis).
3.Results and discussion
Fig. 1 shows the XRD patterns of the final products synthesized at different annealing temperatures.
Fig. 1(a) indicates that the X-ray spectrum is confined to a wide and weak (0 0 2) peak of the
amorphous WS2 synthesized by the oxidation-reduction described by the following reaction:
1) (NH4)2WO4 + 4Na2S + 8HCl (NH4)2WS4 +8NaCl +4H2O
2) (NH4)2WS4 + 2NH2OH + HCl WS2 + N2 + 2NH4Cl + 2H2O + 2H2S
With the temperature increasing from 400 to 900 8C, more peaks appeared and became sharper
meanwhile the peak intensities increased rapidly. The above results indicated that the morphology
became more integrated and 2H-WS2 with characteristic peaks of (0 0 2), (0 0 4), (1 0 0), (1 0 2), (1
5thSASTech 2011, Khavaran Higher-education Institute, Mashhad, Iran. May 12-14.
0 3), (1 0 4), (1 0 5), (1 1 0), (1 1 2), (1 1 6), (2 0 0 (appeared at the annealing temperature of 900 0C
(indexing JCPDS file No. 87-2416 .)But the (0 0 2) peaks during the thermal process have no distinct
shift comparing with the corresponding value (about %2 reported by Tenne et al. With the
temperature increasing to 1000 0C, W2S3 appeared and at 1100 8C W2S3 mostly transformed into Mo.
Pure body-centered cubic metal Mo is obtained at 1200 0C with characteristic peaks of (110) and (2 2
0). Thus, controlling suitable temperature is a key factor to obtain samples with different
morphologies .
Fig. 2 shows the morphologies of the WS2, W2S3 and W obtained at different annealing temperatures.
It was obvious that the morphology and the size of the samples were distinctly different during the
thermal treatment. From Fig. 2(a) it could be seen that the sample annealed at 400 8C were mainly
small nanoparticles with the diameter about 30 nm. While during the thermal treatment at 800 8C, a
majority of nanoparticles grew along c axes then WS2 nanorods can be obtained. Indicated from XRD
and FESEM images, sheet W2S3 and W bended nanorods were obtained at 1000 0C and 1200 0C,
respectively .
The TEM images in Fig. 3 revealed the dramatic changes in the overall appearance of the samples
following the heat treatments. At lower annealing temperature, such as 400 0C, near monodisperse
WS2 nanospheres can be obtained and the majority is with the diameter of 20-70 nm (Fig. 3(a)).
Driven by the thermal energy, the IF shell come into being due to the sulfur bonds breaking for
recrystallization of amorphous WS2 and the elimination of unsaturated bonds at the edge of WS2 (Fig.
3(b)). With the temperature increased to 700 0C, elliptical polyhedron of WS2 with nested core is
produced with the port connected each other (Fig. 3(c)). And the rod-like WS2 is obtained at 800 0 0C
with 30 nm in diameters and 100-200 nm in length (Fig. 3(d)). The electron diffraction pattern of Fig.
3(g) indicates the WS2 with (0 0 2), (1 0 0), (1 0 3) and (1 1 0) planes. With the temperature increased
to 1000 0C, clubbed W2S3 with core-shell structure appears (Fig. 3(e)). The selected area electron
diffraction of Fig. 3(h) shows the Mo2S3 single crystal shell. At elevated temperature of 1200 0C rodlike W is the main phase because of the decomposition of WS2 (Fig. 3(f)). The floc on Mo surface
may be the amorphous sulfide formed during the descent of temperature. Thus, the annealing
temperature should be controlled at 500-700 0C in order to get IF structure .
More details for WS2 at elevated temperatures are illustrated by HRTEM studies in Fig. 4. The series
of images indicated the transformation in IF structures. The morphologies indicate that at the
annealing temperatures of 600 0C the nested and closed structure with several layers of the particle is
relatively integrated but the defect is obvious at the corner (Fig. 4(b)). However, with the annealing
temperature increased to 900 0C, the closed cage is broken and a side in IF structure disappears (Fig.
4(c)). The typical morphology of irregular strip is showed in Fig. 4(d) with fringe distance of 0.62 nm
and 5 nm wide under the same thermal situation. Furthermore, bended rod of W2S3 and open-ended W
rod with core-shell structure was observed at the elevated temperature of 1000 0C and 1100 0C (Fig.
4(e and f)), respectively. And the shell indicates the fringe thickness of 0.21 nm for W (1 1 0) lattice
(Fig. 4f) .The growth mechanism for IF structure can be described as follows: According to XRD and
energy-dispersive spectroscopy results, the initially obtained nanoparticles are amorphous and the
molar ratio of sulfur to molybdenum is about 2.2:1 (the EDS spectra are not shown here). During the
thermal treatment, the rich sulfur bonds broke first for recrystallization of WS2. Driven by thermal
kinetics, the IF nanoparticles were obtained by eliminating the unsaturated bonds to form a
closed structure. While, the IF nanostructures in our experiment have many defects especially at the
corner and periphery. The defects may help to release some strain in the folded layers, so the d(0 0 2 (
have no obvious change and the (0 0 2) peaks in XRD do not show distinct shifts. At elevated
temperature, the closed structures become unstable and the layers brake, thus the nanorods of WS2 are
5thSASTech 2011, Khavaran Higher-education Institute, Mashhad, Iran. May 12-14.
formed. Due to the decomposition of WS2 by controlling the temperature, a special phase of W/WS2
(Fig. 4(f)) and an intergradation of W2S3( Fig. 2(c)) come into being. Because the disconnection
could easily occur at the corner, the metal Mo with branches can be obtained. The unique morphology
for W may be applied as accession to enhance mechanical performance and offer catalytic
applications .Optical characterization of IF-WS2 was performed by UV-vis absorption spectroscopy at
room temperature .Fig. 5 shows the UV-vis absorption spectra of IF-WS2 suspension. The spectral
data show stronger absorption in the blue region and weaker absorption in the red region. It displayed
a strong and broad line from 290 to 400 nm with a maximum centered at about 340 nm and weaker
absorbance during 500-670 nm (in Fig. 5(b and c)). It is known that the direct band gap of WS2 is
located in the visible (ca. 700 nm) and two further more bands occur at ca. 500 nm and 350 nm both
associated with the direct transitions from the valence band to the conduction band [26]. Fig. 5
matched well with the above description, although the band threshold for the less energetic direct
transition is slightly blue shifted from the typical wavelength of 700 nm. This may be because that the
WS2 nanoparticles used in the test have bigger dimensions than 4.5 nm WS2 clusters in reference [26]
and the dimensions should be in the limit for which quantum size effects are expected. In addition, the
absorbance became stronger with annealing temperature increased. This may be due to the size of the
sample and the IF layers increased .
Fig. 1. XRD patterns of (a) as-synthesized WS2 nanoparticles without being annealed; the samples
annealed at different temperatures: (b) 400 0C ;(c) 500 0C; (d) 600 0C; (e) 900 0C; (f) 1000 0C; (g)
1100 0C; (h) 1200 0C.
5thSASTech 2011, Khavaran Higher-education Institute, Mashhad, Iran. May 12-14.
Fig. 2. SEM images for amorphous WS2 annealed at different temperatures: (a) 400 0C, (b) 800 0C,
(c) 1000 0C, and (d) 1200 0C.
Fig. 3. TEM images for amorphous WS2 annealed at different temperatures: (a) 400 0C, (b) 500 0C,
(c) 700 0C, (d) 800 0C, (e) 1000 0C, (f) 12000C, and (g and h) electron diffraction patterns for the
samples of (d) and (e).
5thSASTech 2011, Khavaran Higher-education Institute, Mashhad, Iran. May 12-14.
Fig. 4. HRTEM images of the annealed amorphous WS2 at higher temperatures (a) annealed at 600
0
C with WS2 d(0 0 2) of 0.63 nm; (b) enlarged image of (a) in the rectangle and indicated the obvious
effects at the corner in IF structure; (c) annealed at 900 0C; (d) annealed at 900 0C with
highmagnification and with WS2 d(0 0 2) of 0.62 nm; (e) annealed at 1000 0C; (f) 1100 0C, W
nanorod with core-shell structure and the shell show W (110) lattice of about 0.21 nm.
5thSASTech 2011, Khavaran Higher-education Institute, Mashhad, Iran. May 12-14.
Fig. 5. UV-vis absorption spectroscopy of the IF nanospheres obtained at (a) 500 0C; (b) 600 0C; (c)
700 0C.
4. Conclusions
Amorphous WS2 nanospheres were obtained by a one-step oxidation-reduction reaction in solution.
Because of the rich sulfur bonds broken and the unsaturated bonds eliminated by controlling the
annealing temperatures, a series of products have been synthesized, WS2 nanospheres with diameter
of 20-70 nm, IF-nanospheres with diameter of around 20 nm and single crystal of W rods with length
from hundreds of nanometers to several microns. The IF structure with many defects at the corner and
periphery caused the break driven by the thermal kinetics. This may be the main reason for the
formation of the bended nanorods. The IF WS2 nanospheres obtained by us have a relatively strong
and broad visible absorption spectroscopy from 290 to 400 nm and a weak absorption at the region of
500 - 670 nm with a slightly blue shift. The present route is easy to control over all the reaction
processes and the simple synthesis with low-cost may offer an alternative rout to synthesize the
transition metal dichalcognides with IF structure and transition metal nanorods .
References
]5[ H.W. Kroto, J.R. Heath, S.C. OBrien, R.F. Curl, R.E. Smalley, Nature 318 (1985) 162.
[2] S. Iijima, Nature 354 (1991) 56 .
[3] R. Tenne, L. Margulis, M. Genut, G. Hodes, Nature 360 (1992) 444 .
5thSASTech 2011, Khavaran Higher-education Institute, Mashhad, Iran. May 12-14.
[4] L. Margulis, G. Salitra, R. Tenne, M. Talianker, Nature 365 (1993) 113 .
[5] M. Hershfinkel, L.A. Gheber, V. Volterra, J.L. Hutchison, L. Margulis, R. Tenne, J. Am. Chem.
Soc. 116 (1994) 1914.
[6] J. Chen, N. Kuriyama, H.T. Yuan, H.T. Takeshita, T. Sakai, J. Am. Chem. Soc. 123 (2001)
11813 .
[7] N. Imanishi, K. Kanamura, Z. Takehara, J. Electrochem. Soc. 139 (1992) 2082 .
[8] M. Thomalla, H. Tributsch, J. Phys. Chem. B 110 (2006) 12167 .
[9] F. Kopnov, G. Leitus, A. Yoffe, I. Feldman, A.M. Panich, R. Tenne, Phys. Stat. Sol. 243 (2006)
3290.
[10] L. Rapoport, N. Fleischer, R. Tenne, Adv. Mater. 15 (2003) 651 .
[11] S.E. Skrabalak, K.S. Suslick, J. Am. Chem. Soc. 127 (2005) 9990 .
[12] E. Aharon, A. Albo, M. Kalina, L. Frey Gitti, Adv. Funct. Mater. 16 (2006) 980.
[13] M. Nath, A. Govindaraj, C.N.R. Rao, Adv. Mater. 13 (2001) 283 .
[14] C.M. Zelenski, P.K. Dorhout, J. Am. Chem. Soc. 120 (1998) 743 .
[15] L. Rapoport, Y. Bibik, Y. Feldman, M. Homyonfer, S.R. Cohen, R. Tenne, Nature 387 (1997)
791 .
[16] M. Jose-Yacaman, H. Lorez, P. Santiago, D.H. Galvan, I.L. Garzon, A. Reyes, Appl. Phys. Lett.
69 (1996) 1065.
[17] P.A. Parilla, A.C. Dillon, K.M. Jones, G. Riker, D.L. Schulz, D.S. Ginley, M.J. Heben, Nature
397 (1999) 114.
[18] Y. Tian, Y. He, Y.F. Zhu, Chem. Lett. 8 (2003) 268 .
[19] D. Duphil, S. Bastide, C. Levy-Clment, J. Mater. Chem. 12 (2002) 2430 .
[20] I. Uzcanga, I. Bezverkhyy, P. Afanasiev, C. Scott, M. Vrinat, Chem. Mater. 17 (2005) 3575 .
[21] L.X. Chang, H.B. Yang, J.X. Li, W.Y. Fu, Y.H. Du, K. Du, Q.Y. Yu, J. Xu, M.H. Li,
Nanotechnology 17 (2006) 3827.
[22] X.L. Li, J.P. Ge, Y.D. Li, Chem. Eur. J. 10 (2004) 6163 .
[23] J. Etzkorn, A.H. Therese, F. Rocker, N. Zink, U. Kolb, W. Tremel, Adv. Mater. 17 (2005) 2372.
[24] Q. Li, J.T. Newberg, E.C. Walter, J.C. Hemminger, R.M. Penner, Nano Lett. 4 (2004) 277 .
[25] H.B. Yang, S.K. Liu, J.X. Li, M.H. Li, G. Peng, G.T. Zou, Nanotechnology 17 (2006) 1512.
[26] J.P. Wilcoxon, P.P. Newcomer, G.A. Samara, J. Appl. Phys. 81 (1997) 7934.
Vous aimerez peut-être aussi
- The Subtle Art of Not Giving a F*ck: A Counterintuitive Approach to Living a Good LifeD'EverandThe Subtle Art of Not Giving a F*ck: A Counterintuitive Approach to Living a Good LifeÉvaluation : 4 sur 5 étoiles4/5 (5795)
- The Gifts of Imperfection: Let Go of Who You Think You're Supposed to Be and Embrace Who You AreD'EverandThe Gifts of Imperfection: Let Go of Who You Think You're Supposed to Be and Embrace Who You AreÉvaluation : 4 sur 5 étoiles4/5 (1091)
- Never Split the Difference: Negotiating As If Your Life Depended On ItD'EverandNever Split the Difference: Negotiating As If Your Life Depended On ItÉvaluation : 4.5 sur 5 étoiles4.5/5 (838)
- Hidden Figures: The American Dream and the Untold Story of the Black Women Mathematicians Who Helped Win the Space RaceD'EverandHidden Figures: The American Dream and the Untold Story of the Black Women Mathematicians Who Helped Win the Space RaceÉvaluation : 4 sur 5 étoiles4/5 (895)
- Grit: The Power of Passion and PerseveranceD'EverandGrit: The Power of Passion and PerseveranceÉvaluation : 4 sur 5 étoiles4/5 (588)
- Shoe Dog: A Memoir by the Creator of NikeD'EverandShoe Dog: A Memoir by the Creator of NikeÉvaluation : 4.5 sur 5 étoiles4.5/5 (537)
- The Hard Thing About Hard Things: Building a Business When There Are No Easy AnswersD'EverandThe Hard Thing About Hard Things: Building a Business When There Are No Easy AnswersÉvaluation : 4.5 sur 5 étoiles4.5/5 (345)
- Elon Musk: Tesla, SpaceX, and the Quest for a Fantastic FutureD'EverandElon Musk: Tesla, SpaceX, and the Quest for a Fantastic FutureÉvaluation : 4.5 sur 5 étoiles4.5/5 (474)
- Her Body and Other Parties: StoriesD'EverandHer Body and Other Parties: StoriesÉvaluation : 4 sur 5 étoiles4/5 (821)
- The Emperor of All Maladies: A Biography of CancerD'EverandThe Emperor of All Maladies: A Biography of CancerÉvaluation : 4.5 sur 5 étoiles4.5/5 (271)
- The Sympathizer: A Novel (Pulitzer Prize for Fiction)D'EverandThe Sympathizer: A Novel (Pulitzer Prize for Fiction)Évaluation : 4.5 sur 5 étoiles4.5/5 (121)
- The Little Book of Hygge: Danish Secrets to Happy LivingD'EverandThe Little Book of Hygge: Danish Secrets to Happy LivingÉvaluation : 3.5 sur 5 étoiles3.5/5 (400)
- The World Is Flat 3.0: A Brief History of the Twenty-first CenturyD'EverandThe World Is Flat 3.0: A Brief History of the Twenty-first CenturyÉvaluation : 3.5 sur 5 étoiles3.5/5 (2259)
- LV07 KDocument11 pagesLV07 KSalman Shah83% (6)
- The Yellow House: A Memoir (2019 National Book Award Winner)D'EverandThe Yellow House: A Memoir (2019 National Book Award Winner)Évaluation : 4 sur 5 étoiles4/5 (98)
- Devil in the Grove: Thurgood Marshall, the Groveland Boys, and the Dawn of a New AmericaD'EverandDevil in the Grove: Thurgood Marshall, the Groveland Boys, and the Dawn of a New AmericaÉvaluation : 4.5 sur 5 étoiles4.5/5 (266)
- A Heartbreaking Work Of Staggering Genius: A Memoir Based on a True StoryD'EverandA Heartbreaking Work Of Staggering Genius: A Memoir Based on a True StoryÉvaluation : 3.5 sur 5 étoiles3.5/5 (231)
- Team of Rivals: The Political Genius of Abraham LincolnD'EverandTeam of Rivals: The Political Genius of Abraham LincolnÉvaluation : 4.5 sur 5 étoiles4.5/5 (234)
- On Fire: The (Burning) Case for a Green New DealD'EverandOn Fire: The (Burning) Case for a Green New DealÉvaluation : 4 sur 5 étoiles4/5 (74)
- The Unwinding: An Inner History of the New AmericaD'EverandThe Unwinding: An Inner History of the New AmericaÉvaluation : 4 sur 5 étoiles4/5 (45)
- 53 MugeshDocument5 pages53 MugeshAnonymous t7MdBjnOPas encore d'évaluation
- Tribology of Tungsten Disulfide Films in Humid Environments: The Role of A Tailored Metal-Matrix Composite SubstrateDocument11 pagesTribology of Tungsten Disulfide Films in Humid Environments: The Role of A Tailored Metal-Matrix Composite SubstrateAnonymous t7MdBjnOPas encore d'évaluation
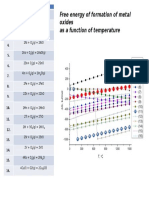
- Free Energy of Formation of Metal Oxides As A Function of TemperatureDocument1 pageFree Energy of Formation of Metal Oxides As A Function of TemperatureAnonymous t7MdBjnOPas encore d'évaluation
- Spectrophotometric Determination of The Copper IonDocument2 pagesSpectrophotometric Determination of The Copper IonAnonymous t7MdBjnOPas encore d'évaluation
- Structures of Solids & X - Ray DiffractionDocument14 pagesStructures of Solids & X - Ray DiffractionAnonymous t7MdBjnOPas encore d'évaluation
- Flexible Conductive Graphene Paper Obtained PDFDocument36 pagesFlexible Conductive Graphene Paper Obtained PDFAnonymous t7MdBjnOPas encore d'évaluation
- CNC RoboticsDocument17 pagesCNC RoboticsKunal DuttPas encore d'évaluation
- Reference Manual: Buildings and Infrastructure Protection SeriesDocument514 pagesReference Manual: Buildings and Infrastructure Protection SeriesmydearteacherPas encore d'évaluation
- Roundel 1961-06 Vol 13 No 5Document36 pagesRoundel 1961-06 Vol 13 No 5TatePas encore d'évaluation
- In Search of The Indo Europeans J P Mallory TextDocument289 pagesIn Search of The Indo Europeans J P Mallory TextHISTORY OF MUSICPas encore d'évaluation
- Allen: Pre Nurture & Career Foundation DivisionDocument7 pagesAllen: Pre Nurture & Career Foundation DivisionSiddharth Singh100% (1)
- Blue A Ks2 Test3Document6 pagesBlue A Ks2 Test3Mateo García-PumarinoPas encore d'évaluation
- Grade 4 Social Sciences-Geography Term 2 Lesson PlansDocument9 pagesGrade 4 Social Sciences-Geography Term 2 Lesson Plansgobedimalapile02Pas encore d'évaluation
- Dist Census Book Latur PDFDocument770 pagesDist Census Book Latur PDFMP100% (1)
- Final Differential Equations (PDF)Document88 pagesFinal Differential Equations (PDF)Tina ShahPas encore d'évaluation
- Data Sheet 3VA2225-5HL32-0AA0: ModelDocument7 pagesData Sheet 3VA2225-5HL32-0AA0: ModelJENNYPas encore d'évaluation
- DAA NotesDocument97 pagesDAA Notesanish.t.pPas encore d'évaluation
- Slide Topic 2 - PR TheoriesDocument26 pagesSlide Topic 2 - PR TheoriesJeneesh RajendranPas encore d'évaluation
- ContemporaryDocument2 pagesContemporaryAlbert PaggaoPas encore d'évaluation
- GALCIT 6-Inch Shock Tube: Bryan SchmidtDocument14 pagesGALCIT 6-Inch Shock Tube: Bryan SchmidtNitesh Kumar PatelPas encore d'évaluation
- Claves Eset 5Document3 pagesClaves Eset 5Orquesta Sensacion CaribePas encore d'évaluation
- Consultant DirectoryDocument20 pagesConsultant DirectoryPreservation MassachusettsPas encore d'évaluation
- Family Tree Template With DetailsDocument8 pagesFamily Tree Template With DetailsJude WellsPas encore d'évaluation
- Manual SomachineDocument228 pagesManual SomachineMauricio NaranjoPas encore d'évaluation
- Business Statistics I: Hypothesis TestingDocument58 pagesBusiness Statistics I: Hypothesis TestingJamesPas encore d'évaluation
- Cambridge O Level: PHYSICS 5054/42Document16 pagesCambridge O Level: PHYSICS 5054/42Lapu LapuPas encore d'évaluation
- Government Polytechnic, Pune: ET2107 - NODocument8 pagesGovernment Polytechnic, Pune: ET2107 - NOG012 Bhise AniketPas encore d'évaluation
- Dwnload Full Business Research Methods 9th Edition Zikmund Solutions Manual PDFDocument35 pagesDwnload Full Business Research Methods 9th Edition Zikmund Solutions Manual PDFerichuel33a100% (14)
- Probability DistributionsDocument167 pagesProbability DistributionslihaiyangPas encore d'évaluation
- Pte Apeuni 20240408 Global Monthly en Kd54Document321 pagesPte Apeuni 20240408 Global Monthly en Kd54Boyes WorldPas encore d'évaluation
- Transient Thermal Analysis On Re-Entry Vehicle Nose Cone With Tps MaterialsDocument10 pagesTransient Thermal Analysis On Re-Entry Vehicle Nose Cone With Tps Materialssiva shankarPas encore d'évaluation
- Variables That Affect Teachers' Attitudes Towards Disability and Inclusive Education in Mumbai, IndiaDocument12 pagesVariables That Affect Teachers' Attitudes Towards Disability and Inclusive Education in Mumbai, IndiaSumaiya HossainPas encore d'évaluation
- Standard Support: Figure 1: Pressing F1 Redirects User To A Bing SearchDocument4 pagesStandard Support: Figure 1: Pressing F1 Redirects User To A Bing SearchDbluepengLumPas encore d'évaluation
- TI 20220909 SG320HX and SG350HX Short-Circuit Current V1 enDocument5 pagesTI 20220909 SG320HX and SG350HX Short-Circuit Current V1 en2D EngenhariaPas encore d'évaluation
- Non-Parametric TestsDocument1 pageNon-Parametric TestssdvdsvPas encore d'évaluation