Académique Documents
Professionnel Documents
Culture Documents
3
Transféré par
api-296833859Copyright
Formats disponibles
Partager ce document
Partager ou intégrer le document
Avez-vous trouvé ce document utile ?
Ce contenu est-il inapproprié ?
Signaler ce documentDroits d'auteur :
Formats disponibles
3
Transféré par
api-296833859Droits d'auteur :
Formats disponibles
Promoters
- If we want an inserted gene to be expressed in a bacterium, a promoter must be added.
- In the case of insulin, the insulin gene was inserted next to the beta-galactosidase gene
(controlled by the lac operon), so they share a promoter. This means that if the bacteria are
grown in a medium containing lactose but no glucose, they would synthesise both beta-
galactosidase and insulin.
- The promoter allows RNA polymerase to bind to the DNA and identify the template strand of the
two antiparallel DNA strands.
- In eukaryotes, transcription factors may also need to bind to the promoter or RNA polymerase
before transcription can occur.
Electrophoresis
- Electrophoresis is used to separate individual charged molecules or ionic substances in a
sample on the basis of their differential movement in an electric field.
- The essential components of an electrophoretic system are:
Two electrodes, the anode (positive) and the cathode (negative) between which a voltage is
applied
A buffer solution through which the current flows and in which the ionic substances move at
constant pH.
A supporting medium which limits diffusion.
- The medium for gel electrophoresis is polyacrylamide (for proteins) or agarose (DNA).
- The movements of the charged molecules in the gel depend on:
The net charge: Negatively charged molecules move towards the anode and positively
charged molecules move towards the cathode. Highly charged molecules move faster
then those with less overall charge.
Size: The smaller the molecules, the faster they move.
Composition of the gel: The size of the pores within the gel determines the speed at
which proteins and DNA fragments move.
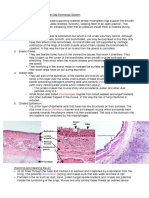
Steps for the Electrophoresis of Proteins
1. The protein sample is mixed with a buffer solution, a substance to cleave disulfide bonds, and
a tracker dye. Then, it is heated to linearise the polypeptide chains.
2. Individual samples are added to the wells using a pipette.
3. The gel (polyacrylamide) is positioned with the wells closest to the cathode. Most proteins have
a net negative charge. Apply the voltage.
4. Fix and stain the gel.
Separated proteins are visible as bands.
5. Examine the results. The position of the bands can be compared with molecular mass
standards to determine the size of each band.
- Amino acids have R groups with different charges (e.g. -COO - and -NH3 +). This allows them to
undergo electrophoresis.
- The haemoglobin of people who have sickle cell anaemia will have a less negative charge
because valine is replaced by glutamic acid. So, the molecules of haemoglobin with the
defective beta-goblin chain will not move as far through the gel as those normal ones.Thus, the
sickle cell allele can be checked for with electrophoresis.
Steps for the Electrophoresis of DNA
1. Prepare the gel (agarose).
2. Prepare the samples and add a tracking dye.
3. Load the samples onto the gel.
4. Load the DNA markers: these standards are of known size and are added to the first and last
wells of gel.
After electrophoresis the relative position of the bands of known size can be used to prepare a
calibration curve.
5. Carry out electrophoresis for 30-60 minutes until the tracking dye has migrated across 80% of
the gel.
6. Examine the results by transferring the gel to a UV transilluminator.
7. Extract only DNA bands of interest.
- DNA fragments are negatively charged due to the charged phosphate groups attached to them.
Thus, the DNA fragments move towards the anode.
Vous aimerez peut-être aussi
- Selective Advantage Natural Selection Selection Pressure: Speciation SpeciesDocument2 pagesSelective Advantage Natural Selection Selection Pressure: Speciation Speciesapi-296833859Pas encore d'évaluation
- TranscriptoneDocument2 pagesTranscriptoneapi-296833859Pas encore d'évaluation
- Negative Effects of Growing Herbicide Resistant Crops IncludeDocument3 pagesNegative Effects of Growing Herbicide Resistant Crops Includeapi-296833859Pas encore d'évaluation
- Selection PressuresDocument1 pageSelection Pressuresapi-296833859Pas encore d'évaluation
- Selective AdvantagesDocument2 pagesSelective Advantagesapi-296833859Pas encore d'évaluation
- Background Genes Progeny TestingDocument1 pageBackground Genes Progeny Testingapi-296833859Pas encore d'évaluation
- Inbreeding Interbreeding Outbreeding Inbreeding DepressionDocument1 pageInbreeding Interbreeding Outbreeding Inbreeding Depressionapi-296833859Pas encore d'évaluation
- Photophosphorylation:: PhotoactivationDocument1 pagePhotophosphorylation:: Photoactivationapi-296833859Pas encore d'évaluation
- The Founder Effect: Genetic DriftDocument1 pageThe Founder Effect: Genetic Driftapi-296833859Pas encore d'évaluation
- Photorespiration: Bundle Sheath CellsDocument1 pagePhotorespiration: Bundle Sheath Cellsapi-296833859Pas encore d'évaluation
- PhotolysisDocument1 pagePhotolysisapi-296833859Pas encore d'évaluation
- Limiting FactorDocument2 pagesLimiting Factorapi-296833859Pas encore d'évaluation
- Coevolution Fitness Natural Selection: Discontinuous VariationDocument2 pagesCoevolution Fitness Natural Selection: Discontinuous Variationapi-296833859Pas encore d'évaluation
- Absorbance Spectrum Action SpectrumDocument2 pagesAbsorbance Spectrum Action Spectrumapi-296833859Pas encore d'évaluation
- High Energy Bonds: Universal Intermediary MoleculeDocument2 pagesHigh Energy Bonds: Universal Intermediary Moleculeapi-296833859Pas encore d'évaluation
- Cytoplasm: GlycolysisDocument4 pagesCytoplasm: Glycolysisapi-296833859Pas encore d'évaluation
- GahhDocument1 pageGahhapi-296833859Pas encore d'évaluation
- Energy DensityDocument1 pageEnergy Densityapi-296833859Pas encore d'évaluation
- AntibioticsDocument2 pagesAntibioticsapi-296833859Pas encore d'évaluation
- Anaerobic Respiration (In The: CytoplasmDocument1 pageAnaerobic Respiration (In The: Cytoplasmapi-296833859Pas encore d'évaluation
- Gas Exchange: One (1.8 CM) Yes Yes Yes Yes No YesDocument2 pagesGas Exchange: One (1.8 CM) Yes Yes Yes Yes No Yesapi-296833859Pas encore d'évaluation
- Overcrowded Substandard Housing Low ImmunityDocument2 pagesOvercrowded Substandard Housing Low Immunityapi-296833859Pas encore d'évaluation
- Cardiac Cycle: PacemakerDocument3 pagesCardiac Cycle: Pacemakerapi-296833859Pas encore d'évaluation
- CholeraDocument1 pageCholeraapi-296833859Pas encore d'évaluation
- Passive Smoking: CarcinogensDocument3 pagesPassive Smoking: Carcinogensapi-296833859Pas encore d'évaluation
- Glycoproteins Called Mucin: DesiccationDocument2 pagesGlycoproteins Called Mucin: Desiccationapi-296833859Pas encore d'évaluation
- Organic Molecule Respiration Potential Energy Chemical Potential EnergyDocument1 pageOrganic Molecule Respiration Potential Energy Chemical Potential Energyapi-296833859Pas encore d'évaluation
- Cardiac Muscle: MyogenicDocument2 pagesCardiac Muscle: Myogenicapi-296833859Pas encore d'évaluation
- Shoe Dog: A Memoir by the Creator of NikeD'EverandShoe Dog: A Memoir by the Creator of NikeÉvaluation : 4.5 sur 5 étoiles4.5/5 (537)
- The Yellow House: A Memoir (2019 National Book Award Winner)D'EverandThe Yellow House: A Memoir (2019 National Book Award Winner)Évaluation : 4 sur 5 étoiles4/5 (98)
- The Subtle Art of Not Giving a F*ck: A Counterintuitive Approach to Living a Good LifeD'EverandThe Subtle Art of Not Giving a F*ck: A Counterintuitive Approach to Living a Good LifeÉvaluation : 4 sur 5 étoiles4/5 (5794)
- The Little Book of Hygge: Danish Secrets to Happy LivingD'EverandThe Little Book of Hygge: Danish Secrets to Happy LivingÉvaluation : 3.5 sur 5 étoiles3.5/5 (400)
- Grit: The Power of Passion and PerseveranceD'EverandGrit: The Power of Passion and PerseveranceÉvaluation : 4 sur 5 étoiles4/5 (588)
- Elon Musk: Tesla, SpaceX, and the Quest for a Fantastic FutureD'EverandElon Musk: Tesla, SpaceX, and the Quest for a Fantastic FutureÉvaluation : 4.5 sur 5 étoiles4.5/5 (474)
- A Heartbreaking Work Of Staggering Genius: A Memoir Based on a True StoryD'EverandA Heartbreaking Work Of Staggering Genius: A Memoir Based on a True StoryÉvaluation : 3.5 sur 5 étoiles3.5/5 (231)
- Hidden Figures: The American Dream and the Untold Story of the Black Women Mathematicians Who Helped Win the Space RaceD'EverandHidden Figures: The American Dream and the Untold Story of the Black Women Mathematicians Who Helped Win the Space RaceÉvaluation : 4 sur 5 étoiles4/5 (895)
- Team of Rivals: The Political Genius of Abraham LincolnD'EverandTeam of Rivals: The Political Genius of Abraham LincolnÉvaluation : 4.5 sur 5 étoiles4.5/5 (234)
- Never Split the Difference: Negotiating As If Your Life Depended On ItD'EverandNever Split the Difference: Negotiating As If Your Life Depended On ItÉvaluation : 4.5 sur 5 étoiles4.5/5 (838)
- The Emperor of All Maladies: A Biography of CancerD'EverandThe Emperor of All Maladies: A Biography of CancerÉvaluation : 4.5 sur 5 étoiles4.5/5 (271)
- Devil in the Grove: Thurgood Marshall, the Groveland Boys, and the Dawn of a New AmericaD'EverandDevil in the Grove: Thurgood Marshall, the Groveland Boys, and the Dawn of a New AmericaÉvaluation : 4.5 sur 5 étoiles4.5/5 (266)
- On Fire: The (Burning) Case for a Green New DealD'EverandOn Fire: The (Burning) Case for a Green New DealÉvaluation : 4 sur 5 étoiles4/5 (74)
- The Unwinding: An Inner History of the New AmericaD'EverandThe Unwinding: An Inner History of the New AmericaÉvaluation : 4 sur 5 étoiles4/5 (45)
- The Hard Thing About Hard Things: Building a Business When There Are No Easy AnswersD'EverandThe Hard Thing About Hard Things: Building a Business When There Are No Easy AnswersÉvaluation : 4.5 sur 5 étoiles4.5/5 (345)
- The World Is Flat 3.0: A Brief History of the Twenty-first CenturyD'EverandThe World Is Flat 3.0: A Brief History of the Twenty-first CenturyÉvaluation : 3.5 sur 5 étoiles3.5/5 (2259)
- The Gifts of Imperfection: Let Go of Who You Think You're Supposed to Be and Embrace Who You AreD'EverandThe Gifts of Imperfection: Let Go of Who You Think You're Supposed to Be and Embrace Who You AreÉvaluation : 4 sur 5 étoiles4/5 (1090)
- The Sympathizer: A Novel (Pulitzer Prize for Fiction)D'EverandThe Sympathizer: A Novel (Pulitzer Prize for Fiction)Évaluation : 4.5 sur 5 étoiles4.5/5 (121)
- Her Body and Other Parties: StoriesD'EverandHer Body and Other Parties: StoriesÉvaluation : 4 sur 5 étoiles4/5 (821)
- Plantas InvasorasDocument271 pagesPlantas InvasorasSamuel Luis SperandioPas encore d'évaluation
- Production of Glutamate and Stereospecific Flavors, (S) - Linalool and (+) - Valencene, by Synechocystis Sp. PCC6803Document7 pagesProduction of Glutamate and Stereospecific Flavors, (S) - Linalool and (+) - Valencene, by Synechocystis Sp. PCC6803Fernando IzquierdoPas encore d'évaluation
- Anp Course Plan PDFDocument10 pagesAnp Course Plan PDFpriyanka bhavsarPas encore d'évaluation
- Apomixis - AgrobiosDocument6 pagesApomixis - AgrobiosSripathy KudekalluPas encore d'évaluation
- Synopsis TemplateDocument6 pagesSynopsis TemplateZia Ashraf ChaudharyPas encore d'évaluation
- Aquaculture SpeciesDocument10 pagesAquaculture SpeciesMitch Panganiban TogniPas encore d'évaluation
- AP Biology Vocabulary TermsDocument41 pagesAP Biology Vocabulary Termsqazedc159Pas encore d'évaluation
- Fruit ScienceDocument3 pagesFruit ScienceYashraj Singh BhadoriyaPas encore d'évaluation
- AP Biology Lab Seven: Genetics of OrganismsDocument8 pagesAP Biology Lab Seven: Genetics of OrganismsCoolAsianDude67% (15)
- Alpha InterferonDocument7 pagesAlpha InterferonLAURA MARCELA BARRENECHE CALLEPas encore d'évaluation
- STS Technology and The Future of HumanityDocument62 pagesSTS Technology and The Future of HumanitymsksjsnskwjnenPas encore d'évaluation
- Life Sciences Fundamentals and Practice - IIDocument161 pagesLife Sciences Fundamentals and Practice - IIPathifnder Publication81% (21)
- Condensed Biology Georgia Eoct Study GuideDocument13 pagesCondensed Biology Georgia Eoct Study GuideTiffany Gallina100% (1)
- Report-Nepal Medical College (PVT.) Ltd. Teaching HospitalDocument1 pageReport-Nepal Medical College (PVT.) Ltd. Teaching HospitalTamang RkPas encore d'évaluation
- NCBI Blast - M22-15059-14-MU-2 - 3FDocument3 pagesNCBI Blast - M22-15059-14-MU-2 - 3FCarlitah CortinaPas encore d'évaluation
- 2012: DNA Upgrade, The Shift and AscensionDocument21 pages2012: DNA Upgrade, The Shift and AscensioniamdinesPas encore d'évaluation
- GCE - O Level BiologyDocument25 pagesGCE - O Level BiologyWayne WeePas encore d'évaluation
- HistidinemiaDocument5 pagesHistidinemiaSanya Dian FirdaPas encore d'évaluation
- The PIC Camborough Family PDFDocument44 pagesThe PIC Camborough Family PDFCristian Camilo GONZALEZ HERNANDEZPas encore d'évaluation
- Grade 8 - 1st Monthly Exams - ScienceDocument23 pagesGrade 8 - 1st Monthly Exams - ScienceInah MasubayPas encore d'évaluation
- IB Biology Genetics Questions (IB Topic 3) : For Mader 10 EditionDocument2 pagesIB Biology Genetics Questions (IB Topic 3) : For Mader 10 EditionmanrajPas encore d'évaluation
- CRISPR Methods and Protocols-Humana Press 2015Document231 pagesCRISPR Methods and Protocols-Humana Press 2015Cyril100% (2)
- Sordaria Lab ReportDocument12 pagesSordaria Lab Reportapi-305324821100% (1)
- Hardy Weinberg Problem Set KEYDocument12 pagesHardy Weinberg Problem Set KEYKapil ShindePas encore d'évaluation
- 2-Mendel Notes For WebsiteDocument29 pages2-Mendel Notes For WebsiteSamantha Isobel TumaganPas encore d'évaluation
- Evolution and Natural Selection TutorialDocument52 pagesEvolution and Natural Selection Tutorialapi-234891239Pas encore d'évaluation
- FST Handbook 2014-Final Copy 1 PDFDocument382 pagesFST Handbook 2014-Final Copy 1 PDFDelvon DownerPas encore d'évaluation
- 3 Transcription-From - DNA - To - RNADocument7 pages3 Transcription-From - DNA - To - RNACHIARA ANDREINA ALFARO PURISACAPas encore d'évaluation
- Protein Synthesis ClozeDocument3 pagesProtein Synthesis Clozeapi-309893409Pas encore d'évaluation
- Human Genome ProjectDocument22 pagesHuman Genome Projecttani84638Pas encore d'évaluation