Académique Documents
Professionnel Documents
Culture Documents
DR Balbir Singh
Transféré par
Anonymous CwJeBCAXpTitre original
Copyright
Formats disponibles
Partager ce document
Partager ou intégrer le document
Avez-vous trouvé ce document utile ?
Ce contenu est-il inapproprié ?
Signaler ce documentDroits d'auteur :
Formats disponibles
DR Balbir Singh
Transféré par
Anonymous CwJeBCAXpDroits d'auteur :
Formats disponibles
SRJIS/BIMONTHLY/ DR.
BALBIR SINGH (2511-2516)
SYNTHESIS OF SOMEMETAL COMPLEXES AND ITS STRUCTURAL
INVESTIGATION
Balbir Singh, Ph. D.
Former HOD, Dept. of Chemistry. SD College Pathankot.
Abstract
Preparation and the structural investigation of
phosphinetelluride as a ligand.
someorgano metallic compounds using triaryl
Scholarly Research Journal's is licensed Based on a work at www.srjis.com
Introduction:-The field of coordination chemistry is growing exponentially. Particularly in the last two
decades development in bio -organic chemistry has gathered muchmomentumin the subject.
Thus the present topic has been selected.
The complexing nature of organophosphineis due to the lone pair of electrons on the
phosphorous atom. But when phosphorous attaches with elements (O, S, Se, Teetc) it
becomes pentavalent and it makes double bond with the metal thus making it to act as a
donor. Due to this bonding P becomes slightly positive. And will result in the contraction of
its 3d-orbitals. The ii charge cloud is expected to be distorted in favour of metal thus making
the metal an excellent5 donor and a good complexing agent. Survey study shows that various
metal complexes of phosphine oxide, sulphide, & selenide have been reported where as very
little efforts have been made to prepare phosphine telluride complexes (14).Te is short of
two electrons than octet, high electronegativity and small size show it has non-metallic
covalent chemistry.
In the present series of investigation Cu ll, Ni ll,Coll, Cr lll and Zn ll halides have been
chosen as metal ions to form complexes with tri-xylyl phosphine telluride.
JULY-AUG 2016, VOL-4/25
www.srjis.com
Page 2511
SRJIS/BIMONTHLY/ DR. BALBIR SINGH (2511-2516)
Experimental:----IR spectra is recorded on Perkin-Elmer-577 IR spectrometer. (range 200--4000/cm) . Percentage composition of the complex is determined by usual gravimetric
&instrumental analyser method. Magnetic moment is measured by Guoys balance method.
Preparation of ligand:---The recrystallizedtriphenylphosphine has been dissolved in xylene
and to this added calculated amount of tellurium metal. The mixture is refluxed on water bath
for 2 to 3 hours. The contents are allowed to cool and filtered.The filtrate is concentrated to
give crystals of titled complex which are recrystallized from dry alcohol.
Preparation of complexes:---A ligand is dissolved in minimum quantity of dry alcohol and
the salt of metal M ( M= CoCl2,NiCl2,CuCl2,CrCl2and ZnCl2) in minimum quantity of
water. Both are mixed in equimolar quantity in a round bottomed flask fitted with water
condenser. The contents were refluxedfor 3 to 4 hours. Contents are then removed stirred
well and allowed to stand overnight. Complex is separated out which was recrystallized from
dry alcohol.. Crystals are filtered & dried gently in the folds of filter papers.
JULY-AUG 2016, VOL-4/25
www.srjis.com
Page 2512
SRJIS/BIMONTHLY/ DR. BALBIR SINGH (2511-2516)
TABLE 1
Assignm
ent
C--H,stre
C=
C,
stre
C-H,inplan
e defor
C-H,outpla
ne defor
liga
nd
3047
,m
1478
,v s
C-P,asym
C--p,sym
P-Te,stre
M--X,stre
M--Te,
stre
510,
vs
451,
s
440,
s
----
Cu(l
l)
3046
,s
1472
,(v s)
1305
,s
119
0,s
759,
s
723,
v s
s69
8, v
s6
111
0,v
s
698,
vs
109
3,v
s
630,
m
--
1310
,v s
118
8,s
751,
vs
741,
s
507,
sh
448,
s
427
,v s
378,
m
220,
vs
503,
vs
432,
s
109
7,v
s
718,
vs
103
0,s
Ni(l
l)
304
0,s
147
5,v
s
131
1,s
119
1,s
698,
vs
744,
vs
710,
vs
508,
sh
446,
s
435,
vs
350,
w
206,
m
504,
vs
430,
s
109
7,v
s
702,
vs
103
0,s
Co(l
l)
304
7,s
157
6,v
s
131
0,s
119
2,s
620,
s
745,
vs
710,
vs
508,
sh
445,
s
437,
vs
351,
w
206,
m
503,
vs
420,
s
stre= streching
m= medium
asym=symetric
w= wesk
sym=symetric
v w=veryeesk
defor= deformation
sh= soldier
109
8,v
s
702,
vs
103
1,s
620,
s
Cr(l
ll)
304
8,s
147
3,v
s
131
5,v
s
746,
s
503,
w
450,
sh
435,
vs
352,
s
221,
s
Zn(ll
)
3048,
vw
1478,
s
109
2,s
109
0,s
108
0w
1315,
w
1190
,w
719,
sh
698,
s
609,
s
740,s
720,
m
512,v
s
498,
s
450,
w
426,
vs
392,
m
309,
s
110
8,v
s
699,
m
100
5,
621
,w
v s=very strong
JULY-AUG 2016, VOL-4/25
www.srjis.com
Page 2513
SRJIS/BIMONTHLY/ DR. BALBIR SINGH (2511-2516)
TABLE 2
compl
ex
v
eleme
nts
>
metal
halogen
(theo)obs
(theo)obs
--'------
(55.49)55.59
CuCl2
complex
NiCl2 complex
--------(6.48)6
54
(6.45)6.49
(7.79)7.84
(47.53)47.62
7.79)7.83
47.54)47.60
CoCl2 complex
(3.92)3.97
3.97)4.01
(48.83)48.88
Cr
Cl2
complex
ZnCl2 complex
3.75)3.
79
(7.13)7.20
(3.79)4.18
(51.05)51.11
(7.20)7.74
(47.20)47.30
ligand
carb
an
(theo)obs
hyddro
gen
(theo)ob
s
(3.85)3.
94
(3.32)3.
33
(3.32)3.
41
(3.41)3.
46
(3.57)3.
63
(3.30)3.
34
phosphorous
(theo)obs
(795)7.97
telluri
um
(theo)obs
(6.81)6.87
(32.75)32.80(28.05)28.
12
(28.05)28.13
(6.81)6.87
(28.06)28.12
(6.99)7.00
28.82)28.89
(7.31)7.31(
(30.12)30.21
(6.70)6.72
(27.85)27.91
Elemental analysis
theo = theoretical
obs = observed
JULY-AUG 2016, VOL-4/25
www.srjis.com
Page 2514
SRJIS/BIMONTHLY/ DR. BALBIR SINGH (2511-2516)
TABLE 3
compond
stocheonetric
ratio
Cu(ll) compond
Ni(ll) compond
Co(ll) compond
Cr(lll) compond
Zn(ll) compond
CuCl2.4Ph(CH3)2PTe,
1:4
NiCl2.2Ph(CH3)2PTe, 1
:2
CoCl2.2Ph(CH3)2PTe,
1:2
CrCl3.3Ph(CH3)2PTe, 1
:3
ZnCl2. Ph(CH3)2PTe, 1
:1
m.pt c.grade
colour
238
240
light
blue
blue
244
intence blue
222
orange yellow
230
white
TABLE 4
JULY-AUG 2016, VOL-4/25
complex
mag.moment (BM)
Cu(ll)complex
1.9
stereo
chemistry
octahedral
Ni(ll)complex
3.1
tetrahedral
Co(ll)complex
4.82
tetrahedral
Cr(lll)complex
Zn(ll)complex
3.86
diamag.
tetrahedral
bridged
www.srjis.com
Page 2515
SRJIS/BIMONTHLY/ DR. BALBIR SINGH (2511-2516)
Discussion: ---1) In triarylphosphinetellurideligand,Te has alone pair which it can use for coordination. The
basic character of ligand is increased by two methyl groups due to its inductive effect. IR
spectra of two alkyl groups(of xylene) in the ligand remains same due p iidii bonding
between P &Te.
2) Vib. of the aromatic ring remain the same both in the ligand and the cimplex showing the
ring is intact in the complex.
3) Metal tellurium frequencies observed in the complexes which are absent in the ligand
shows that metalcoardinates through Te.
4) Metal halide frequencies are lower in complexes than in metal halide which further
supports MlTelinkage.
5) Lower wave number observed in case of Zn complex suggests the bridged structure
through halogen. The same is further suggested by its diamag. Character (due to d10
config.)thus sp3 hybridisation used by Zn and formatjonof 1:1 complex. Mag character of
complexes show that Cu(ll) complex is octahedral and Ni(ll),Co(ll)&Cr(ll) has tetrahedral
geometries.
References: --SAhrland , J Chatt and N R Devies,Quart Rev. Chem. Soc. 12,(1988)265
G E Coates,J.1951, 2003
R G Pearson,J.Am.Chem.Soc.85(1963),3533.
C K Jorgenson,Inorg.Chem.2(1964)1201
JULY-AUG 2016, VOL-4/25
www.srjis.com
Page 2516
Vous aimerez peut-être aussi
- Hidden Figures: The American Dream and the Untold Story of the Black Women Mathematicians Who Helped Win the Space RaceD'EverandHidden Figures: The American Dream and the Untold Story of the Black Women Mathematicians Who Helped Win the Space RaceÉvaluation : 4 sur 5 étoiles4/5 (895)
- 13.nasir RasheedDocument9 pages13.nasir RasheedAnonymous CwJeBCAXpPas encore d'évaluation
- Never Split the Difference: Negotiating As If Your Life Depended On ItD'EverandNever Split the Difference: Negotiating As If Your Life Depended On ItÉvaluation : 4.5 sur 5 étoiles4.5/5 (838)
- Customers' Consciousness About Financial Cyber Frauds in Electronic Banking: An Indian Perspective With Special Reference To Mumbai CityDocument13 pagesCustomers' Consciousness About Financial Cyber Frauds in Electronic Banking: An Indian Perspective With Special Reference To Mumbai CityAnonymous CwJeBCAXpPas encore d'évaluation
- The Yellow House: A Memoir (2019 National Book Award Winner)D'EverandThe Yellow House: A Memoir (2019 National Book Award Winner)Évaluation : 4 sur 5 étoiles4/5 (98)
- Homesh RaniDocument7 pagesHomesh RaniAnonymous CwJeBCAXpPas encore d'évaluation
- The Subtle Art of Not Giving a F*ck: A Counterintuitive Approach to Living a Good LifeD'EverandThe Subtle Art of Not Giving a F*ck: A Counterintuitive Approach to Living a Good LifeÉvaluation : 4 sur 5 étoiles4/5 (5794)
- 29.yuvraj SutarDocument4 pages29.yuvraj SutarAnonymous CwJeBCAXpPas encore d'évaluation
- Shoe Dog: A Memoir by the Creator of NikeD'EverandShoe Dog: A Memoir by the Creator of NikeÉvaluation : 4.5 sur 5 étoiles4.5/5 (537)
- 25.suresh ChenuDocument9 pages25.suresh ChenuAnonymous CwJeBCAXpPas encore d'évaluation
- Devil in the Grove: Thurgood Marshall, the Groveland Boys, and the Dawn of a New AmericaD'EverandDevil in the Grove: Thurgood Marshall, the Groveland Boys, and the Dawn of a New AmericaÉvaluation : 4.5 sur 5 étoiles4.5/5 (266)
- 19.Dr Ibrahim Aliyu ShehuDocument29 pages19.Dr Ibrahim Aliyu ShehuAnonymous CwJeBCAXpPas encore d'évaluation
- The Little Book of Hygge: Danish Secrets to Happy LivingD'EverandThe Little Book of Hygge: Danish Secrets to Happy LivingÉvaluation : 3.5 sur 5 étoiles3.5/5 (400)
- 1.prof. Ajay Kumar AttriDocument8 pages1.prof. Ajay Kumar AttriAnonymous CwJeBCAXpPas encore d'évaluation
- Elon Musk: Tesla, SpaceX, and the Quest for a Fantastic FutureD'EverandElon Musk: Tesla, SpaceX, and the Quest for a Fantastic FutureÉvaluation : 4.5 sur 5 étoiles4.5/5 (474)
- Technostress, Computer Self-Efficacy and Perceived Organizational Support Among Secondary School Teachers: Difference in Type of School, Gender and AgeDocument13 pagesTechnostress, Computer Self-Efficacy and Perceived Organizational Support Among Secondary School Teachers: Difference in Type of School, Gender and AgeAnonymous CwJeBCAXpPas encore d'évaluation
- A Study of Effect of Age and Gender On Stress of AdolescentsDocument5 pagesA Study of Effect of Age and Gender On Stress of AdolescentsAnonymous CwJeBCAXpPas encore d'évaluation
- A Heartbreaking Work Of Staggering Genius: A Memoir Based on a True StoryD'EverandA Heartbreaking Work Of Staggering Genius: A Memoir Based on a True StoryÉvaluation : 3.5 sur 5 étoiles3.5/5 (231)
- 4.Dr Gagandeep KaurDocument13 pages4.Dr Gagandeep KaurAnonymous CwJeBCAXpPas encore d'évaluation
- Grit: The Power of Passion and PerseveranceD'EverandGrit: The Power of Passion and PerseveranceÉvaluation : 4 sur 5 étoiles4/5 (588)
- Aggression Among Senior Secondary School Students in Relation To Their Residential BackgroundDocument8 pagesAggression Among Senior Secondary School Students in Relation To Their Residential BackgroundAnonymous CwJeBCAXpPas encore d'évaluation
- The Emperor of All Maladies: A Biography of CancerD'EverandThe Emperor of All Maladies: A Biography of CancerÉvaluation : 4.5 sur 5 étoiles4.5/5 (271)
- 29 Balwinder SinghDocument8 pages29 Balwinder SinghAnonymous CwJeBCAXpPas encore d'évaluation
- The Unwinding: An Inner History of the New AmericaD'EverandThe Unwinding: An Inner History of the New AmericaÉvaluation : 4 sur 5 étoiles4/5 (45)
- A Lookout at Traditional Games Played by Tribes in IndiaDocument4 pagesA Lookout at Traditional Games Played by Tribes in IndiaAnonymous CwJeBCAXpPas encore d'évaluation
- On Fire: The (Burning) Case for a Green New DealD'EverandOn Fire: The (Burning) Case for a Green New DealÉvaluation : 4 sur 5 étoiles4/5 (74)
- The Need of Remote Voting Machine in Indian Voting SystemDocument7 pagesThe Need of Remote Voting Machine in Indian Voting SystemAnonymous CwJeBCAXpPas encore d'évaluation
- The Hard Thing About Hard Things: Building a Business When There Are No Easy AnswersD'EverandThe Hard Thing About Hard Things: Building a Business When There Are No Easy AnswersÉvaluation : 4.5 sur 5 étoiles4.5/5 (345)
- Women Empowerment and Religion's Role in Gender Relations in KargilDocument10 pagesWomen Empowerment and Religion's Role in Gender Relations in KargilAnonymous CwJeBCAXpPas encore d'évaluation
- Team of Rivals: The Political Genius of Abraham LincolnD'EverandTeam of Rivals: The Political Genius of Abraham LincolnÉvaluation : 4.5 sur 5 étoiles4.5/5 (234)
- Effect of Life Skill Training On Mental Health Among B.ed. Interns in Relation To Their Impulsive BehaviourDocument9 pagesEffect of Life Skill Training On Mental Health Among B.ed. Interns in Relation To Their Impulsive BehaviourAnonymous CwJeBCAXpPas encore d'évaluation
- Historical Development of Play Schools in IndiaDocument11 pagesHistorical Development of Play Schools in IndiaAnonymous CwJeBCAXpPas encore d'évaluation
- 31 Dr. Suman Kumari, Prof. Sudarshana Rana & Ms. Anita VermaDocument9 pages31 Dr. Suman Kumari, Prof. Sudarshana Rana & Ms. Anita VermaAnonymous CwJeBCAXpPas encore d'évaluation
- The Gifts of Imperfection: Let Go of Who You Think You're Supposed to Be and Embrace Who You AreD'EverandThe Gifts of Imperfection: Let Go of Who You Think You're Supposed to Be and Embrace Who You AreÉvaluation : 4 sur 5 étoiles4/5 (1090)
- The Use of English Language During PandemicDocument7 pagesThe Use of English Language During PandemicAnonymous CwJeBCAXpPas encore d'évaluation
- 26 Dr. Reni Francis Mr. Rajendra DeshmukhDocument5 pages26 Dr. Reni Francis Mr. Rajendra DeshmukhAnonymous CwJeBCAXpPas encore d'évaluation
- 28 Shailaja KanwarDocument12 pages28 Shailaja KanwarAnonymous CwJeBCAXpPas encore d'évaluation
- The World Is Flat 3.0: A Brief History of the Twenty-first CenturyD'EverandThe World Is Flat 3.0: A Brief History of the Twenty-first CenturyÉvaluation : 3.5 sur 5 étoiles3.5/5 (2259)
- 30 Trishala BhaskarDocument7 pages30 Trishala BhaskarAnonymous CwJeBCAXpPas encore d'évaluation
- 24 Adv Raj KumarDocument5 pages24 Adv Raj KumarAnonymous CwJeBCAXpPas encore d'évaluation
- 25 AshaDocument5 pages25 AshaAnonymous CwJeBCAXpPas encore d'évaluation
- 22 Pavithra.g ArticleDocument8 pages22 Pavithra.g ArticleAnonymous CwJeBCAXpPas encore d'évaluation
- 23 JayalakshmiDocument9 pages23 JayalakshmiAnonymous CwJeBCAXpPas encore d'évaluation
- The Sympathizer: A Novel (Pulitzer Prize for Fiction)D'EverandThe Sympathizer: A Novel (Pulitzer Prize for Fiction)Évaluation : 4.5 sur 5 étoiles4.5/5 (121)
- Suyambukani VDocument6 pagesSuyambukani VAnonymous CwJeBCAXpPas encore d'évaluation
- Deepa AnwarDocument17 pagesDeepa AnwarAnonymous CwJeBCAXpPas encore d'évaluation
- Payal BhatiDocument10 pagesPayal BhatiAnonymous CwJeBCAXpPas encore d'évaluation
- 21 DR ReniDocument6 pages21 DR ReniAnonymous CwJeBCAXpPas encore d'évaluation
- Level & Section: The Lesson Proper)Document2 pagesLevel & Section: The Lesson Proper)Adine Jeminah LimonPas encore d'évaluation
- Science Puz Twist TeasDocument3 pagesScience Puz Twist Teasapi-249777358Pas encore d'évaluation
- Simplified Chemistry 2019. (5124) - 10 - 12.Document176 pagesSimplified Chemistry 2019. (5124) - 10 - 12.Ck ChiyesuPas encore d'évaluation
- Activity 1 Functional Groups and Lewis StructureDocument6 pagesActivity 1 Functional Groups and Lewis StructurePearl NecolePas encore d'évaluation
- Chemical Bonding and Molecular StructureDocument79 pagesChemical Bonding and Molecular StructureGanesh V GaonkarPas encore d'évaluation
- Periodic Shobhit NirwanDocument22 pagesPeriodic Shobhit NirwanGauravPas encore d'évaluation
- Percdc Cns Geas 1Document9 pagesPercdc Cns Geas 1Charles Adrian CPas encore d'évaluation
- Electronegativity PDFDocument42 pagesElectronegativity PDFNehaPas encore d'évaluation
- Exploring Chemical Bonding: Lesson OverviewDocument13 pagesExploring Chemical Bonding: Lesson OverviewCherry Pie Muñoz JimenoPas encore d'évaluation
- CBSE Class 7 Science The Language of Chemistry Exam Notes: IntroductionDocument8 pagesCBSE Class 7 Science The Language of Chemistry Exam Notes: Introductionanika varshneyPas encore d'évaluation
- Her Body and Other Parties: StoriesD'EverandHer Body and Other Parties: StoriesÉvaluation : 4 sur 5 étoiles4/5 (821)
- The Nature of MoleculesDocument42 pagesThe Nature of Moleculeseasy99Pas encore d'évaluation
- Chemistry Chapter 1.exercise 1ADocument28 pagesChemistry Chapter 1.exercise 1AAsifPas encore d'évaluation
- CHEM 103M - Compilation of Lecture Notes (Midterms)Document110 pagesCHEM 103M - Compilation of Lecture Notes (Midterms)nuggetPas encore d'évaluation
- Revision For First Term 9GCE 2010 11Document30 pagesRevision For First Term 9GCE 2010 11Anonymous 8VJhV1eI2yPas encore d'évaluation
- Forces of Attraction and Chemical BondingDocument15 pagesForces of Attraction and Chemical BondingchanPas encore d'évaluation
- Mat Sci Slides 2Document29 pagesMat Sci Slides 2abcdPas encore d'évaluation
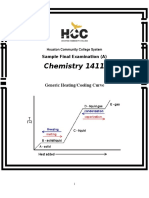
- Chemistry 1411: Generic Heating/Cooling CurveDocument19 pagesChemistry 1411: Generic Heating/Cooling CurveKinal PatelPas encore d'évaluation
- Chapter Test A: Chemical BondingDocument4 pagesChapter Test A: Chemical BondingPencils of PromisePas encore d'évaluation
- Periodic Table - 62760 - 2023 - 05 - 06 - 21 - 02Document31 pagesPeriodic Table - 62760 - 2023 - 05 - 06 - 21 - 02Tae KookPas encore d'évaluation
- Chemical BondingDocument108 pagesChemical BondingAlbert Jade Pontimayor LegariaPas encore d'évaluation
- Chem Essentials 00 Hess RichDocument700 pagesChem Essentials 00 Hess RichChristina ThompsonPas encore d'évaluation
- Ncert Solutions Class 9 Science Chapter 4Document11 pagesNcert Solutions Class 9 Science Chapter 4Mukesh KharbPas encore d'évaluation
- 03 - Atoms and MoleculesDocument20 pages03 - Atoms and MoleculesAtharv SoniPas encore d'évaluation
- On The Principle of Maximum Overlap in Molecular Orbital TheoryDocument4 pagesOn The Principle of Maximum Overlap in Molecular Orbital Theoryleizar_death64Pas encore d'évaluation
- (Q1) MODULE 4 - Chemical and Structural Formulas PDFDocument18 pages(Q1) MODULE 4 - Chemical and Structural Formulas PDFJewel SantiagoPas encore d'évaluation
- Spe 113976 Losal Enhanced Oil Recovery: Evidence of Enhanced Oil Recovery at The Reservoir ScaleDocument12 pagesSpe 113976 Losal Enhanced Oil Recovery: Evidence of Enhanced Oil Recovery at The Reservoir ScaleNitin SharmaPas encore d'évaluation
- Intermetallische Phasen - Antworten-1-30 en-USDocument30 pagesIntermetallische Phasen - Antworten-1-30 en-USLorena juárezPas encore d'évaluation
- Progressive GR 9 2nd Q CDocument21 pagesProgressive GR 9 2nd Q CRAMIL BAUTISTAPas encore d'évaluation
- Class VII OPJGS WORKSHEETS 2020Document50 pagesClass VII OPJGS WORKSHEETS 2020manishaPas encore d'évaluation
- Covalent Bonding Lewis Structure WebquestDocument16 pagesCovalent Bonding Lewis Structure WebquestDean JezerPas encore d'évaluation
- Taste: Surprising Stories and Science About Why Food Tastes GoodD'EverandTaste: Surprising Stories and Science About Why Food Tastes GoodÉvaluation : 3 sur 5 étoiles3/5 (20)
- AP® Chemistry Crash Course, For the 2020 Exam, Book + Online: Get a Higher Score in Less TimeD'EverandAP® Chemistry Crash Course, For the 2020 Exam, Book + Online: Get a Higher Score in Less TimeÉvaluation : 5 sur 5 étoiles5/5 (1)
- The Nature of Drugs Vol. 1: History, Pharmacology, and Social ImpactD'EverandThe Nature of Drugs Vol. 1: History, Pharmacology, and Social ImpactÉvaluation : 5 sur 5 étoiles5/5 (5)
- Periodic Tales: A Cultural History of the Elements, from Arsenic to ZincD'EverandPeriodic Tales: A Cultural History of the Elements, from Arsenic to ZincÉvaluation : 3.5 sur 5 étoiles3.5/5 (137)
- It's Elemental: The Hidden Chemistry in EverythingD'EverandIt's Elemental: The Hidden Chemistry in EverythingÉvaluation : 4 sur 5 étoiles4/5 (10)
- Is That a Fact?: Frauds, Quacks, and the Real Science of Everyday LifeD'EverandIs That a Fact?: Frauds, Quacks, and the Real Science of Everyday LifeÉvaluation : 5 sur 5 étoiles5/5 (4)
- Chemistry for Breakfast: The Amazing Science of Everyday LifeD'EverandChemistry for Breakfast: The Amazing Science of Everyday LifeÉvaluation : 4.5 sur 5 étoiles4.5/5 (14)