Académique Documents
Professionnel Documents
Culture Documents
Stability Study With SAP Quality Management
Transféré par
torr123100%(1)100% ont trouvé ce document utile (1 vote)
497 vues54 pagesThe document discusses configuring SAP Quality Management for stability studies. Key points include:
1. Configuring inspection types, user status profiles, and notification types for stability studies.
2. Defining primary packaging, physical sample containers, storage conditions, and storage locations.
3. Creating a maintenance strategy, assigning inspection types and plans to materials, and assigning maintenance packages to inspection plans to generate planned stations.
4. The standard process of creating a quality notification to start a stability study.
Description originale:
SAP
Titre original
323234961 Stability Study With SAP Quality Management
Copyright
© © All Rights Reserved
Formats disponibles
TXT, PDF, TXT ou lisez en ligne sur Scribd
Partager ce document
Partager ou intégrer le document
Avez-vous trouvé ce document utile ?
Ce contenu est-il inapproprié ?
Signaler ce documentThe document discusses configuring SAP Quality Management for stability studies. Key points include:
1. Configuring inspection types, user status profiles, and notification types for stability studies.
2. Defining primary packaging, physical sample containers, storage conditions, and storage locations.
3. Creating a maintenance strategy, assigning inspection types and plans to materials, and assigning maintenance packages to inspection plans to generate planned stations.
4. The standard process of creating a quality notification to start a stability study.
Droits d'auteur :
© All Rights Reserved
Formats disponibles
Téléchargez comme TXT, PDF, TXT ou lisez en ligne sur Scribd
100%(1)100% ont trouvé ce document utile (1 vote)
497 vues54 pagesStability Study With SAP Quality Management
Transféré par
torr123The document discusses configuring SAP Quality Management for stability studies. Key points include:
1. Configuring inspection types, user status profiles, and notification types for stability studies.
2. Defining primary packaging, physical sample containers, storage conditions, and storage locations.
3. Creating a maintenance strategy, assigning inspection types and plans to materials, and assigning maintenance packages to inspection plans to generate planned stations.
4. The standard process of creating a quality notification to start a stability study.
Droits d'auteur :
© All Rights Reserved
Formats disponibles
Téléchargez comme TXT, PDF, TXT ou lisez en ligne sur Scribd
Vous êtes sur la page 1sur 54
Stability Study with SAP Quality Management
Stability Study with SAP QM
1. Configuration
1.1. Inspection types
Although the inspection types for stability are preconfigured in SAP QM. Still,
you can configure the custom
inspection types for stability.
Follow the Path: IMG >> Quality Management >> Quality Inspection >> Inspection L
ot Creation >> Maintain
Inspection Types
16
Inspection for Storage Condition (Stabi)
1601 Initial Test (Stability Studies)
1602
Manual Insp. Lot for Storage Cond (Stab)
1.2. User Status Profile
The preconfigured user status profile has limitation to the fixed process for st
ability study. You can copy and
configure custom status profile as per the business requirement.
Run Transaction BS02 to access the status profiles.
Add/Remove/Modify the business transaction for a status by double clicking the s
tatus.
Generated by Jive on 2016-07-26Z
1
Stability Study with SAP Quality Management
Generated by Jive on 2016-07-26Z
2
Stability Study with SAP Quality Management
1.3. Notification type and the Action box
There are standard preconfigured notification type available. You can refer the
same and use it as a reference
to create a custom notification type. Also, you can add/remove/modify the action
box for the notification type.
Generated by Jive on 2016-07-26Z
3
Stability Study with SAP Quality Management
Generated by Jive on 2016-07-26Z
4
Stability Study with SAP Quality Management
Generated by Jive on 2016-07-26Z
5
Stability Study with SAP Quality Management
1.4. Primary Packaging
You need to define the primary packaging for Physical Samples
Definition: The packing material which is in direct contact with the product is
considered as primary packaging.
There may be container (bottle, ampule), closure (bottle cap) and desiccant (sil
ica gel)
Follow the menu path IMG >> Quality Management >> Stability Study >> Basic Data
>> Define Primary
Packaging
Generated by Jive on 2016-07-26Z
6
Stability Study with SAP Quality Management
1.5. Physical Sample Container
You need to define the physical sample container for physical sample
Definition: The containing material in which the samples are placed in stability
chamber.
1. E.g.
10 strips are packed in carton and placed in stability chamber then the carton i
s the physical sample container
10 strips (Aluminum and PVC blister) are placed in the stability chamber then th
e Aluminum and PVC blister is
the physical sample container
Follow the menu path IMG >> Quality Management >> Quality Inspection >> Sample M
anagement >> Define
Physical-Sample Container
Generated by Jive on 2016-07-26Z
7
Stability Study with SAP Quality Management
1.6. Storage Condition
You need to define the storage condition for Physical sample. It is the most imp
ortant configuration for stability
study
Definition: The temperature and humidity level in which the physical samples are
placed is called the storage
condition
1. E.g. Temperature 25C 2C and Relative Humidity 65% 5% is one of the storage cond
ition. It can
be a long term/real time storage condition for a zone or it can be an accelerate
d storage condition for
a zone. The storage condition has nothing to do with Long term / Intermediate /
Accelerated storage
condition. These word should not be the part of the description of a storage con
dition.
Follow the menu path IMG >> Quality Management >> Stability Study >> Basic Data
>> Define storage
Conditions
Generated by Jive on 2016-07-26Z
8
Stability Study with SAP Quality Management
1.7. Physical Sample Storage Location
You need to define the physical sample storage location for physical sample.
Definition: The stability chamber in which the samples are placed is called Phys
ical sample storage location.
E.g. it has nothing to do with the material storage location. It is the identifi
cation of a stability chamber.
It is helpful to have tractability of the sample
Follow the menu path IMG >> Quality Management >> Quality Inspection >> Sample M
anagement >> Define
Physical-Sample Locations
It is a plant specific configuration. You need to first define a plant for which
you are maintaining the stability
chamber. If you have a common chamber for multiple plant. You need to create the
same stability chamber for
each of plants.
Generated by Jive on 2016-07-26Z
9
Stability Study with SAP Quality Management
2. Master Data
2.1. Maintenance Strategy
You need to create a maintenance strategy for stability study. It is the most im
portant master data for the
stability study
Run Transaction IP11 or follow the menu path SAP Menu >> Logistics >> Quality Ma
nagement >> Stability
Study >> Stability Planning >> Strategy >> Edit (IP11)
The important part is the scheduling indicator. You need to select 1-Time-Key da
te to get the schedule on
key date. E.g. if you start the study on 12.07.2016 and you have 1month, 2 month
s, 3 months, 6 months, 9
months and 12 months stations, you will get 12.08.2016(1month), 12.09.2016(2mont
hs), 12.10.2016(3months),
12.01.2017(6months), 12.04.2017(9months) and 12.07.2017(12months). For remaining
two options (time and
2-time-factory calendar) you will not get the stations on the key date.
Generated by Jive on 2016-07-26Z
10
Stability Study with SAP Quality Management
Generated by Jive on 2016-07-26Z
11
Stability Study with SAP Quality Management
2.2. Material Master and Inspection type
You must assign and activate inspection type for a material which you intended f
or stability study.
You must assign inspection type 16 (Inspection lot for Storage condition), 1601
(Initial inspection lot) and 1602
(Manual or additional inspection lot) or custom inspection types substitute for
each one.
Generated by Jive on 2016-07-26Z
12
Stability Study with SAP Quality Management
2.3. Inspection plan
You need to have inspection plan for inspection processing for stability study
I hope everyone on this forum for Quality Management must be aware of the inspec
tion plan. So, I will not
describe the same in details. But, the following points must be considered.
No Dynamic Modification Rule work for stability Study as the task list usage is
3-universal
The inspection plan should have sample drawing procedure in order to support the
physical samples
The most important part is to assign the maintenance strategy and maintenance pa
ckage to the
inspection plan which decide the planned stations for the particular storage con
dition.
Generated by Jive on 2016-07-26Z
13
Stability Study with SAP Quality Management
2.4. Assign Maintenance Packages to the Inspection
plan
You must assign a maintenance strategy and required maintenance package to an in
spection plan in order to
create maintenance plan with the required planned stations.
1. E.g. you need to assign maintenance packages 3 Months, 6 Months, 9 Months and
12 Months for an
Accelerated stability study.
Also, you need to create different inspection plan for each strategy. E.g. if yo
u are planning for Long
term(3,6,9,12,18,24,36 months), intermediate(3,6,9,12,18,24 months) and Accelera
ted (3,6,9,12 months)
strategy. You need to create three different inspection plan for each one of the
m.
Run the transaction CWBQM or follow the menu path SAP Menu >> Logistics >> Quali
ty Management >>
Stability Study >> Stability Planning >> Inspection Planning >> Workbench (CWBQM
)
You get the first screen as follows to select either Header area or Operations a
rea for the inspection plan
workbench
Select always Q_TSK_000000000010 - Insp. plan, mater.-routing alloc., lock and c
lick on Continue
You get the below screen.
Enter the selection criteria and execute. E.g. Material, Plant, Task list type (
always Q-Inspection plan), Group
and Group Counter and execute (load task lists)
Generated by Jive on 2016-07-26Z
14
Stability Study with SAP Quality Management
You get the below screen. Here you have all the inspection plan against the sele
ction criteria. Select the
required inspection plan and scroll horizontally to your right.
Generated by Jive on 2016-07-26Z
15
Stability Study with SAP Quality Management
You scroll horizontally to your right till you get see the column for Strategy l
ike below screen. Enter/choose the
same maintenance strategy (e.g. QSTABI) for all the selected inspection plan(s).
Generated by Jive on 2016-07-26Z
16
Stability Study with SAP Quality Management
Now Follow the menu path Task List >> Maintenance Packages like below screen.
Generated by Jive on 2016-07-26Z
17
Stability Study with SAP Quality Management
The system will navigates to below screen in which you can assign the required p
ackages. You can assign and
navigate to other selected inspection plans using navigation buttons on the tran
saction tool bar.
Generated by Jive on 2016-07-26Z
18
Stability Study with SAP Quality Management
Generated by Jive on 2016-07-26Z
19
Stability Study with SAP Quality Management
Generated by Jive on 2016-07-26Z
20
Stability Study with SAP Quality Management
Also, dont forget to save and exit from the transaction.
This will add maintenance packages in your inspection plans. That decides the pl
anned stations for the storage
condition for which the inspection plan is assigned.
3. Stability Study Processing
The standard stability study process flow is as below
Generated by Jive on 2016-07-26Z
21
Stability Study with SAP Quality Management
3.1. Create Quality Notification
Use Transaction QM01 and Notification Type QS-Stability Study with Material
Generated by Jive on 2016-07-26Z
22
Stability Study with SAP Quality Management
In Below screen, Enter notification type QS.
In Below Screen, Enter the material and plant and hit enter key. The system will
open up the batch number
field
Generated by Jive on 2016-07-26Z
23
Stability Study with SAP Quality Management
In below screen, enter the batch number and Save
Generated by Jive on 2016-07-26Z
24
Stability Study with SAP Quality Management
Notification is created
Generated by Jive on 2016-07-26Z
25
Stability Study with SAP Quality Management
3.2. Create Initial Sample
Open the notification in change using transaction QM02
Generated by Jive on 2016-07-26Z
26
Stability Study with SAP Quality Management
In Below screen, enter the notification number and enter
In below screen, in Action Box, Click on Action Create Initial Sample
The system should show a pop-up dialog box for initial sample. On the pop-up dia
log box, Enter Primary
Packaging, Sample Container and the sample quantity (the total sample quantity f
or the stability study i.e. total
Generated by Jive on 2016-07-26Z
27
Stability Study with SAP Quality Management
of all the storage condition sample). Storage Condition and Storage Location is
optional as this is the initial
sample. If the product required to be kept in particular storage condition (e.g.
product need refrigeration) then
you must define the same as the product need to be kept inside the refrigerator
before placing into the stability
chamber.
Click on Save and change notification. This will create the initial sample for t
he stability study.
Note: You must create initial sample when you decide to do stability study as th
e system do not allow
to start the study in back date.
3.3. Confirm Initial Sample
In below screen, in Action Box, Click on Action Confirm Initial Sample
Generated by Jive on 2016-07-26Z
28
Stability Study with SAP Quality Management
The system should show a pop-up dialog box for the sample confirmation with all
the details.
Generated by Jive on 2016-07-26Z
29
Stability Study with SAP Quality Management
Click on save and change notification button. The system should show another pop
-up dialog box for physical
sample drawing confirmation
Click on continue to confirm the sample
Generated by Jive on 2016-07-26Z
30
Stability Study with SAP Quality Management
3.4. Create Initial Inspection Lot
In below screen, in Action Box, Click on Action Initial test Inspection lot
The system shows a pop-up dialog box for specification selection. Enter the Grou
p and Group
counter and put Initial Inspection lot as Short Text and click on Create Inspectio
n lot.
Generated by Jive on 2016-07-26Z
31
Stability Study with SAP Quality Management
The System show another pop-up dialog box for flexible specification selection s
creen where you can select
the required test as well as change the specification of the test as per the req
uirement and click on copy to
create inspection lot. Alternatively, you can click on copy all button to copy a
ll the specification if you are using
the required inspection plan.
The system should create the inspection lot. You need to process the inspection
lot separately.
Generated by Jive on 2016-07-26Z
32
Stability Study with SAP Quality Management
I hope everyone on this forum for Quality Management must be aware of the inspec
tion lot result recording and
Usage Decision. So, I will not describe the same in details.
3.5. Complete Initial Inspection Lot
You need not to perform the this action from the Quality Notification action box
if you use the standard UD code
provided for making usage decision for stability study as those UD code contains
the follow up action to do this.
You can see the same in the below screen shots.
Generated by Jive on 2016-07-26Z
33
Stability Study with SAP Quality Management
Generated by Jive on 2016-07-26Z
34
Stability Study with SAP Quality Management
If you use custom UD codes without follow up action, you need to click on the ac
tion box for Complete Initial
Test as per below screenshot
Generated by Jive on 2016-07-26Z
35
Stability Study with SAP Quality Management
3.6. Define Storage Conditions
In below screen, in Action Box, Click on Action Specify Storage Conditions
The System shows a pop-up dialog box for defining the storage conditions. Select
the required Storage
Conditions and distribute the sample quantity for the selected storage condition
s. Click on save and change
notification button to save the same.
Generated by Jive on 2016-07-26Z
36
Stability Study with SAP Quality Management
3.7. Create Testing Schedule
In below screen, in Action Box, Click on Action Create Testing Schedule
Generated by Jive on 2016-07-26Z
37
Stability Study with SAP Quality Management
The system shows a pop-up dialog box for create test schedule. Select the Mainte
nance Strategy and Define
the Scheduling period and Click on create Testing schedule, save and change the
notification button to create
the testing schedule.
Note: You must define the scheduling period greater than last station. E.g. If y
ou are planning for 3, 6,
9, 12, 18, 24, 36, 48, 60 Months then you must put value greater than 60 Months.
In this case I am putting 72 Months (I always ask the client for the maximum per
iod for stability study
and ask them to standardize a fixed value to be defined as scheduling period to
avoid any confusion)
E.g. if client say that they do stability maximum up to 60 Months, I tell them t
o standardize 72 Months. If
they say 36 Months then I tell to standardize 48 Months.
Generated by Jive on 2016-07-26Z
38
Stability Study with SAP Quality Management
Now, you should be have a question in your mind Why????
Because, if you are using time-key date in maintenance strategy then system will
not create last station
in maintenance schedule.
E.g. If you have 3, 6, 9, 12, 18, 24, 36, 48, 60 months stations and if you defi
ne the scheduling period as
60 Months. The System will create the test schedule till 48 months and will not
show 60 Months station.
Because the system consider 1 month = 30 days and if you calculate 30 days x 60
Months = 1800 days.
So, if your start date is 12.07.2016 then you are scheduling up to 1800 days i.e
. till date 16.06.2021.
But as per the time-key date strategy it should be 12.07.2021 which is greater t
han 16.06.2021. So, the
system do not create schedule for 60 months station. Although, the system will g
enerate the inspection
lot for 60 Months when 12.07.2021 date is reached. So, it is up to you to follow
the same or not.
But, I recommend to use my suggestion if you are using the scheduling data for r
eporting purpose.
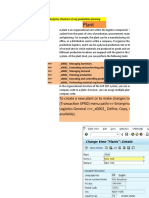
3.8. Change Testing Schedule (Assign Inspection Plan)
In below screen, in Action Box, Click on Action Change Testing Schedule (here I
have changed the
text for the action to fit to my clients requirement)
The system shows a pop-up dialog box asking for Do you want to edit the testing s
chedule for
the study and return to the study? click on Yes.
Generated by Jive on 2016-07-26Z
39
Stability Study with SAP Quality Management
The system navigates to Change Maintenance Plan screen.
Here, you select the Maintenance Item (each maintenance item represents a storag
e conditions selected in
previous step) and click on Item Tab
Generated by Jive on 2016-07-26Z
40
Stability Study with SAP Quality Management
In below screen, you can see the details of the maintenance item.
Click on Cycle Item, you will see nothing in that tab.
Generated by Jive on 2016-07-26Z
41
Stability Study with SAP Quality Management
Come back to Item tab. Enter Group and Group Counter (basically assign the inspe
ction plan to a storage
condition) then navigate to cycle item tab to see the assigned time points
Generated by Jive on 2016-07-26Z
42
Stability Study with SAP Quality Management
Generated by Jive on 2016-07-26Z
43
Stability Study with SAP Quality Management
Now, use the Navigation buttons to navigate between items.
Generated by Jive on 2016-07-26Z
44
Stability Study with SAP Quality Management
Generated by Jive on 2016-07-26Z
45
Stability Study with SAP Quality Management
Generated by Jive on 2016-07-26Z
46
Stability Study with SAP Quality Management
Now, Click on Save on below screen.
The System saves the maintenance plan and navigates back to the notification scr
een.
Generated by Jive on 2016-07-26Z
47
Stability Study with SAP Quality Management
3.9. Store Stability Samples
In below screen, in Action Box, Click on Action Store Stability Sample
Generated by Jive on 2016-07-26Z
48
Stability Study with SAP Quality Management
The system shows a pop-up dialog box for Store stability Sample. Select all the
storage conditions and choose
the sample location and click on save and change the notification button to stor
e the sample.
Generated by Jive on 2016-07-26Z
49
Stability Study with SAP Quality Management
3.10. Complete Test Scheduling
In below screen, in Action Box, Click on Action Complete Test Scheduling
Generated by Jive on 2016-07-26Z
50
Stability Study with SAP Quality Management
The System shows a pop-up dialog box asking for Do you want to set the status "Te
st Scheduling
Completed" and return to the stability study? click on yes.
The system perform the action of completion of test scheduling.
3.11. Define Start Date of Study
In below screen, in Action Box, Click on Action Define Start Date of Study
Generated by Jive on 2016-07-26Z
51
Stability Study with SAP Quality Management
The system shows a pop-up dialog box for defining start of cycle. Check / Change
date and click on Save and
Change notification button to complete the action.
Note: The dates are very important for the stability study. Below is the order f
or the different dates. If
you deviate from the order then you will mess up the stability study notificatio
n and you may need to
close the same and create new one for the same.
Initial Sample Creation Date <= Initial Sample Confirmation Date <= Store Stabil
ity Sample Date <= Start
Date of Study.
You cannot define the start date of the study as back date (if initial sample is
created on current date) /
Future date
3.12. Deadline Monitoring for Due Stations
You need to schedule a daily background Job with Transaction IP30 with proper va
riant to create inspection lot
for the stability.
You need to process the generated inspection lot for Result recording and Usage
Decision.
Generated by Jive on 2016-07-26Z
52
Stability Study with SAP Quality Management
I hope everyone on this forum for Quality Management must be aware of scheduling
a background job as well
as inspection lot processing. So, I will not describe the same in details.
3.13. Complete Study and Reporting
Once all the stations are completed, you can use action box item. Complete Stabi
lity Study to complete the
study.
Also, you can Use Actions Display Objects for Study to see the object linked to
the stability study notification.
It shows Quality Notification, Initial Physical Sample, Initial Inspection lot,
Physical Samples for Storage
Conditions and Their inspection lots and Maintenance Plan.
You can use the action Display Stability History to display all the result recor
ding history till date for the
stability study. This action is also available while result recording of the sta
bility study inspection lot. There is a
command button below the sample.
There are many other things that you can customize as per your requirements.
I hope this document will be helpful. There are many possibility in the stabilit
y study with SAP QM. You can
explore the same.
Regards,
Dipeshkumar Bhavsar
Generated by Jive on 2016-07-26Z
53
Vous aimerez peut-être aussi
- Stability Study With SAP Quality ManagementDocument53 pagesStability Study With SAP Quality ManagementSandeep89% (9)
- Stability StudyDocument18 pagesStability StudyPramod ShettyPas encore d'évaluation
- Sap PP SyllabusDocument4 pagesSap PP SyllabusZaheer AhamedPas encore d'évaluation
- SAP Quality Management Course SyllabusDocument3 pagesSAP Quality Management Course Syllabusyash50% (2)
- Production Kitting Using WM-PP Interface: Setting Up SAP WM PP Interface Is Covered in This Online SAP WM Training CourseDocument6 pagesProduction Kitting Using WM-PP Interface: Setting Up SAP WM PP Interface Is Covered in This Online SAP WM Training Courseanon_672875766Pas encore d'évaluation
- Automatic Batch Determination Shelf LifeDocument18 pagesAutomatic Batch Determination Shelf LifeKhalil EL KARAMAPas encore d'évaluation
- Global Sap Kanban ProcessDocument52 pagesGlobal Sap Kanban ProcessAbhijitPas encore d'évaluation
- Active Ingredient Management and Batch BalancingDocument24 pagesActive Ingredient Management and Batch Balancingmrivo100% (1)
- Implementing Integrated Business Planning: A Guide Exemplified With Process Context and SAP IBP Use CasesD'EverandImplementing Integrated Business Planning: A Guide Exemplified With Process Context and SAP IBP Use CasesPas encore d'évaluation
- SAP PP Serial Number ManagementDocument14 pagesSAP PP Serial Number Managementhoney50% (2)
- Handling Unit Management in SAP MMDocument15 pagesHandling Unit Management in SAP MMRULER KINGSPas encore d'évaluation
- LO710-V46c-En Problem Processing With Quality NotificationsDocument318 pagesLO710-V46c-En Problem Processing With Quality NotificationsWinston Kong Gao100% (1)
- Planning With Shelflife in PPDSDocument3 pagesPlanning With Shelflife in PPDSJintasit HearPas encore d'évaluation
- SAP PP REM Profile AssistanceDocument19 pagesSAP PP REM Profile AssistanceKanapon GunpromPas encore d'évaluation
- Sap PP Vijay ResumeDocument2 pagesSap PP Vijay Resumesonu87000% (1)
- Name of Candidate SAP - PP Functional Consultant Professional SummaryDocument5 pagesName of Candidate SAP - PP Functional Consultant Professional SummaryVasuPas encore d'évaluation
- Notification - Sending Mail Process FlowDocument14 pagesNotification - Sending Mail Process FlowshafisfjPas encore d'évaluation
- QM-WM Interface IssuesDocument28 pagesQM-WM Interface IssuesJagannadh Pradeep Ivaturi100% (2)
- Multilevel Subcontracting Process in SAP S4 HANADocument15 pagesMultilevel Subcontracting Process in SAP S4 HANASiva Thunga100% (1)
- MRP With HanaDocument9 pagesMRP With HanaNeeraj KumarPas encore d'évaluation
- Sap QM CFR Audit Management PDFDocument28 pagesSap QM CFR Audit Management PDFAnywhere100% (1)
- Key Data Structure List (KDS) : PurposeDocument8 pagesKey Data Structure List (KDS) : PurposeAditya100% (2)
- SAP Batch Derivation-Bath DeterminationDocument27 pagesSAP Batch Derivation-Bath Determinationrakesh1618Pas encore d'évaluation
- EHP6 for SAP ERP 6.0 MRP LIVE Process DocumentationDocument18 pagesEHP6 for SAP ERP 6.0 MRP LIVE Process Documentationrahul shendage100% (1)
- HUMDocument35 pagesHUMPranaya Barik100% (1)
- Batch Specific Active Ingredient ManagementDocument35 pagesBatch Specific Active Ingredient Managementrakeshp73Pas encore d'évaluation
- Sample SD BBPDocument30 pagesSample SD BBProlsonlewisPas encore d'évaluation
- Restrictions For SAP CWM in SAP S/4HANADocument11 pagesRestrictions For SAP CWM in SAP S/4HANATuấn Nguyễn Khắc MinhPas encore d'évaluation
- Stock Transfer Between Same PlantDocument25 pagesStock Transfer Between Same PlantamitjpPas encore d'évaluation
- New SAP Manufacturing ERP Features and Functions For Discrete Manufacturers - Execution StepsDocument34 pagesNew SAP Manufacturing ERP Features and Functions For Discrete Manufacturers - Execution Stepsliram299188Pas encore d'évaluation
- PP BBPDocument20 pagesPP BBPSiva Sankar Mohapatra100% (1)
- Batch Determination PPDocument13 pagesBatch Determination PPrvk386Pas encore d'évaluation
- QM OverviewDocument79 pagesQM OverviewRakesh MusalePas encore d'évaluation
- SAP Handling Unit Management Integration With Production PlanningDocument23 pagesSAP Handling Unit Management Integration With Production Planningsjobsvn100% (1)
- Sap PP Enterprise Structure 1Document24 pagesSap PP Enterprise Structure 1NASEER ULLAHPas encore d'évaluation
- RN Ewm 900 enDocument16 pagesRN Ewm 900 enAakriti ChPas encore d'évaluation
- EWM - Wave Management - SAP BlogsDocument18 pagesEWM - Wave Management - SAP BlogsAadityaa ChintapalliPas encore d'évaluation
- Handling Unit Management in SAP Packing During Outbound ProcessDocument48 pagesHandling Unit Management in SAP Packing During Outbound ProcessManibudh Sankasem0% (1)
- Alternate and Parallel Sequence in RoutingDocument27 pagesAlternate and Parallel Sequence in Routingkrishna100% (1)
- REM ProfilesDocument9 pagesREM ProfilesNishant PatilPas encore d'évaluation
- Warehouse Management With SAP EWMDocument695 pagesWarehouse Management With SAP EWMDurga SPas encore d'évaluation
- WM-QM Interface in SAP WMDocument8 pagesWM-QM Interface in SAP WMsumitjain_25Pas encore d'évaluation
- Sap MM Business Blue Print SampleDocument79 pagesSap MM Business Blue Print SampleNiranjan BeheraPas encore d'évaluation
- Manage QM Data in SAP Material MasterDocument40 pagesManage QM Data in SAP Material MasterRohit shahi100% (1)
- Sap QM End User ManualDocument70 pagesSap QM End User ManualDemoPas encore d'évaluation
- Check for Certificate Receipt in SAP QMDocument1 pageCheck for Certificate Receipt in SAP QMganeshPas encore d'évaluation
- The Up & Away Advisors’ Guide to Implementing and Executing Sap’s Vehicle Management SystemD'EverandThe Up & Away Advisors’ Guide to Implementing and Executing Sap’s Vehicle Management SystemPas encore d'évaluation
- Step by Step Sap QM End User ManualDocument63 pagesStep by Step Sap QM End User ManualAditya DeshpandePas encore d'évaluation
- Calibration CertificateDocument29 pagesCalibration CertificateSrinivasa Rao MullapudiPas encore d'évaluation
- Alert and MacroDocument11 pagesAlert and MacroDennis RoyPas encore d'évaluation
- SAP QM Questionnaire for BlueprintDocument10 pagesSAP QM Questionnaire for BlueprintchameladeviPas encore d'évaluation
- Automotive QM Manual Config GuideDocument342 pagesAutomotive QM Manual Config Guidesureva65Pas encore d'évaluation
- 03.inbound POSCDocument5 pages03.inbound POSCmjPas encore d'évaluation
- SAP Batch Management OverviewDocument70 pagesSAP Batch Management Overviewlibran_15Pas encore d'évaluation
- C Ewm 94 Study Guide and How To Crack Ex PDFDocument5 pagesC Ewm 94 Study Guide and How To Crack Ex PDFmaniPas encore d'évaluation
- SAP Repetitive Manufacturing With Reporting Point BackflushDocument12 pagesSAP Repetitive Manufacturing With Reporting Point BackflushDevidas Karad100% (1)
- Material Quantity CalculationDocument33 pagesMaterial Quantity Calculationbalu4indians100% (1)
- Sap QM IDI Interface With LIMSDocument21 pagesSap QM IDI Interface With LIMSHTE100% (1)
- Warehouse Management Systems A Complete Guide - 2019 EditionD'EverandWarehouse Management Systems A Complete Guide - 2019 EditionPas encore d'évaluation
- EulaDocument3 pagesEulaBrandon YorkPas encore d'évaluation
- UnconsciousnessDocument91 pagesUnconsciousnessFitri Amelia RizkiPas encore d'évaluation
- Fortiap S Fortiap w2 v6.0.5 Release Notes PDFDocument10 pagesFortiap S Fortiap w2 v6.0.5 Release Notes PDFtorr123Pas encore d'évaluation
- Fortiap v6.0.5 Upgrade Table PDFDocument2 pagesFortiap v6.0.5 Upgrade Table PDFtorr123Pas encore d'évaluation
- Management of Unconscious PatientDocument51 pagesManagement of Unconscious PatientEmenike Donald Ejieji50% (2)
- Vishnu Priya Anguraj Tsmu Grp-14Document39 pagesVishnu Priya Anguraj Tsmu Grp-14torr123100% (1)
- Dr. Praveen Kumar DoddamaniDocument81 pagesDr. Praveen Kumar Doddamanitorr123100% (2)
- DGFT IndiaDocument33 pagesDGFT IndiaKarthik RedPas encore d'évaluation
- Presented By: Dr. Vishal Bathma Asst. Professor Community Medicine PCMS&RCDocument42 pagesPresented By: Dr. Vishal Bathma Asst. Professor Community Medicine PCMS&RCtorr123Pas encore d'évaluation
- Hypertension: PharmacotherapyDocument23 pagesHypertension: Pharmacotherapytorr123Pas encore d'évaluation
- Understanding Parkinson's DiseaseDocument52 pagesUnderstanding Parkinson's Diseasetorr123Pas encore d'évaluation
- Cranial Osteomyelitis Diagnosis and Treatment OptionsDocument5 pagesCranial Osteomyelitis Diagnosis and Treatment OptionsRajanish Kumar50% (2)
- EEM Best PracticesDocument26 pagesEEM Best Practicestorr123Pas encore d'évaluation
- Neoadjuvant Therapies for Gastric CancerDocument64 pagesNeoadjuvant Therapies for Gastric Cancertorr123Pas encore d'évaluation
- Bonetumoursandprinciplesoflimbsalvagesurgery 140504075940 Phpapp01Document87 pagesBonetumoursandprinciplesoflimbsalvagesurgery 140504075940 Phpapp01torr123Pas encore d'évaluation
- DGFT IndiaDocument33 pagesDGFT IndiaKarthik RedPas encore d'évaluation
- India Bank Recruitment of Specialist Officers EngDocument25 pagesIndia Bank Recruitment of Specialist Officers Engajay kumarPas encore d'évaluation
- Contactdermatitis 130920140849 Phpapp01Document65 pagesContactdermatitis 130920140849 Phpapp01torr123Pas encore d'évaluation
- Hspowerpoint 140424161028 Phpapp01Document25 pagesHspowerpoint 140424161028 Phpapp01torr123Pas encore d'évaluation
- HaemodialysisDocument98 pagesHaemodialysisKo Zin100% (1)
- CP R75.40VS SmartViewTracker AdminGuideDocument31 pagesCP R75.40VS SmartViewTracker AdminGuidetorr123Pas encore d'évaluation
- Administration Guide: 17 February 2013Document303 pagesAdministration Guide: 17 February 2013torr123Pas encore d'évaluation
- India Bank Recruitment of Specialist Officers EngDocument25 pagesIndia Bank Recruitment of Specialist Officers Engajay kumarPas encore d'évaluation
- Liver Diseases With Pregnancy3603Document138 pagesLiver Diseases With Pregnancy3603torr123Pas encore d'évaluation
- Afib574903profslideshareversiondaspeakerbureauslidedeck10 131017131248 Phpapp02Document31 pagesAfib574903profslideshareversiondaspeakerbureauslidedeck10 131017131248 Phpapp02torr123Pas encore d'évaluation
- CP R75.40VS SmartViewTracker AdminGuideDocument31 pagesCP R75.40VS SmartViewTracker AdminGuidetorr123Pas encore d'évaluation
- Preventing AFib Related Strokes SlideDocument48 pagesPreventing AFib Related Strokes Slidetorr123Pas encore d'évaluation
- Physiological Aspect of Foods & Drugs As Aphrodisiacs: Guided By-Presented byDocument21 pagesPhysiological Aspect of Foods & Drugs As Aphrodisiacs: Guided By-Presented bytorr123Pas encore d'évaluation
- Preventing AFib Related Strokes SlideDocument48 pagesPreventing AFib Related Strokes Slidetorr123Pas encore d'évaluation
- CCNP Work BookDocument94 pagesCCNP Work Booktorr123100% (1)
- 02 CV Tracking SETUPDocument10 pages02 CV Tracking SETUPAbraham SegundoPas encore d'évaluation
- Rust Programming Cheat Sheet: IncludesDocument2 pagesRust Programming Cheat Sheet: IncludesmutecamelPas encore d'évaluation
- 8-Port Spectrum Analyzer with Carrier MonitoringDocument2 pages8-Port Spectrum Analyzer with Carrier MonitoringarzeszutPas encore d'évaluation
- Interfacing LCD With F PgaDocument22 pagesInterfacing LCD With F PgaadityagulkotwarPas encore d'évaluation
- Lab Assignment #1 Introduction To Geographic Data 1. LogisticsDocument16 pagesLab Assignment #1 Introduction To Geographic Data 1. LogisticsIgo CandeeiroPas encore d'évaluation
- Xcams - EBookDocument34 pagesXcams - EBookmerarib342Pas encore d'évaluation
- IBM 3500 M2 User's GuideDocument126 pagesIBM 3500 M2 User's GuidekelvislonghiPas encore d'évaluation
- Week 5 Lecture MaterialDocument97 pagesWeek 5 Lecture MaterialSmita Chavan KhairnarPas encore d'évaluation
- Ramdump Venus 2022-01-04 10-22-26 PropsDocument15 pagesRamdump Venus 2022-01-04 10-22-26 PropsYousles maniaPas encore d'évaluation
- 1 s2.0 S2212017312004604 MainDocument9 pages1 s2.0 S2212017312004604 MainTHAPELO PROSPERPas encore d'évaluation
- AI Technologies Transform Military OperationsDocument20 pagesAI Technologies Transform Military OperationsKady YadavPas encore d'évaluation
- Order 7870856 Post SIEM Implementation FinalDocument19 pagesOrder 7870856 Post SIEM Implementation FinalQuinter WandiaPas encore d'évaluation
- Android SQLite Database TutorialDocument6 pagesAndroid SQLite Database TutorialJean claude onanaPas encore d'évaluation
- Studio 5000: Ordering GuideDocument3 pagesStudio 5000: Ordering Guideluis.chuquimiaPas encore d'évaluation
- Catia V5 Tutorial Work BookDocument17 pagesCatia V5 Tutorial Work Bookavaakayi100% (2)
- Virtuoso Foundation IP Characterization PDFDocument7 pagesVirtuoso Foundation IP Characterization PDFMike CainPas encore d'évaluation
- ml310 Vxworks QuickstartDocument32 pagesml310 Vxworks Quickstartjumbo_hydPas encore d'évaluation
- Mysql Security Excerpt 5.1 enDocument85 pagesMysql Security Excerpt 5.1 enEduardo De AmatPas encore d'évaluation
- Operator ManualDocument84 pagesOperator Manualswoessner1100% (1)
- GridWorld AP CompScienceDocument129 pagesGridWorld AP CompScienceJoseph VandegriftPas encore d'évaluation
- R24 System Diagram: AMD OntarioDocument36 pagesR24 System Diagram: AMD OntarioRicardo SilvaPas encore d'évaluation
- Question 1:-Answer True or False For The Following StatementsDocument5 pagesQuestion 1:-Answer True or False For The Following StatementsMony JosephPas encore d'évaluation
- Oracle SQL Developer VMDocument3 pagesOracle SQL Developer VMKhoa DiepPas encore d'évaluation
- Triconex DocumentDocument51 pagesTriconex DocumentsendutdutPas encore d'évaluation
- Optimal performance internet with Global InternetDocument2 pagesOptimal performance internet with Global InternetDony KurniawanPas encore d'évaluation
- Examen Analista FuncionalDocument7 pagesExamen Analista FuncionalAlexia HaedoPas encore d'évaluation
- DX DiagDocument30 pagesDX DiagPepito Pepon PepoidePas encore d'évaluation
- 928TCDocument116 pages928TCJuan David DiazPas encore d'évaluation
- TTLEdit LicenseDocument2 pagesTTLEdit LicenseTrung DuongPas encore d'évaluation
- Mcq4 Questions on Spring Boot, Spring MVC, Microservices and REST APIsDocument40 pagesMcq4 Questions on Spring Boot, Spring MVC, Microservices and REST APIsAbcdPas encore d'évaluation