Académique Documents
Professionnel Documents
Culture Documents
Pka Chart
Transféré par
SayTitre original
Copyright
Formats disponibles
Partager ce document
Partager ou intégrer le document
Avez-vous trouvé ce document utile ?
Ce contenu est-il inapproprié ?
Signaler ce documentDroits d'auteur :
Formats disponibles
Pka Chart
Transféré par
SayDroits d'auteur :
Formats disponibles
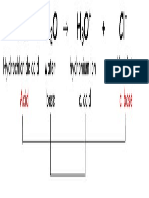
pKa Chart
sulfuric acid
H2SO4
conjugate acid
-10
HSO4
HI
-9
HBr
Br
-8
hydroiodic acid
hydrobromic acid
conjugate base
protonated
ketone
hydrochloric acid
HCl
-7.3
weakest bases
strongest acids
conjugate acid
nitric acid
HNO3
tosic acid
TsOH
-0.6
3.2
hydrogen nitride
HN3
N3
4.7
O
O
4.8
H
H
carbocations
protonated
alcohol
TsO-
-3.6
-1.3
NO3
HF
carboxylic acids
protonated
ether
conjugate base
hydrofluoric acid
-7
Cl
-3
-2.4
protonated
pyridine
carbonic acid
NH
H2CO3
HCO3
5.2
6.37
(bicarbonate)
hydronium ion
O
H
hydrogen sulfide
H
-1.7
H2S
SH
conjugate acid
O
conjugate base
O
water
1,3-diketones
hydrogen cyanide
conjugate base
O
primary alcohols
9.1
15.7
16
(alkoxides)
9.4
tertiary alcohols
(hydroxide)
(cyanide)
ammonium ions
conjugate acid
(tert-butoxide)
O
OH
ketones
10
phenols
18
18-21
(enolates)
O
bicarbonate
-2
HCO3
10.25
CO3
esters
25
O
(ester enolates)
thiols
10.5
alkynes
26
(acetylide anions)
protonated
amines
H
H3C
H
H3C
10.6
hydrogen
H
one way only
(hydride)
35
H
H
15
(aromatic)
O
amides
NH2
15
NH
ammonia/amines
N
R
H
(amide bases)
alkanes
36
~60
H
one way only
MgBr
(Grignards/ organolithium reagents)
strongest bases
weakest acids
cyclopentadiene
Vous aimerez peut-être aussi
- Common Chemical Formula ListDocument3 pagesCommon Chemical Formula Listaran9280% (5)
- Ch18Acid Base (A)Document45 pagesCh18Acid Base (A)Sam H. SalehPas encore d'évaluation
- Acid and Base Strengths: A B B CDocument1 pageAcid and Base Strengths: A B B CVIKAS GARGPas encore d'évaluation
- Acizi Si BazeDocument4 pagesAcizi Si BazeKristanna123Pas encore d'évaluation
- Weak Acids, Weak Bases and BuffersDocument23 pagesWeak Acids, Weak Bases and BuffersCansın UğurPas encore d'évaluation
- Pka TableDocument1 pagePka TableEman BadryPas encore d'évaluation
- Base Ionization Constants Substance Formula KDocument5 pagesBase Ionization Constants Substance Formula KKhelly Shan C. Sta. RitaPas encore d'évaluation
- Acids&BasesDocument3 pagesAcids&BasesJosh CatolinPas encore d'évaluation
- Acidity Constants Data Part 1Document2 pagesAcidity Constants Data Part 1AcePas encore d'évaluation
- CHPT 16Document12 pagesCHPT 16Duaa RajaPas encore d'évaluation
- Table of Pka ValuesDocument3 pagesTable of Pka ValuesjVPas encore d'évaluation
- The Table of Acid and BaseDocument3 pagesThe Table of Acid and BaseDimas MuhamadPas encore d'évaluation
- Notes Solutions Chapter 07Document15 pagesNotes Solutions Chapter 07Syllvia SunnivaPas encore d'évaluation
- Constantes de Equilibrioacido-Base (25 C) : Acid Formula N Pk1 Pk2 Pk3 Pk4Document2 pagesConstantes de Equilibrioacido-Base (25 C) : Acid Formula N Pk1 Pk2 Pk3 Pk4Naty AvalosPas encore d'évaluation
- 51 HMW Solutions Ch14Document43 pages51 HMW Solutions Ch14Cbn NoonPas encore d'évaluation
- Acids and Bases NotesDocument17 pagesAcids and Bases NotesNap DoPas encore d'évaluation
- Video NotesDocument45 pagesVideo Notesjim tannerPas encore d'évaluation
- Acid BasesDocument2 pagesAcid BasesseruPas encore d'évaluation
- An Incomplete Polyatomic Ion ChartDocument1 pageAn Incomplete Polyatomic Ion ChartGAT TutoringPas encore d'évaluation
- Acid-Base Equilibrium: See Aqueousions in Chemistry 1110 Online Notes For Review of Acid-Base Fundamentals!Document31 pagesAcid-Base Equilibrium: See Aqueousions in Chemistry 1110 Online Notes For Review of Acid-Base Fundamentals!Janna EchavezPas encore d'évaluation
- Lecture 5 Acids and BasesDocument39 pagesLecture 5 Acids and BasesAllen SiaPas encore d'évaluation
- 3.ii. BASES AND ACIDS OF CHOICEDocument110 pages3.ii. BASES AND ACIDS OF CHOICEKeith OmwoyoPas encore d'évaluation
- Conjugate Acid and Base PairsDocument1 pageConjugate Acid and Base Pairsfelisita wisangPas encore d'évaluation
- HSC Chemistry Lesson Plan 16Document8 pagesHSC Chemistry Lesson Plan 16Ali HaidarPas encore d'évaluation
- Chapter 3 Acid - BaseDocument98 pagesChapter 3 Acid - BaseKhoa Nguyen Viet DangPas encore d'évaluation
- Phenol SDocument32 pagesPhenol SBangaloredPas encore d'évaluation
- Acid and BaseDocument2 pagesAcid and BaseMaraPas encore d'évaluation
- Ch14 Study QuestionsDocument3 pagesCh14 Study QuestionsКанат ТютеновPas encore d'évaluation
- PPP1A 8.1 Arrhenius and NamingDocument13 pagesPPP1A 8.1 Arrhenius and NamingRichard LindemannPas encore d'évaluation
- Chapter 3 Acid - BaseDocument98 pagesChapter 3 Acid - BasePHƯƠNG ĐẶNG YẾNPas encore d'évaluation
- Chapter 3 Acid - BaseDocument98 pagesChapter 3 Acid - BaseNhan PhướcPas encore d'évaluation
- Asam LemahDocument2 pagesAsam LemahAddeSRPas encore d'évaluation
- Solutions 222Document8 pagesSolutions 222estellasr00Pas encore d'évaluation
- Acid Base Notes (Use For Chem 123)Document16 pagesAcid Base Notes (Use For Chem 123)Nate JamesPas encore d'évaluation
- Acid Equilibrium and PH: Søren SørensenDocument16 pagesAcid Equilibrium and PH: Søren Sørensenmonster40lbsPas encore d'évaluation
- New 104 Nucleophilic SubstDocument11 pagesNew 104 Nucleophilic SubstEva PannemanPas encore d'évaluation
- Table of Ka and KBDocument1 pageTable of Ka and KBkarimhandoyoPas encore d'évaluation
- Chapter 3Document124 pagesChapter 3Fariz SharudinPas encore d'évaluation
- Lecture# PhenolDocument16 pagesLecture# PhenolRao Wazim AkramPas encore d'évaluation
- PhenolsDocument34 pagesPhenolsLoran Prelya TengayPas encore d'évaluation
- Natural Produc Chemistry - Alkaloid Biosynthesis - Nila HudaDocument23 pagesNatural Produc Chemistry - Alkaloid Biosynthesis - Nila HudaNilaHudaBaqirPas encore d'évaluation
- Asam BasaDocument13 pagesAsam Basaaliefyan4769Pas encore d'évaluation
- Name: KEY Nomenclature - Covalent (Molecular) Compounds Part A: Name The Following Covalent Compounds. Part B: Write The Chemical Formula For Each of The Following CompoundsDocument2 pagesName: KEY Nomenclature - Covalent (Molecular) Compounds Part A: Name The Following Covalent Compounds. Part B: Write The Chemical Formula For Each of The Following CompoundsTrung LuongPas encore d'évaluation
- 1288 PhenolsDocument29 pages1288 PhenolsX ThrxPas encore d'évaluation
- Activity 2.1 - Acids and Bases NMNMDocument3 pagesActivity 2.1 - Acids and Bases NMNMClarise CanAPas encore d'évaluation
- Chapter 6-Acid and Base PDFDocument47 pagesChapter 6-Acid and Base PDFWhafimsPas encore d'évaluation
- Chapter 3 Acid - BaseDocument96 pagesChapter 3 Acid - BaseAnh NhamPas encore d'évaluation
- Chapter 18 Silberberg AnswersDocument51 pagesChapter 18 Silberberg AnswersKevin DashPas encore d'évaluation
- EMAN YASSIN - Chemistry 12 Unit 4 PacketDocument11 pagesEMAN YASSIN - Chemistry 12 Unit 4 PacketSherine GamalPas encore d'évaluation
- Physical Chemistry QuestionsDocument2 pagesPhysical Chemistry QuestionsClinton Softleigh JrPas encore d'évaluation
- 7.0 Ionic Equilibria (Students)Document187 pages7.0 Ionic Equilibria (Students)Supia Nazma100% (1)
- Acids and Bases Note SapDocument30 pagesAcids and Bases Note SapNabilah MustafaPas encore d'évaluation
- Acids Bases SaltsDocument11 pagesAcids Bases Saltsabiodun olaokePas encore d'évaluation
- Acids BasesDocument30 pagesAcids BasesHaniel GalzotePas encore d'évaluation
- Practice Makes Perfect in Chemistry: Acids, Bases, and SaltsD'EverandPractice Makes Perfect in Chemistry: Acids, Bases, and SaltsPas encore d'évaluation
- Recent Developments in the Chemistry of Natural Phenolic Compounds: Proceedings of the Plant Phenolics Group SymposiumD'EverandRecent Developments in the Chemistry of Natural Phenolic Compounds: Proceedings of the Plant Phenolics Group SymposiumW. D. OllisPas encore d'évaluation
- Monohydric Alcohols Their Ethers and Esters Sulphur Analogues Nitrogen Derivatives Organometallic Compounds: A Modern Comprehensive TreatiseD'EverandMonohydric Alcohols Their Ethers and Esters Sulphur Analogues Nitrogen Derivatives Organometallic Compounds: A Modern Comprehensive TreatisePas encore d'évaluation
- Aliphatic Compounds: A Modern Comprehensive TreatiseD'EverandAliphatic Compounds: A Modern Comprehensive TreatisePas encore d'évaluation
- "And, When You Want Something, ALL The: UniverseDocument1 page"And, When You Want Something, ALL The: UniverseSayPas encore d'évaluation
- Gases AnswersDocument8 pagesGases AnswersSayPas encore d'évaluation
- Key 3Document7 pagesKey 3SayPas encore d'évaluation
- Social Science Nmat ReviewerDocument13 pagesSocial Science Nmat ReviewerSay77% (13)