Académique Documents
Professionnel Documents
Culture Documents
Department of Instrumentation Engineering, Faculty of Engineering, Keio University, 3-14-1 Hiyoshi, Kohoku, Yokohama 223, Japan
Transféré par
Vu Anh KienTitre original
Copyright
Formats disponibles
Partager ce document
Partager ou intégrer le document
Avez-vous trouvé ce document utile ?
Ce contenu est-il inapproprié ?
Signaler ce documentDroits d'auteur :
Formats disponibles
Department of Instrumentation Engineering, Faculty of Engineering, Keio University, 3-14-1 Hiyoshi, Kohoku, Yokohama 223, Japan
Transféré par
Vu Anh KienDroits d'auteur :
Formats disponibles
Volume 96A, number 2
PHYSICS LETTERS
13 June 1983
COLOURATION OF RHODAMINE B IN PMMA BY AQUEOUS HCI SOLUTION
AND IN THE COPOLYMER OF METHYL METHACRYLATE WITH METHACRYLIC ACID
Jun'ichiro MUTO and Fumito HIGUCHI
Department of Instrumentation Engineering, Faculty of Engineering, Keio University,
3-14-1 Hiyoshi, Kohoku, Yokohama 223, Japan
Received 1 July 1982
A rhodamine B doped PMMAsample is made by the solution polymerization method. It is found that the reddish
colottr of rhodamine B molecules reduces greatly during polymerization and that the once reduced colour of rhodamine
B recovers by immersion of the sample in aqueous HCI solution. Discolouration of rhodamine B is diminished in the
copolymer of methyl methacrylate with methacrylie acid.
The use of a solid matrix for dye lasers obviates
many of the problems associated with static or flowing
solution dye systems, e.g., convective schlieren, evaporation, flow fluctuation, solvent or dye poisoning,
system ruggedness and maintenance. Polymethyl
methacrylate (PMMA) is a highly transparent plastic
and is used as a solid dye laser matrix and optical fibre.
Rhodamine B(RhB) and rhodamine 6G(Rh6G), most
well-known laser dyestuffs, have both been succeeded
to lase in PMMA [ 1].
Methods of producing dye impregnated plastics
may be characterized as either bulk or solution polymerization methods whereby laser dyes are added to
a solution of catalysts and monomers before polymerization, or at some point in time before the final polymerization of the monomers into the solid matrix.
The dye may also be doped in the plastic by a concentration-difference diffusion method in which a plastic
matrix is immersed in a solution of dissolved organic
lasing dye and a solvent at a suitable dye concentration,
pH and temperature.
The polarity of RhB is reduced by the coordination
of methacrylic acid(MA), oleic acid or stearic acid, and
then the solubility of RhB in methyl methacrylate (MMA)
is increased by the addition of these acids. Actually
it became possible to dope RhB in the copolymer of
MMA with MA at sufficient concentration for lasing
performance [2]. A decrease in photo and thermal
0 031-9163/83/0000-0000]$ 03.00 1983 North-Holland
bleaching of Rh6G was realized in P(MMA + MA) [3].
Corresponding photo and thermal deterdation results
of RhB in P(MMA + MA) have not yet been obtained.
In view of the above situations, we have fabricated
a RhB doped PMMA sample by the solution polymeriza.
tion method and find that the reddish colour of RhB
molecules decreases greatly during the polymerization
process and that the once reduced colour of RhB/PMMA
recovers by immersion of the sample in aqueous hydrochloric acid solution. RhB is observed to be less
discoloured in P(MMA + MA).
RhB(Eastman Kodak, laser grade) was dissolved in
vacuum distilled MMA solution. Polymerization catalyst was azobisisobutyronytrile(AIBN). 1 mg of AIBN
was used for 1 ml solution. To obtain a RhB doped
P(MMA + MA) sample, vacuum distilled MA was added to the solution. The solution was bubbled with nitrogen gas and kept in a glass container. Polymerization
was performed in a water bath at 40C for 4 days. The
samples obtained were cut and finally polished with 0.3
m alumina.
The samples were 30 mm in diameter and 5 mm
thick. The RhB concentration of the sample is typicall y 7 X 10 -5 M.
Absorption spectra of RhB/PMMA immersed in
7.2 M aqueous HC1 solution at 40C for different immersion times are shown in fig. 1. Absorption coefficients of the sample are found to increase with in101
Volume 96A, number 2
PHYSICS LETTERS
1
HCt
7214
~E
1c]
1:
740 h
2:
265h
3:
~,3h
4:
g,
g_ 5
2,
o
0 ~,
-r
400
--
450
500
Wavelength
550
600
( nrn )
Fig. 1. Absorption spectra of RhB soaked in 7.2 M aqueous
HCI solution at 40C for different immersion times between
0 and 74.0 h.
creasing immersion time in the measured wavelength
region between 600 and 400 nm. Similar spectral behaviour is seen for different immersion temperatures
between 5 and 56C and HC1 concentrations between
0 and 7.2 M. It may be remarked that the maximum
molar absorption coefficient of RhB in ethanol is
about 2.5 X 105 M-1 cm -1, whereas the corresponding
value in PMMA, as seen in fig. 1, is very low, i.e. 0.3
104 M-1 cm -1 . RhB is exposed to HC1 vapour at
room temperature, colouration of the sample is clearly
recognized.
Rhodamine dyes that carry the COOH group become colourless lactonic state in non-polar solvents,
because the n-electron system of the dye chromophore
is interrupted (see fig. 2). Actually, lactonic RhB is
formed in benzoic solution in a reversible reaction [4].
In polar solvents, such as alcohol and water, the lactonic RhB has a tendency to open the closed ring
with a resulting separation of charges, forming a coloured zwitterion.
(C 2 H 5 ) 2 N , ~ O , , ~ . ~ I ( C 2 1 . . 1 5 ) 2
[~
[A]
13 June 1983
Our PMMA sample is in an amorphous state and
shows atactic form, and owing to steric hindrances,
the polymer chain cannot take all configurations allowed by free rotation about the chain molecules. It
is observed that the dipole moment of the long-chain
PMMA molecule per monomer unit is smaller than that
of MMA [5]. Presumably, the polarity of PMMA
molecules surrounding RhB is lessened as polymerization proceeds and then the dye molecules become
colourless lactone. The Rh6G/PMMA sample, made
by the solution polymerization method, is observed
to reduce its colour scarcely during polymerization,
which is reasonable since Rh6G has esterified the
COOH group and the formation of a lactonic state is
prevented. By the introduction of aqueous HC1 solution into PMMA, water molecules form H-bonds
between HC1 and the segments of the polymeric
material and causes the loosening of PMMA material
[6].
Fig. 3. shows a change in peak molar absorption
coefficient of RhB/PMMA, Act, as a function of the
immersion time between 5 and 56C and at a HC1 concentration of 7.2 M. Here Act = (peak molar absorption coefficient of the sample before immersion)
- (that after t hours immersion). In fig. 3, Act seems
to increase as the square root of the immersion time.
In fig. 4, Act versus immersion time of RhB/PMMA
soaked in 7.2 M solution at temperatures between 5 and
56C is presented, in which Act increases approximately
as the square root of the immersion time at smaller immersion times and decreases, at larger immersion times.
Similar behaviours are seen for RhB/PMMA soaked in
the solution of HCI concentration below 7.2 M.
Taking into consideration the reversible reaction
of lactonic RhB in fig. 2, and also the non-polar nature
of PMMA mentioned above in addition to the chemically active behaviour of the hydrogen ion, we may sup-
CI-
f
%/',,c//-,,f
COOH
COOH
l..~. 0
[BJ
Fig. 2. Structural formulae of RhB. [A] shows salt form and [B], formation of lactonic RhB in a reversible way.
102
Volume 96A, number 2
PHYSICS LETTERS
the sample is observed for RhB/PMMA soaked in pure
aqueous solution (see fig. 4). Perhaps,water has also
some activity, small as it is, to open the lactonic ring
of the RhB molecule.
When diffusion of a solvent occurs in a polymer
slab under isothermal conditions, and the slab contains
a uniform initial distribution of a solvent, Fick's law
of diffusion for small diffusion times becomes [8]
xl~ 4
56 ,.C~
//i;30C
Immersion Ti
}1/2
(1)
Wt/W.o = 2 (Dtprd2) 1/2 ,
13 June 1983
(hI/2)
Fig. 3. Change in peak molar absorption coefficient of RhB/
PMMA, AS, versus immersion time at different solution temperatures between 5 and 56C. HC1 concentration: 7.2 M.
pose that by immersion of the RhB/PMMA sample in
aqueous HCI solution, the H + ion diffuses into the
sample and effectively works to open the closed ring
of lactonic RhB resulting in the recovery of the RhB
colour in the sample.
It is said that water in PMMA acts as a plasticizer
for the cooperative motion of the polymer backbone
and appears to exist as isolated molecules or small
clusters [7]. A slight increase in the absorption of
where Wt denotes the total amount of diffusing substance which has entered into the sheet at time t; W=,
the corresponding quantity after infinite time; d, onehalf of the slab thickness, and D is the diffusion coefficient.
Our results in figs. 3 and 4, that Act is proportional
to the square root of the immersion time at small immersion times, may be reasonable provided that the
H ion enters into the sample followed by Fick's law
of diffusion and that Aa increases linearly with the
amount of diffusion substance, i.e. H ion in the solution. The decrease of Act in fig. 4 at larger immersion
times may be due to the destruction of RhB by HC1.
Further detailed studies concerning the diffusion of
~10~
10
10~
u.
c
o
k~
o.
1[
2:
2.4 M
~
o
(Immersion Timel/e
10
(h1/~1
Fig. 4. Change in peak molar absorption coefficient of RhB/
PMMA, AS, versus immersion time at HC1 concentration
between 0 and 7.2 M. Solution temperature: 40C.
~
MA Concentrotion
'
10
(% )
15
Fig. 5. Maximum molar absorption coefficient of RhB/P(MMA
+ MA) versus MA concentration.
103
Volume 96A, number 2
PHYSICS LETTERS
H 2 0 - H C 1 into PMMA will be needed to understand
fully the colouration o f the sample.
Now, we examine the absorption behaviour of
RhB in P(MMA + MA). Discolouration o f RhB in
P(MMA + MA) tends to become less with increasing
MA concentration (see fig. 5). In our experiments,
RhB concentration is below the maximum solubility
in MMA solution, and the addition of MA to MMA
does not seem to improve the solubility of RhB
molecules in the solution. MA is different from MMA
in that one of the COOCH 3 groups is replaced b y
COOH, i.e., non-esterified carboxyl group. The H
ion in the' COOH group of MA is thought to be effective to open the mentioned colourless lactonic RhB
in the polymer matrix.
Hydrolysis of PMMA in sulphuric acid gave a polymer containing 36% MA [9]. Colouration of RhB/
PMMA is expected when the sample is immersed in
H2SO 4 solution. Unfortunately, RhB in PMMA is
found destroyed by the diffusion of H2SO 4 into the
RhB/PMMA sample.
104
13 June 1983
The authors wish to thank Mr.S. Iwata for his assistance in the present work.
References
[1 ] O.G. Peterson and B.B. Snavely, Appl. Phys. Lett. 12
(1968) 232.
[2] K. It0h, H. Kusakawa and T. Takahashi, Oyo Butsuri 39
(1970) 1067 (in Japanese).
[3] F. Higuchi and J. Muto, Phys. Lett. 81A (1981) 95; 86A
(1981) 51.
[4] R.W. Ramette and E.B. Sandel, J. Am. Chem. Soc. 78
(1956) 4872.
[5 ] W.R. Wringham and J.V. Pawkins, Polymer handbook,
Vol. 4, eds. J. Braundrup and E.H. Immergut (Interscience, New York, 1967) p. 331.
[6] V.N. Kestl'man, I.A. Kershunov, N.F. Novotorov, R.A.
Lazareva and L.N. Magazinova, Sint. Fiz. Khim. Polim.
11 (1973)57 [Chem. Abstr. 80 (1974) 3938z].
[7] A.S, Gilbert, R.A. Pethric and D.W. Phillips, J. Appl.
Polym. Sci. 21 (1977) 319.
[8] J. Crank, The mathematics of diffusion (Oxford Univ.
Press, London, 1967) ch. 4.
[9] A.E. Kurikova and D.A. Aranovich, Tr. Khim. Khim.
Tekhnol. 1 (1973) 102 [Chem. Abstr. 80 (1974)
83852e].
Vous aimerez peut-être aussi
- New MTBE Production DesignDocument13 pagesNew MTBE Production DesignImad Sami100% (2)
- Autocatalytic Oxidation of Ethers With Sodium BromateDocument6 pagesAutocatalytic Oxidation of Ethers With Sodium Bromatebebabebic45Pas encore d'évaluation
- Refresh Refresh (Dynamic - Bypass - Reload) Click Here If You Are Not Automatically Redirected. For Assistance, Contact Your Network Support Team.Document9 pagesRefresh Refresh (Dynamic - Bypass - Reload) Click Here If You Are Not Automatically Redirected. For Assistance, Contact Your Network Support Team.Kelvin LimPas encore d'évaluation
- New Strategies For The Hofmann ReactionDocument7 pagesNew Strategies For The Hofmann ReactionCasper JorckPas encore d'évaluation
- (20672446 - Acta Chemica Iasi) Aldol Condensation Reactions Effectively Catalysed by Lewis AcidDocument10 pages(20672446 - Acta Chemica Iasi) Aldol Condensation Reactions Effectively Catalysed by Lewis AcidNurannisa NisaPas encore d'évaluation
- Determining The Dissociation Constant of A Weak Acid Using The SpectrophotometerDocument23 pagesDetermining The Dissociation Constant of A Weak Acid Using The SpectrophotometerLaila Faeizah100% (7)
- Kinetic, Thermodynamic and Equilibrium Studies On Uptake of Rhodamine B Onto ZNCL Activated Low Cost CarbonDocument9 pagesKinetic, Thermodynamic and Equilibrium Studies On Uptake of Rhodamine B Onto ZNCL Activated Low Cost CarbonSeptian Perwira YudhaPas encore d'évaluation
- Communications To The EditorDocument3 pagesCommunications To The EditorSowo SesPas encore d'évaluation
- Methyl RedDocument13 pagesMethyl RedAirthSpark3000100% (2)
- Surface Charge Properties of Red Mud Particles Generated From Chinese Diaspore BauxiteDocument5 pagesSurface Charge Properties of Red Mud Particles Generated From Chinese Diaspore BauxiteArvind KumarPas encore d'évaluation
- 1955 - Boyes - Methods For The AnalysisDocument8 pages1955 - Boyes - Methods For The Analysisjlcheefei9258Pas encore d'évaluation
- Optimization in The Absorption and DesorptionDocument20 pagesOptimization in The Absorption and DesorptionShamsMohdPas encore d'évaluation
- 4 J Chem Res November 2008Document6 pages4 J Chem Res November 2008Arjun paudelPas encore d'évaluation
- Ashworth1992 PDFDocument6 pagesAshworth1992 PDFEka HerlinaPas encore d'évaluation
- 1) Potassium Carbonate Phase in Estuarine Sediments: Composition, Formation and Chemical ReactivityDocument8 pages1) Potassium Carbonate Phase in Estuarine Sediments: Composition, Formation and Chemical Reactivitymark_idananPas encore d'évaluation
- Snamprogetti New MTBE Production Design PDFDocument13 pagesSnamprogetti New MTBE Production Design PDFViệt HàPas encore d'évaluation
- Polylactic Acid Synthesis With PolymerizationDocument9 pagesPolylactic Acid Synthesis With PolymerizationnierzaPas encore d'évaluation
- Kinetics of Amines With Primary and Secondary in Aqueous SolutionsDocument7 pagesKinetics of Amines With Primary and Secondary in Aqueous SolutionsJack SutherlandPas encore d'évaluation
- Relationship Between HBP Structure and Chrome UptakeDocument9 pagesRelationship Between HBP Structure and Chrome UptakeSundarapandiyan SundaramoorthyPas encore d'évaluation
- 36 - Expantion - Calculate MethodDocument10 pages36 - Expantion - Calculate Methodmehran janPas encore d'évaluation
- Tobey-The Acid Dissociation Constant of Methyl Red. A Spectrophotometric Measurement-58Document2 pagesTobey-The Acid Dissociation Constant of Methyl Red. A Spectrophotometric Measurement-58Debora Cores Carrera0% (1)
- Mercarptans X Active CarbonDocument3 pagesMercarptans X Active CarbonAntonio CasPas encore d'évaluation
- Selective Oxidation of Methylal As A New Catalytic Route To Concentrated FormaldehydeDocument5 pagesSelective Oxidation of Methylal As A New Catalytic Route To Concentrated FormaldehydeAnonymous vWSYmPPas encore d'évaluation
- Bromination of TPM Lab ReportDocument5 pagesBromination of TPM Lab ReportImmanuel GreenePas encore d'évaluation
- Buffer solution explainedDocument27 pagesBuffer solution explainedSohel AnsariPas encore d'évaluation
- Dyes and Pigments 21 (1993) 45-66 PDFDocument22 pagesDyes and Pigments 21 (1993) 45-66 PDFAmitPas encore d'évaluation
- O-Hydroxyethylation of 1 1-DihydroperfluDocument10 pagesO-Hydroxyethylation of 1 1-DihydroperfluDeepak CharanPas encore d'évaluation
- Copper Extraction by Liquid MembraneDocument8 pagesCopper Extraction by Liquid MembraneSaad ShahPas encore d'évaluation
- Determining the Acid Dissociation Constant of Methyl Red Using Spectrophotometry (pKa = 4.86Document18 pagesDetermining the Acid Dissociation Constant of Methyl Red Using Spectrophotometry (pKa = 4.86vanessa olgaPas encore d'évaluation
- Dispersion For CeramicDocument5 pagesDispersion For CeramicThanhPas encore d'évaluation
- Exp18 NotesDocument4 pagesExp18 NotesjanePas encore d'évaluation
- Handbook of Coordination Catalysis in Organic ChemistryD'EverandHandbook of Coordination Catalysis in Organic ChemistryPas encore d'évaluation
- 05 Chapter2Document115 pages05 Chapter2Aaron AsnePas encore d'évaluation
- pH Standards at Various Temperatures: Aqueous Solutions of Acid Potassium PhthalateDocument16 pagespH Standards at Various Temperatures: Aqueous Solutions of Acid Potassium PhthalateAri CleciusPas encore d'évaluation
- PCC Technical PaperDocument6 pagesPCC Technical Papersrikant palakurthiPas encore d'évaluation
- Expt 4 Pka of Methyl Red Lab ManualDocument5 pagesExpt 4 Pka of Methyl Red Lab ManualAditya BasuPas encore d'évaluation
- 1 s2.0 0095852256900162 Main PDFDocument5 pages1 s2.0 0095852256900162 Main PDFFashihPas encore d'évaluation
- ChemistrySelect 2021 Hazer SynthesisofBiobasedBlockCopolymersUsingANovelMethacrylatedMethylSalicylateandDocument12 pagesChemistrySelect 2021 Hazer SynthesisofBiobasedBlockCopolymersUsingANovelMethacrylatedMethylSalicylateandhokagehashirama8Pas encore d'évaluation
- Buffer SolutionDocument7 pagesBuffer SolutionFerisa Wisuda NingtyasPas encore d'évaluation
- Matrix Acidizing Using HCL and Other AicdsDocument9 pagesMatrix Acidizing Using HCL and Other AicdsRamanamurthy PalliPas encore d'évaluation
- Carbonate LabDocument6 pagesCarbonate Labsamantha davidsonPas encore d'évaluation
- Bromination of AcetanilideDocument7 pagesBromination of Acetanilideaustingoewert93% (15)
- Bromination of AcetanilideDocument7 pagesBromination of AcetanilideaustingoewertPas encore d'évaluation
- New Boron (III) - Catalyzed Amide and Ester Condensation ReactionsDocument13 pagesNew Boron (III) - Catalyzed Amide and Ester Condensation ReactionsAngélica Andrea SalinasPas encore d'évaluation
- Polylactide - Viscosity RelationshipsDocument7 pagesPolylactide - Viscosity RelationshipsQasim RiazPas encore d'évaluation
- Arylation Ionic LiquidsDocument4 pagesArylation Ionic Liquidshector5palaPas encore d'évaluation
- Study On Decolorization of Methylene Blue by H2O2 Catalyzed With IronDocument6 pagesStudy On Decolorization of Methylene Blue by H2O2 Catalyzed With IrondumitriuPas encore d'évaluation
- Bahan Metilen BlueDocument5 pagesBahan Metilen Blue4uvq5j3lPas encore d'évaluation
- Lee1997 - 97 - Removal of Ethylene Blue Onto HExane Extracted Spent Bleaching EarthDocument7 pagesLee1997 - 97 - Removal of Ethylene Blue Onto HExane Extracted Spent Bleaching EarthMark Le PetitPas encore d'évaluation
- Carbon Dioxide Absorption Into Promoted Carbonate SolutionsDocument10 pagesCarbon Dioxide Absorption Into Promoted Carbonate SolutionsDunyu LiuPas encore d'évaluation
- Decolorizing Industrial Dye WastewaterDocument7 pagesDecolorizing Industrial Dye WastewaterizatPas encore d'évaluation
- Cui 2014Document7 pagesCui 2014Carolina Velásquez QuinteroPas encore d'évaluation
- Fatty Acid Analysis of Cooking OilsDocument3 pagesFatty Acid Analysis of Cooking OilsMohammad AliPas encore d'évaluation
- Kristofic Et Al - Modification of PA-6 Fibres With Alkiline CopolyamidesDocument6 pagesKristofic Et Al - Modification of PA-6 Fibres With Alkiline Copolyamidespanagiota memouPas encore d'évaluation
- Aluminum doped SBA-15 for removing remazol yellow dyeDocument40 pagesAluminum doped SBA-15 for removing remazol yellow dyeMagui Ros MonPas encore d'évaluation
- Jps 03 41 1677 AcarDocument4 pagesJps 03 41 1677 AcarMerry PaembonanPas encore d'évaluation
- Laboratory Report: Sko3023: Organic Chemistry I Semester II Session 2018/2019Document7 pagesLaboratory Report: Sko3023: Organic Chemistry I Semester II Session 2018/2019Ayuni Nadrah Bt KamarujamanPas encore d'évaluation
- Nanoporous Catalysts for Biomass ConversionD'EverandNanoporous Catalysts for Biomass ConversionFeng-Shou XiaoPas encore d'évaluation
- Homogeneous Catalysis: Mechanisms and Industrial ApplicationsD'EverandHomogeneous Catalysis: Mechanisms and Industrial ApplicationsPas encore d'évaluation
- An Overview of Chemical Additives Present in Plastics: Migration, Release, Fate and Environmental Impact During Their Use, Disposal and RecyclingDocument21 pagesAn Overview of Chemical Additives Present in Plastics: Migration, Release, Fate and Environmental Impact During Their Use, Disposal and RecyclingChris BurgerPas encore d'évaluation
- 1 s2.0 S0956053X12001304 Main PDFDocument13 pages1 s2.0 S0956053X12001304 Main PDFVu Anh KienPas encore d'évaluation
- 1 s2.0 S0956053X12001304 Main PDFDocument13 pages1 s2.0 S0956053X12001304 Main PDFVu Anh KienPas encore d'évaluation
- IE & SE Chapter 2Document17 pagesIE & SE Chapter 2Vu Anh KienPas encore d'évaluation
- Immune Function in Cigarette Smokers Who Quit Smoking For 31 DaysDocument10 pagesImmune Function in Cigarette Smokers Who Quit Smoking For 31 DaysVu Anh KienPas encore d'évaluation
- Analytical Methods PDFDocument14 pagesAnalytical Methods PDFoptisearchPas encore d'évaluation
- Chapter 1 - Crittenden's TextbookDocument8 pagesChapter 1 - Crittenden's TextbookVu Anh KienPas encore d'évaluation
- Calibration Curve 1Document2 pagesCalibration Curve 1Vu Anh KienPas encore d'évaluation
- Term Paper Grading RubricDocument1 pageTerm Paper Grading RubricVu Anh KienPas encore d'évaluation
- Ozonation of U.S. Drinking Water SourcesDocument8 pagesOzonation of U.S. Drinking Water SourcesVu Anh KienPas encore d'évaluation
- Toxicology in Vitro: Subhashini Arimilli, Brad E. Damratoski, G.L. PrasadDocument13 pagesToxicology in Vitro: Subhashini Arimilli, Brad E. Damratoski, G.L. PrasadVu Anh KienPas encore d'évaluation
- Reflections on a Tenure-Track Job SearchDocument18 pagesReflections on a Tenure-Track Job SearchrnatellaPas encore d'évaluation
- HEC-RAS 4.1 Reference Manual - DesbloqueadoDocument417 pagesHEC-RAS 4.1 Reference Manual - Desbloqueadohenrysoll999Pas encore d'évaluation
- TWE TOEFL Writing TemplatesDocument16 pagesTWE TOEFL Writing TemplatesVu Anh KienPas encore d'évaluation
- Speaking TemplateDocument2 pagesSpeaking TemplateVu Anh KienPas encore d'évaluation
- Yale Student's Cover Letter for WRI Energy & Climate Change InternshipDocument1 pageYale Student's Cover Letter for WRI Energy & Climate Change InternshipVu Anh KienPas encore d'évaluation
- ArcGIS authorization codesDocument2 pagesArcGIS authorization codesVu Anh KienPas encore d'évaluation
- Hiragana WorksheetDocument7 pagesHiragana WorksheetVu Anh KienPas encore d'évaluation
- Volunteer Job - Roles and ResponsibilitiesDocument2 pagesVolunteer Job - Roles and ResponsibilitiesVu Anh KienPas encore d'évaluation
- f1 CPT FinalDocument3 pagesf1 CPT FinalVu Anh KienPas encore d'évaluation
- CWR 4120 5125 HW1 Fa14Document1 pageCWR 4120 5125 HW1 Fa14Vu Anh KienPas encore d'évaluation
- Quiz For LabDocument2 pagesQuiz For LabVu Anh KienPas encore d'évaluation
- 05 Independent Study Quiz - Kien VuDocument5 pages05 Independent Study Quiz - Kien VuVu Anh KienPas encore d'évaluation
- Teaching Strategies and Insights for New TADocument4 pagesTeaching Strategies and Insights for New TAVu Anh KienPas encore d'évaluation
- 01 Independent Study ChecklistDocument1 page01 Independent Study ChecklistVu Anh KienPas encore d'évaluation
- Cover Letter Advice - Colgate UniversityDocument7 pagesCover Letter Advice - Colgate UniversityVu Anh KienPas encore d'évaluation
- Preparation Process of E. ColiDocument1 pagePreparation Process of E. ColiVu Anh KienPas encore d'évaluation
- How To Enjoy Your Life and Your Job (Summary) - Dale CarnegieDocument9 pagesHow To Enjoy Your Life and Your Job (Summary) - Dale CarnegieVu Anh Kien100% (3)
- Dale Carnegie Golden Book-Se PDFDocument7 pagesDale Carnegie Golden Book-Se PDFRanjith Kumar GoudPas encore d'évaluation
- Newton's Laws of Motion Lab Questions Answer KeyDocument2 pagesNewton's Laws of Motion Lab Questions Answer KeyAbdulla NassPas encore d'évaluation
- Adobe After Effects CS3 Keyboard Shortcuts GuideDocument14 pagesAdobe After Effects CS3 Keyboard Shortcuts GuideBrandon Sirota100% (1)
- Novel Proteinaceous Infectious Particles Cause ScrapieDocument10 pagesNovel Proteinaceous Infectious Particles Cause ScrapieMikey HaveyPas encore d'évaluation
- Primary Reformer TubesDocument10 pagesPrimary Reformer TubesAhmed ELmlahyPas encore d'évaluation
- 03a IGCSE Maths 4MB1 Paper 2R - January 2020 Examination PaperDocument32 pages03a IGCSE Maths 4MB1 Paper 2R - January 2020 Examination PaperMehwish ArifPas encore d'évaluation
- Eclipse RCPDocument281 pagesEclipse RCPjpradeebanPas encore d'évaluation
- Air Suspension Benefits Over Steel SpringsDocument3 pagesAir Suspension Benefits Over Steel SpringsBejoy G NairPas encore d'évaluation
- Vorplex - MST - Airblowing and Water FlushingDocument14 pagesVorplex - MST - Airblowing and Water FlushingAmirHakimRusliPas encore d'évaluation
- Line Tension and Pole StrengthDocument34 pagesLine Tension and Pole StrengthDon BunPas encore d'évaluation
- Junction boxes gas group IIC selectionDocument16 pagesJunction boxes gas group IIC selectionkskadryPas encore d'évaluation
- Lab Report Mass Spring System Omar Nizar2Document31 pagesLab Report Mass Spring System Omar Nizar2Omar MustafaPas encore d'évaluation
- The 2009 ACM ASIA Programming Contest Dhaka Site: Hosted by North South University Dhaka, BangladeshDocument21 pagesThe 2009 ACM ASIA Programming Contest Dhaka Site: Hosted by North South University Dhaka, BangladeshPhạm Hữu Thạnh ĐạtPas encore d'évaluation
- Mark SchemeDocument12 pagesMark SchemeNdanji SiamePas encore d'évaluation
- Reinforcement Detailing in BeamsDocument9 pagesReinforcement Detailing in Beamssaheed tijaniPas encore d'évaluation
- Class XI Chemistry Question BankDocument71 pagesClass XI Chemistry Question BankNirmalaPas encore d'évaluation
- Continuous DeploymentDocument2 pagesContinuous DeploymentVenkat Reddy0% (1)
- Assignment No.3 Bolted JointsDocument6 pagesAssignment No.3 Bolted JointsYash SahuPas encore d'évaluation
- Name Source Description Syntax Par, Frequency, Basis)Document12 pagesName Source Description Syntax Par, Frequency, Basis)alsaban_7Pas encore d'évaluation
- Chapter 5: Work, Energy and PowerDocument4 pagesChapter 5: Work, Energy and PowerPriyaa JayasankarPas encore d'évaluation
- Lecture 2 - Kinematics Fundamentals - Part ADocument30 pagesLecture 2 - Kinematics Fundamentals - Part ASuaid Tariq BalghariPas encore d'évaluation
- Dss Paper 1Document2 pagesDss Paper 1hemalPas encore d'évaluation
- Temperature Effect On Voc and IscDocument5 pagesTemperature Effect On Voc and IscAnonymous bVLovsnPas encore d'évaluation
- Principles of Differential RelayingDocument115 pagesPrinciples of Differential RelayingelitesankarPas encore d'évaluation
- SERIES-90 180cc SERVICE BLN-2-41695 1997-05Document52 pagesSERIES-90 180cc SERVICE BLN-2-41695 1997-05thailanPas encore d'évaluation
- FOUNDATION REPAIR AND REGROUT FOR BODYMAKER AT CARNDAUD METALBOX SINGAPOREDocument15 pagesFOUNDATION REPAIR AND REGROUT FOR BODYMAKER AT CARNDAUD METALBOX SINGAPORETrúc NguyễnPas encore d'évaluation
- Laing Electric Heater-CirculatorDocument20 pagesLaing Electric Heater-Circulatorkamilawehbe100% (1)
- Dev - Mag - 09Document26 pagesDev - Mag - 09georgpiorczynskiPas encore d'évaluation
- Priority Academic Student Skills Mathematics Grades 1-5Document44 pagesPriority Academic Student Skills Mathematics Grades 1-5faithinhim7515Pas encore d'évaluation
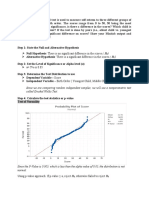
- Step 1: State The Null and Alternative HypothesisDocument3 pagesStep 1: State The Null and Alternative HypothesisChristine Joyce BascoPas encore d'évaluation
- Assignment 3Document2 pagesAssignment 3Spring RollsPas encore d'évaluation