Académique Documents
Professionnel Documents
Culture Documents
P A H P B C: Salt Solubility in Water
Transféré par
annatsubaki0 évaluation0% ont trouvé ce document utile (0 vote)
6 vues1 pageThis document discusses the solubility of different types of salts in water. It states that sodium, potassium, and ammonium salts along with nitrate and chloride salts are all soluble in water. While most sulphate and carbonate salts are soluble, exceptions include lead, barium, and calcium sulphates and all carbonates except sodium, potassium, and ammonium carbonates.
Description originale:
how to remember the solubility of salts
Titre original
Acroynm of Salts
Copyright
© © All Rights Reserved
Formats disponibles
DOCX, PDF, TXT ou lisez en ligne sur Scribd
Partager ce document
Partager ou intégrer le document
Avez-vous trouvé ce document utile ?
Ce contenu est-il inapproprié ?
Signaler ce documentThis document discusses the solubility of different types of salts in water. It states that sodium, potassium, and ammonium salts along with nitrate and chloride salts are all soluble in water. While most sulphate and carbonate salts are soluble, exceptions include lead, barium, and calcium sulphates and all carbonates except sodium, potassium, and ammonium carbonates.
Droits d'auteur :
© All Rights Reserved
Formats disponibles
Téléchargez comme DOCX, PDF, TXT ou lisez en ligne sur Scribd
0 évaluation0% ont trouvé ce document utile (0 vote)
6 vues1 pageP A H P B C: Salt Solubility in Water
Transféré par
annatsubakiThis document discusses the solubility of different types of salts in water. It states that sodium, potassium, and ammonium salts along with nitrate and chloride salts are all soluble in water. While most sulphate and carbonate salts are soluble, exceptions include lead, barium, and calcium sulphates and all carbonates except sodium, potassium, and ammonium carbonates.
Droits d'auteur :
© All Rights Reserved
Formats disponibles
Téléchargez comme DOCX, PDF, TXT ou lisez en ligne sur Scribd
Vous êtes sur la page 1sur 1
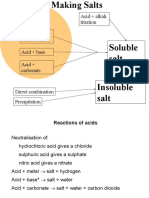
Salt Solubility in water
Sodium, potassium and All are soluble
ammonium salts
(Na+, K+, NH4+)
All are soluble
-
Nitrate salt (NO3 )
All chloride salts are soluble in water
Chloride salt (Cl -)
except PbCl2, AgCl and HgCl2
All sulphate salts are soluble in water
Sulphate salt (SO42-)
except PbSO4, BaSO4 and CaSO4
All carbonate salts are insoluble except
Carbonate salt (CO32-) Na2CO3, K2CO3 and (NH4)2CO3
Vous aimerez peut-être aussi
- Stuff I Should Know For The AP Chemistry ExamDocument2 pagesStuff I Should Know For The AP Chemistry Examhunterkiller3100% (1)
- Stuff I Should Know For The Ap Test But Do Not Know Yet: Ions ListDocument1 pageStuff I Should Know For The Ap Test But Do Not Know Yet: Ions ListScott AllredPas encore d'évaluation
- Chemical Elements Pocket Guide: Detailed Summary of the Periodic TableD'EverandChemical Elements Pocket Guide: Detailed Summary of the Periodic TablePas encore d'évaluation
- Chapter 8 SALTSDocument75 pagesChapter 8 SALTSSiti Hajar Abd HamidPas encore d'évaluation
- Notes Updates SaltsDocument32 pagesNotes Updates SaltsLim Jing YeePas encore d'évaluation
- P A H P B C: Sodium, Potassium and Ammonium Salts (Na All Are Soluble Semua LarutDocument1 pageP A H P B C: Sodium, Potassium and Ammonium Salts (Na All Are Soluble Semua LarutZekZanaPas encore d'évaluation
- Chemistry: Form 4-Chapter 8Document7 pagesChemistry: Form 4-Chapter 8Muhamad AazrilPas encore d'évaluation
- Chapter 8: SaltsDocument14 pagesChapter 8: SaltsLynn HengPas encore d'évaluation
- Solubility TableDocument1 pageSolubility TableNontando MendiePas encore d'évaluation
- S-Block Elements: Solubility TrendsDocument2 pagesS-Block Elements: Solubility TrendsHansel GoyalPas encore d'évaluation
- Solubility RulesDocument1 pageSolubility RulesAdamPas encore d'évaluation
- Chapter 8: Salts: Flow Chart of Preparation of SaltsDocument7 pagesChapter 8: Salts: Flow Chart of Preparation of SaltsPrincess Ting TingPas encore d'évaluation
- Salt AnalysisDocument4 pagesSalt AnalysisMarietta ChristopherPas encore d'évaluation
- Must Know For Chapter 9 - Salts (And C11 Qualitative Analysis)Document4 pagesMust Know For Chapter 9 - Salts (And C11 Qualitative Analysis)Chaw Wei HengPas encore d'évaluation
- Solubility Guidelines RevisedDocument1 pageSolubility Guidelines RevisedConnor AndersonPas encore d'évaluation
- Salts PreparationDocument7 pagesSalts PreparationCynthia RoneyPas encore d'évaluation
- Net Ionic EquationsDocument15 pagesNet Ionic EquationsAikaterine SmithPas encore d'évaluation
- Data Sheet Revision PDFDocument2 pagesData Sheet Revision PDFShifa RizwanPas encore d'évaluation
- Solubility Rules: Except: Ag HG, PBDocument1 pageSolubility Rules: Except: Ag HG, PBrickyPas encore d'évaluation
- Stuff I Should Know For The Ap Test But Do Not Know Yet: Ions ListDocument2 pagesStuff I Should Know For The Ap Test But Do Not Know Yet: Ions ListedecePas encore d'évaluation
- Periodic TableDocument2 pagesPeriodic TableAlliyah vidanesPas encore d'évaluation
- Salts FormationDocument19 pagesSalts FormationUrwa Abdul MannanPas encore d'évaluation
- Notes On SaltsDocument4 pagesNotes On SaltsFelix S100% (1)
- Water Solubility ChartDocument3 pagesWater Solubility ChartLuzarro BPas encore d'évaluation
- Garam Bab 8Document29 pagesGaram Bab 8ctohPas encore d'évaluation
- Wan Noor Afifah BT Wan YusoffDocument33 pagesWan Noor Afifah BT Wan YusoffThilagavathyPas encore d'évaluation
- Ariana's Rules For Solubility of Ionic Compounds in WaterDocument2 pagesAriana's Rules For Solubility of Ionic Compounds in Waterakavi1Pas encore d'évaluation
- Acids, Bases and SaltsDocument8 pagesAcids, Bases and Saltsaakashb1918Pas encore d'évaluation
- Solubility Rules: Strong & in WaterDocument1 pageSolubility Rules: Strong & in WaterChelsea MartinezPas encore d'évaluation
- Analisis Anion PDFDocument25 pagesAnalisis Anion PDFNunungNurjanahPas encore d'évaluation
- Stuff I Should Know For The Ap Test But Do Not Know Yet: Ions ListDocument1 pageStuff I Should Know For The Ap Test But Do Not Know Yet: Ions ListShubham MangalPas encore d'évaluation
- Acids - 2Document2 pagesAcids - 2Anushka ShendagePas encore d'évaluation
- Qualitative AnalysisDocument90 pagesQualitative AnalysisMahesh100% (1)
- AnionsDocument90 pagesAnionsAnish RaoPas encore d'évaluation
- 8.1 Definitions of SaltsDocument5 pages8.1 Definitions of Saltsscta94Pas encore d'évaluation
- AcidsBases - Oxides and SaltsDocument20 pagesAcidsBases - Oxides and SaltsZain AhmadPas encore d'évaluation
- 6.8 Salt Crystal and Daily LifeDocument17 pages6.8 Salt Crystal and Daily LifeNur ShahirahPas encore d'évaluation
- StuffDocument1 pageStuffrgeahreahPas encore d'évaluation
- Class X Phycw - Le.3.Document2 pagesClass X Phycw - Le.3.siddhusai999Pas encore d'évaluation
- 6 Kiseline Baze Soli 3Document42 pages6 Kiseline Baze Soli 3Emir Melisa100% (1)
- Acids and Bases SummaryDocument2 pagesAcids and Bases SummaryTan Yong KhaiPas encore d'évaluation
- Form 2 Introduction To SaltsDocument11 pagesForm 2 Introduction To Saltsemilykwamboka500Pas encore d'évaluation
- Solubility Rules2 PDFDocument1 pageSolubility Rules2 PDFFernandaIbarraVázquezPas encore d'évaluation
- Solubility RulesDocument1 pageSolubility Rulesamin_zamanPas encore d'évaluation
- Acids, Bases and Salts AKHS 2020 - Complete NotesDocument27 pagesAcids, Bases and Salts AKHS 2020 - Complete NotesKim SewoonPas encore d'évaluation
- SPM Form 5 Chemistry SaltDocument6 pagesSPM Form 5 Chemistry SaltCt Sophie PheaPas encore d'évaluation
- Final Revision Acids, Bases and Salts (Repaired) PDFDocument13 pagesFinal Revision Acids, Bases and Salts (Repaired) PDFRawan Abd ElaatyPas encore d'évaluation
- Grade 10 Chemistry Week 12 Lesson 1Document4 pagesGrade 10 Chemistry Week 12 Lesson 1nesiaroberts903Pas encore d'évaluation
- Chapter 11 - Acids, Bases, and Salts PDFDocument7 pagesChapter 11 - Acids, Bases, and Salts PDFAarush SharmaPas encore d'évaluation
- Acid Bases and Salts Igcse Chemistry 0620Document15 pagesAcid Bases and Salts Igcse Chemistry 0620Aminah ShahzadPas encore d'évaluation
- Appendix II Solubility Rules PDFDocument1 pageAppendix II Solubility Rules PDFP.shashangPas encore d'évaluation
- 10 Nature of Oxides2Document28 pages10 Nature of Oxides2James WongPas encore d'évaluation
- MetalsDocument13 pagesMetalsTvrtkoPas encore d'évaluation
- AcidsDocument3 pagesAcidsPratham GoradiaPas encore d'évaluation
- 4th Form Qualitative Analysis Sheet Summary SheetDocument2 pages4th Form Qualitative Analysis Sheet Summary SheetFrank MassiahPas encore d'évaluation
- Basic Inorganic Chemistry PHR 125: Prof. Dr. Mona BedairDocument33 pagesBasic Inorganic Chemistry PHR 125: Prof. Dr. Mona BedairAvvari AnnamaniPas encore d'évaluation
- Complete and Net Ionic Equations PracticeDocument2 pagesComplete and Net Ionic Equations PracticeActivePas encore d'évaluation
- Oxides, Acids, Bases and SaltsDocument8 pagesOxides, Acids, Bases and Salts12&13 SciencesPas encore d'évaluation