Académique Documents
Professionnel Documents
Culture Documents
Sickle Cell Anemia Assignment
Transféré par
Princess Janine Catral0 évaluation0% ont trouvé ce document utile (0 vote)
12 vues2 pagesanemia
Copyright
© © All Rights Reserved
Formats disponibles
DOCX, PDF, TXT ou lisez en ligne sur Scribd
Partager ce document
Partager ou intégrer le document
Avez-vous trouvé ce document utile ?
Ce contenu est-il inapproprié ?
Signaler ce documentanemia
Droits d'auteur :
© All Rights Reserved
Formats disponibles
Téléchargez comme DOCX, PDF, TXT ou lisez en ligne sur Scribd
0 évaluation0% ont trouvé ce document utile (0 vote)
12 vues2 pagesSickle Cell Anemia Assignment
Transféré par
Princess Janine Catralanemia
Droits d'auteur :
© All Rights Reserved
Formats disponibles
Téléchargez comme DOCX, PDF, TXT ou lisez en ligne sur Scribd
Vous êtes sur la page 1sur 2
Princess Janine B.
Catral BSChE V Assignment in Biochemical Engineering
Sickle Cell Anemia
Sickle cell anemia is a genetic disease that affects hemoglobin, the
oxygen transport molecule in the blood. The disease gets its name from to the
shape of the red blood cells under certain conditions. Some red blood cells
become sickle-shaped and these elongated cells get stuck in small blood
vessels so that parts of the body don't get the oxygen they need. Sickle cell
anemia is caused by a single code letter change in the DNA. This in turn alters
one of the amino acids in the hemoglobin protein. Valine sits in the position
where glutamic acid should be as shown in Figure 1. The valine makes the
hemoglobin molecules stick together, forming long fibers that distort the shape
of the red blood cells, and this brings on an attack.
Figure 1. Valine replaces glutamic acid resulting in a sickle cell hemoglobin
In a molecular perspective, the mutation causing sickle cell anemia is a
single nucleotide substitution (A to T) in the codon for amino acid 6 as shown in
Figure 2. The change converts a glutamic acid codon (GAG) to a valine codon
(GTG). The form of hemoglobin in persons with sickle cell anemia is referred to as
HbS. The nomenclature for normal adult hemoglobin protein is HbA1.
Figure 2. Change in codon 6 by single nucleotide substitution (A to T)
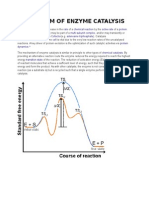
Active site of enzymes
This is a small portion of the surface of an enzyme where a specific chemical
reaction is catalyzed.
Some physical and chemical interactions occur at this site to catalyze a
certain chemical reaction for a certain enzyme.
Lock and Key Hypothesis
Enzymes are thought to operate on a geometric principle. The tertiary
and quaternary structures of an enzyme have the substrate binding sites,
which have exactly the complementary shape of the substrate
molecules.
This helps in the binding of the appropriate substrate to the active centers
just like a key fits in the keyhole as shown in Figure 3.
Figure 3. An enzyme and a substrate fits together as dictated by the lock and key hypothesis
Induced Fit Theory
Some enzymes change the shape of the active center slightly to
accommodate the substrate molecules, a process known as induced fit.
The enzyme substrate complex thus formed lowers the energy of activation
by either stressing an existing bond or correctly orienting two molecules to
favor a reaction.
The enzyme holds the substrate molecules in exactly the right position
relative to each other to facilitate the reaction due to geometric and
electrical configuration.
Vous aimerez peut-être aussi
- General Principles of CatalysisDocument12 pagesGeneral Principles of CatalysisOLUWASEGUN K AfolabiPas encore d'évaluation
- Enzymes Mbc213Document13 pagesEnzymes Mbc213prosperosamegiePas encore d'évaluation
- Cell MetabolismDocument6 pagesCell MetabolismelsayidPas encore d'évaluation
- BindingDB: Collecting Protein-Ligand Binding DataDocument12 pagesBindingDB: Collecting Protein-Ligand Binding Datadelphinas8Pas encore d'évaluation
- Chap. 6B Enzymes: - Introduction To Enzymes - How Enzymes Work - Enzyme Kinetics As An Approach ToDocument28 pagesChap. 6B Enzymes: - Introduction To Enzymes - How Enzymes Work - Enzyme Kinetics As An Approach ToRock SmithPas encore d'évaluation
- Biochemistry PDFDocument15 pagesBiochemistry PDFJaz SantosPas encore d'évaluation
- Sickle Cell AnemiaDocument9 pagesSickle Cell Anemiaتجربة أولىPas encore d'évaluation
- Biology ProjectDocument41 pagesBiology ProjectGanesan Siva67% (12)
- Enzyme ActivityDocument5 pagesEnzyme Activityjohn_flanaganPas encore d'évaluation
- Mechanism of Enzyme ActionDocument4 pagesMechanism of Enzyme ActionrohiniPas encore d'évaluation
- Principles of Pharmacology: The Pathophysiologic Basis of Drug TherapyDocument12 pagesPrinciples of Pharmacology: The Pathophysiologic Basis of Drug TherapyKayedAhmadPas encore d'évaluation
- Enzyme InhibitionDocument4 pagesEnzyme InhibitionNisha ManasPas encore d'évaluation
- Mechanism of Enzyme Catalysis MadhuDocument9 pagesMechanism of Enzyme Catalysis MadhumengelhuPas encore d'évaluation
- Proteins: What Do They Look Like and What Do They Do?Document59 pagesProteins: What Do They Look Like and What Do They Do?donaldPas encore d'évaluation
- Study On EnzymesDocument21 pagesStudy On EnzymesPragyan Kumar PradhanPas encore d'évaluation
- Mutation (Sickle Cell Anaemia)Document4 pagesMutation (Sickle Cell Anaemia)Arielle LewisPas encore d'évaluation
- Sickle Cell InfoDocument2 pagesSickle Cell InfoDeanLemanPas encore d'évaluation
- Enzyme TechnologyDocument19 pagesEnzyme TechnologySusmit NimjePas encore d'évaluation
- Summary Essential Cell Biology Chapters 4678121516 1718 and 20Document36 pagesSummary Essential Cell Biology Chapters 4678121516 1718 and 20mohamed arbi taifPas encore d'évaluation
- An Abzyme (From Antibody and Enzyme), Also Called Catmab (FromDocument6 pagesAn Abzyme (From Antibody and Enzyme), Also Called Catmab (Fromdeepali_m100% (1)
- 4 - EnzymesDocument31 pages4 - EnzymescheckmatePas encore d'évaluation
- Enzymes H2 BIOLOGY NOTESDocument3 pagesEnzymes H2 BIOLOGY NOTESSheila ChaiPas encore d'évaluation
- Group Two'S Seminar Work: Topic: Enzyme Regulation Allosteric Regulation and Models OutlineDocument13 pagesGroup Two'S Seminar Work: Topic: Enzyme Regulation Allosteric Regulation and Models OutlineOluwasegun ModupePas encore d'évaluation
- AP BIO Febraury Break With AnswersDocument12 pagesAP BIO Febraury Break With AnswersMehak BectorPas encore d'évaluation
- Michael R. Caplan Et Al - Control of Self-Assembling Oligopeptide Matrix Formation Through Systematic Variation of Amino Acid SequenceDocument9 pagesMichael R. Caplan Et Al - Control of Self-Assembling Oligopeptide Matrix Formation Through Systematic Variation of Amino Acid SequenceMdo3Pas encore d'évaluation
- 3-BIOL 101 Study Guide Quiz 3Document8 pages3-BIOL 101 Study Guide Quiz 3Suraj NaikPas encore d'évaluation
- Introduction To Enzymatic CatalysisDocument16 pagesIntroduction To Enzymatic Catalysisqodirashirquliyev0Pas encore d'évaluation
- Mechanism: Substrate BindingDocument3 pagesMechanism: Substrate BindingDharmendra SinghPas encore d'évaluation
- Problem Set 4 DK Spring 2024 Feb9Document3 pagesProblem Set 4 DK Spring 2024 Feb9Princess BellaPas encore d'évaluation
- Quiz 2Document2 pagesQuiz 2ElainaPas encore d'évaluation
- Enzymology: Theresa May Chin TanDocument20 pagesEnzymology: Theresa May Chin TanVejerla PriyankaPas encore d'évaluation
- BioOrganic Notes Part-4 FinalDocument16 pagesBioOrganic Notes Part-4 FinalAnupama GuptaPas encore d'évaluation
- Exam Iii: Only V VDocument8 pagesExam Iii: Only V Vss shinodaPas encore d'évaluation
- P4 13.4Document2 pagesP4 13.4lilahsilkPas encore d'évaluation
- 4.2 - Metabolism and Metabolic PathwaysDocument3 pages4.2 - Metabolism and Metabolic PathwayscarlPas encore d'évaluation
- Enzyme Catalysis: Mechanisms and Factors that Accelerate Reaction RatesDocument14 pagesEnzyme Catalysis: Mechanisms and Factors that Accelerate Reaction RatesAVPas encore d'évaluation
- Molecular Cell Biology Lodish 6th Edition Test BankDocument6 pagesMolecular Cell Biology Lodish 6th Edition Test BankCharles BlairPas encore d'évaluation
- Ma2003 Zainab Omolara Sanni - Practical Lesson 3Document6 pagesMa2003 Zainab Omolara Sanni - Practical Lesson 3geddy D.Pas encore d'évaluation
- Protein Denaturation: EnzymesDocument7 pagesProtein Denaturation: EnzymescharlesPas encore d'évaluation
- Bio201 Lecture 12 Enzymes - Energy & MetabolismDocument34 pagesBio201 Lecture 12 Enzymes - Energy & MetabolismPaulPas encore d'évaluation
- Module 3 Sec 2 v2Document14 pagesModule 3 Sec 2 v2James MagnoPas encore d'évaluation
- BCH 401 - Advanced EnzymologyDocument15 pagesBCH 401 - Advanced EnzymologyOLUWASEGUN K Afolabi100% (2)
- Lecture 15: Enzyme Inhibition: Biochemistry I Fall Term, 2003Document4 pagesLecture 15: Enzyme Inhibition: Biochemistry I Fall Term, 2003archerofthestarsPas encore d'évaluation
- Introduction To Enzymatic CatalysisDocument10 pagesIntroduction To Enzymatic Catalysisqodirashirquliyev0Pas encore d'évaluation
- Final Study On EnzymeDocument9 pagesFinal Study On Enzymerosariopraveen007Pas encore d'évaluation
- Enzymes RegulationDocument87 pagesEnzymes RegulationWahyu KurniawatiPas encore d'évaluation
- Aysegül Zeyno Epigenetic and Nutritient Lecture - New SunumDocument26 pagesAysegül Zeyno Epigenetic and Nutritient Lecture - New SunumzeynoleeePas encore d'évaluation
- C9e Answers Active Reading 08Document7 pagesC9e Answers Active Reading 08melissa_chow_1100% (6)
- Enzyme: Enzymes AreDocument11 pagesEnzyme: Enzymes ArePiyush BhallaPas encore d'évaluation
- Enzymes Essay Questions Answers GuideDocument3 pagesEnzymes Essay Questions Answers GuideZhiTing96Pas encore d'évaluation
- Assignment 1 - NguyenTanPhuBTBCIU16057Document4 pagesAssignment 1 - NguyenTanPhuBTBCIU16057Phú NguyễnPas encore d'évaluation
- MetabolismDocument27 pagesMetabolismAnum RasoolPas encore d'évaluation
- MutationsDocument2 pagesMutationsRajput On HuntPas encore d'évaluation
- 4.2 - Metabolism and Metabolic PathwaysDocument3 pages4.2 - Metabolism and Metabolic PathwayscarlPas encore d'évaluation
- Bai Giang Chuong 06Document36 pagesBai Giang Chuong 06Minh TâmPas encore d'évaluation
- ENZYMES NOTES (Recovered)Document10 pagesENZYMES NOTES (Recovered)zipporahwaithera404Pas encore d'évaluation
- The Genetic Basis of Haematological CancersD'EverandThe Genetic Basis of Haematological CancersSabrina TosiPas encore d'évaluation
- RAMP RESPONSE (Single Model) Ramp Parameters Values: Slope 1 Start Time 0 Initial Output 0 Tau 0.1 K 1Document7 pagesRAMP RESPONSE (Single Model) Ramp Parameters Values: Slope 1 Start Time 0 Initial Output 0 Tau 0.1 K 1Princess Janine CatralPas encore d'évaluation
- Simulink Exercise - Princess Janine B. CatralDocument4 pagesSimulink Exercise - Princess Janine B. CatralPrincess Janine CatralPas encore d'évaluation
- Nuevo Series ProloguesDocument2 pagesNuevo Series ProloguesPrincess Janine CatralPas encore d'évaluation
- Downstream Bioprocessing StepsDocument2 pagesDownstream Bioprocessing StepsPrincess Janine CatralPas encore d'évaluation
- Chapter 1Document6 pagesChapter 1Princess Janine CatralPas encore d'évaluation
- Effect of PH On Enzyme ActivityDocument1 pageEffect of PH On Enzyme ActivityPrincess Janine CatralPas encore d'évaluation
- Quiz #3: Biochemical Engineering Fall 2003Document5 pagesQuiz #3: Biochemical Engineering Fall 2003Princess Janine CatralPas encore d'évaluation
- DTI Project Proposal Edit 1Document9 pagesDTI Project Proposal Edit 1Princess Janine CatralPas encore d'évaluation
- Calibration of Rectangular NotchDocument4 pagesCalibration of Rectangular NotchPrincess Janine CatralPas encore d'évaluation
- Michaelis-Menten vs Full MechanismDocument2 pagesMichaelis-Menten vs Full MechanismPrincess Janine CatralPas encore d'évaluation
- Board Questions November 2003Document3 pagesBoard Questions November 2003Princess Janine CatralPas encore d'évaluation
- House probes DOT's plans to promote sustainable tourism in BoracayDocument12 pagesHouse probes DOT's plans to promote sustainable tourism in BoracayPrincess Janine CatralPas encore d'évaluation
- Biodegradation in A Packed Bed ColumnDocument2 pagesBiodegradation in A Packed Bed ColumnPrincess Janine CatralPas encore d'évaluation
- Online Biology BookDocument3 pagesOnline Biology BookPrincess Janine CatralPas encore d'évaluation
- Quiz #4: Biochemical Engineering Fall 2003Document5 pagesQuiz #4: Biochemical Engineering Fall 2003Princess Janine CatralPas encore d'évaluation
- PUV Modernization ProgramDocument7 pagesPUV Modernization ProgramPrincess Janine CatralPas encore d'évaluation
- 2017 Events in PHDocument5 pages2017 Events in PHPrincess Janine CatralPas encore d'évaluation
- Timeline of Marawi CrisisDocument8 pagesTimeline of Marawi CrisisPrincess Janine CatralPas encore d'évaluation
- Dengvaxia IssueDocument11 pagesDengvaxia IssuePrincess Janine CatralPas encore d'évaluation
- Wireless Power-The BasicsDocument18 pagesWireless Power-The BasicsPrincess Janine CatralPas encore d'évaluation
- Environmental Constraints and AnalysisDocument1 pageEnvironmental Constraints and AnalysisPrincess Janine CatralPas encore d'évaluation
- InsightDocument36 pagesInsightHuzaifa JalilPas encore d'évaluation
- Supercritical CO2 Extraction: A Clean Extraction MethodDocument6 pagesSupercritical CO2 Extraction: A Clean Extraction MethodPrincess Janine CatralPas encore d'évaluation
- ChE Review - Chem and Phy Prin 2Document4 pagesChE Review - Chem and Phy Prin 2Princess Janine CatralPas encore d'évaluation
- Chapter 1Document6 pagesChapter 1Princess Janine CatralPas encore d'évaluation
- Methodology MORDocument1 pageMethodology MORPrincess Janine CatralPas encore d'évaluation
- Research PracticumDocument4 pagesResearch PracticumPrincess Janine CatralPas encore d'évaluation
- Research NotesDocument2 pagesResearch NotesPrincess Janine CatralPas encore d'évaluation
- Tut. 05 - Cost Targeting FinalDocument4 pagesTut. 05 - Cost Targeting Finalpeterbookworm2014Pas encore d'évaluation
- Solved Problems in Fluid MechanicsDocument19 pagesSolved Problems in Fluid MechanicsPrincess Janine CatralPas encore d'évaluation
- NAtural LogarithmDocument15 pagesNAtural Logarithmjohn tanPas encore d'évaluation
- Second Group Project - Risk and ReturnDocument3 pagesSecond Group Project - Risk and ReturnJeff RuncornPas encore d'évaluation
- Emergent Reader Case Summary SheetDocument2 pagesEmergent Reader Case Summary Sheetapi-503192153Pas encore d'évaluation
- Radiology Imaging Techniques and PositioningDocument6 pagesRadiology Imaging Techniques and PositioningSatish PaswanPas encore d'évaluation
- Glenn Pullin'S Statement: EX344 EX344Document2 pagesGlenn Pullin'S Statement: EX344 EX344Richard HughesPas encore d'évaluation
- Tchaikovsky Piano Concerto No 1 in BB Major Op 23 Mov 1Document95 pagesTchaikovsky Piano Concerto No 1 in BB Major Op 23 Mov 1Marta FloresPas encore d'évaluation
- Componentes de Ingles Pregunta 2Document14 pagesComponentes de Ingles Pregunta 2Andres Salazar RoviraPas encore d'évaluation
- Report - Inclusive Education Experiences of Parents in MsiaDocument13 pagesReport - Inclusive Education Experiences of Parents in MsiaSuhana AhmadPas encore d'évaluation
- Jyotish - Astrology - Destiny and The Wheel of Time - BanDocument204 pagesJyotish - Astrology - Destiny and The Wheel of Time - BanIndira Ranka88% (8)
- FIN Act 2 - OdysseyDocument2 pagesFIN Act 2 - OdysseyJanwyne NgPas encore d'évaluation
- BITS Pilani MBA Project Work Course HandoutDocument20 pagesBITS Pilani MBA Project Work Course Handoutshashank_khatri3728Pas encore d'évaluation
- Indian Political SystemDocument25 pagesIndian Political SystemAyeza AmerPas encore d'évaluation
- Static Magnetic FieldsDocument89 pagesStatic Magnetic FieldsAli Ahmad100% (1)
- Opici Lecture 16 PDFDocument3 pagesOpici Lecture 16 PDFMingu JeongPas encore d'évaluation
- I Don't Care - GapfillDocument3 pagesI Don't Care - GapfillmrbalintPas encore d'évaluation
- ITC Strategic Implementation GuideDocument13 pagesITC Strategic Implementation GuidePranav Goyal100% (1)
- Secured Transactions Torts and Damages Preference of Credits Examination QuestionsDocument14 pagesSecured Transactions Torts and Damages Preference of Credits Examination QuestionsPilacan KarylPas encore d'évaluation
- Brochure For KRC Lang Classes 1Document2 pagesBrochure For KRC Lang Classes 1Jason FranklinPas encore d'évaluation
- RELATIONSHIP BETWEEN CENTRIPETAL FORCE, FREQUENCY, RADIUS AND MASSDocument14 pagesRELATIONSHIP BETWEEN CENTRIPETAL FORCE, FREQUENCY, RADIUS AND MASSKenneth NyuPas encore d'évaluation
- Day 3 Las - Ratio and ProportionDocument7 pagesDay 3 Las - Ratio and ProportionPrecious Joy Torayno BayawaPas encore d'évaluation
- Stephen A. Douglas Homecoming'' Speech Summary: By: Miri Cooper, Rivky Schmidt, and Shiffy HeckmanDocument8 pagesStephen A. Douglas Homecoming'' Speech Summary: By: Miri Cooper, Rivky Schmidt, and Shiffy HeckmanChrisPas encore d'évaluation
- BJDC Construction v. Lanuzo PDFDocument21 pagesBJDC Construction v. Lanuzo PDFAgent BluePas encore d'évaluation
- Modernism in LiteratureDocument4 pagesModernism in LiteratureValeriaPas encore d'évaluation
- Thesis On Critical Discourse AnalysisDocument225 pagesThesis On Critical Discourse Analysissepisendiri100% (4)
- 2005 - Bartels, Sunkar - Drought and Salt Tolerance in PlantsDocument36 pages2005 - Bartels, Sunkar - Drought and Salt Tolerance in PlantsmaurolassoPas encore d'évaluation
- DARPA Instant Flame Suppression Phase II - Final RepostDocument23 pagesDARPA Instant Flame Suppression Phase II - Final Repostyikes salPas encore d'évaluation
- The Paradigmatic Crisis in Chinese Studies: Paradoxes in Social and Economic HistoryDocument44 pagesThe Paradigmatic Crisis in Chinese Studies: Paradoxes in Social and Economic HistoryYadanarHninPas encore d'évaluation
- Office AdministrationDocument3 pagesOffice AdministrationSchool AccountPas encore d'évaluation
- Common Problems in Constructing SentencesDocument6 pagesCommon Problems in Constructing SentencesmimslawPas encore d'évaluation
- Legal Realism Study NotesDocument4 pagesLegal Realism Study Noteskapil shrivastavaPas encore d'évaluation