Académique Documents
Professionnel Documents
Culture Documents
FEED ARUS <36> Absorption Rate and Equilibrium Data
Transféré par
Hafizh PrashantyoDescription originale:
Titre original
Copyright
Formats disponibles
Partager ce document
Partager ou intégrer le document
Avez-vous trouvé ce document utile ?
Ce contenu est-il inapproprié ?
Signaler ce documentDroits d'auteur :
Formats disponibles
FEED ARUS <36> Absorption Rate and Equilibrium Data
Transféré par
Hafizh PrashantyoDroits d'auteur :
Formats disponibles
Lo
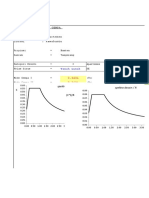
FEED ARUS <36>
Komponen Massa (kg) n (Kmol) Xn
H2O 20046.7791261748 1113.71 0.7147152274
HCl 16225.9644796636 444.547 0.2852847726
Total 36272.7436058384 1558.257 1
Rate air yang dipakai 2 kali rate minimum
Ln,Xn
V n +1 = 1558.2569235 Kmol/jam
Yn+1 = 0.2852847726 1122.593
Asumsi recovery = 98% 1113.702
V1Y1 = 8.8909394409
V' = 1113.7099515 Kmol/jam
V1 = V'/(1-Y1)
Y1 = 0.0079199469
HCl yang terabsorb kedalam air = (Vn+1.Yn+1)-(Vi.Y1)
435.6560326 Kmol/jam
data kesetimbangan uap cair HCl
P total 700
X HCl P Ya X Y
0 0 0 0.00 0.00
0.275 110 0.157143 0.38 0.19
0.336 70 0.1 0.51 0.11
0.418 20 0.028571 0.72 0.03
Yn+1 0.3991586602 Xn Max 0.57
Y1 0.0079831732
L' min = (Yn+1 - Y1) . V'
(Xn max - X0)
= 764.30882913 kmol/jam
Lo min = L'min / (1-X0)
= 764.30882913 kmol/jam
Lo Operasi = 2 Lo Min
= 1528.6176583 Kmol/jam
L'op = Lo op (1-Xo)
= 1528.6176583 kmol/jam
Rate air yang diperlukan = 27515.117849 kg/jam
HCl yang terabsorb ke air = 435.6560326 kmol/jam
Xn . Ln = 435.6560326 kmol/jam
Xn . L'op = 435.6560326 kmol/jam
1-Xn
Xn = 0.2217898833 fraksi mol
Ln = L'op
1-Xn
= 1964.2736909 kmol/jam
= 35356.926436 Kg / jam
HCl terabsorb ke air = 15465.789157 Kg / jam
Jumlah plate ideal = 9 plate
effisinesi = 60%
plate aktual = 15 plate
V1 , y1
Vn+1 ; Yn+1
KESETIMBANGAN UAP CAIR HCl
0.20
0.18
0.16
0.14
0.12
0.10
0.08
0.06
0.04
0.02
0.00
0.00 0.10 0.20 0.30 0.40 0.50 0.60 0.70 0.80
Lo V1 , y1
FEED ARUS <36>
Komponen Massa (kg) n (Kmol) Xn
H2O 760.1753222 42.23196 0.0714030516
HCl 20046.779126 549.2268 0.9285969484
Total 20806.954448 591.4588 1
Rate air yang dipakai 2 kali rate minimum
Ln,Xn Vn+1 ; Yn+1
V n +1 = 591.45878772 Kmol/jam 549.22683 10.984537
Yn+1 = 0.9285969484 53.010087
Asumsi recovery = 98% 42.02555
V1Y1 = 10.984536507
V' = 42.231962344 Kmol/jam
V1 = V'/(1-Y1) 53.216499
Y1 = 0.2064122358
HCl yang terabsorb kedalam a = (Vn+1.Yn+1)-(Vi.Y1)
538.24228887 Kmol/jam
data kesetimbangan uap cair HCl
P total 700 y= 0.1571429
X HCl P Ya X Y x y
0 0 0 0.00 0.00 0 0
0.275 110 0.1571429 0.38 0.19 0.1 0.0157143
0.336 70 0.1 0.51 0.11 0.2 0.0314286
0.418 20 0.0285714 0.72 0.03 0.3 0.0471429
0.4 0.0628571
Yn+1 13.005003672 Xn Max 0.25 0.5 0.0785714
Y1 0.2601000734 0.6 0.0942857
0.7 0.11
L' min = (Yn+1 - Y1) . V' 0.8 0.1257143
(Xn max - X0) 0.9 0.1414286
= 2152.9691555 kmol/jam 1 0.1571429
Lo min = L'min / (1-X0)
= 2152.9691555 kmol/jam
Lo Operasi = 2 Lo Min
= 4305.9383109 Kmol/jam
L'op = Lo op (1-Xo)
= 4305.9383109 kmol/jam
Rate air yang diperlukan = 77506.889597 kg/jam
HCl yang terabsorb ke air = 538.24228887 kmol/jam 101.95923
Xn . Ln = 538.24228887 kmol/jam 0.125
Xn . L'op = 538.24228887 kmol/jam
1-Xn
Xn = 0.1111111111 fraksi mol 0.1111111
Ln = L'op
1-Xn
= 4844.1805998 kmol/jam
= 87195.250796 Kg / jam
HCl terabsorb ke air = 19107.601255 Kg / jam
Jumlah plate ideal = 9 plate
effisinesi = 60%
plate aktual = 15 plate
Vn+1 ; Yn+1
EQUILIBRIUM LINE HCL-WATER
0.09
0.08
0.07
0.06
0.05
0.04
x 0.03
X 0.02 Y
0 0.01 0
0.1111111 0.0159652
0
0.25 0.0324484
0 0.1 0.2 0.3 0.4 0.5 0.6 0.7 0.8 0.9 1
0.4285714 0.0494753
0.6666667 0.0670732
1 0.0852713
1.5 0.1041009
2.3333333 0.1235955
4 0.1437908
9 0.1647255
Vous aimerez peut-être aussi
- Trabajo de Clase (Absorción)Document5 pagesTrabajo de Clase (Absorción)MontsePas encore d'évaluation
- Design of packed absorption columnDocument29 pagesDesign of packed absorption columnJu Naid MalikPas encore d'évaluation
- Video Absorbsi 2Document8 pagesVideo Absorbsi 2Nabilla Putri AndiniPas encore d'évaluation
- Distillation TowerDocument28 pagesDistillation TowerSYED ASGHAR ALI SULTAN100% (1)
- Tugas OTK 3 - AbsorbsiDocument10 pagesTugas OTK 3 - AbsorbsiArganesa Erniko PPas encore d'évaluation
- Metanol - agua separation and drying process optimizationDocument10 pagesMetanol - agua separation and drying process optimizationJAIME ANTONIO VALVERDE RAMOSPas encore d'évaluation
- Where P : A. Flowrate Air Merubah SCFM Menjadi Lbmol/minDocument4 pagesWhere P : A. Flowrate Air Merubah SCFM Menjadi Lbmol/minCarissa EilinPas encore d'évaluation
- Reaktor EsterDocument48 pagesReaktor EsterRofi AriyantoPas encore d'évaluation
- Ou3 2019 IDocument291 pagesOu3 2019 IElizabeth Ttito NuñezPas encore d'évaluation
- EtapasDocument15 pagesEtapasFrank Cesar Delgado MontalvoPas encore d'évaluation
- Problema 1Document8 pagesProblema 1SALMA MAGALÍ DE LA ROSA POSADASPas encore d'évaluation
- Ejemplo Fde C2 NodosDocument1 pageEjemplo Fde C2 NodosVictor Hugo RojasPas encore d'évaluation
- Ntu Vs L/Ga NtuvstDocument2 pagesNtu Vs L/Ga NtuvstEka TianPas encore d'évaluation
- Ullia Nurul - 11.7.6 OtkDocument8 pagesUllia Nurul - 11.7.6 OtkUllia Ismala0% (1)
- From Equation C 1/vmax 2.225 Vmax 0.449 M Km/vmax 3.039 KM M Vmax 1.366Document7 pagesFrom Equation C 1/vmax 2.225 Vmax 0.449 M Km/vmax 3.039 KM M Vmax 1.366Umar FarooqPas encore d'évaluation
- Salsabila Salwa Yusriandi - 2015041070 - Tugas OTK 3 GenapDocument4 pagesSalsabila Salwa Yusriandi - 2015041070 - Tugas OTK 3 GenapSalsabila Salwa YusriandiPas encore d'évaluation
- Indice de Refraccion Vs X: Ch3Oh Chcl3 3Document2 pagesIndice de Refraccion Vs X: Ch3Oh Chcl3 3roman pintorPas encore d'évaluation
- Voltage (V) Vs Current (A) P2Document4 pagesVoltage (V) Vs Current (A) P2Raphael SevillaPas encore d'évaluation
- ASPEN SIMULATION OF MULTITUBULAR PLUGFLOW REACTORDocument8 pagesASPEN SIMULATION OF MULTITUBULAR PLUGFLOW REACTORJay MaradiyaPas encore d'évaluation
- 7Document7 pages7Rommel Sherón RodriguezPas encore d'évaluation
- Ca (Campuran) Konduktivitas 0.005 0.45 0.01 0.85 0.015 1.19 0.02 1.68 0.025 1.91Document3 pagesCa (Campuran) Konduktivitas 0.005 0.45 0.01 0.85 0.015 1.19 0.02 1.68 0.025 1.91MuhammadAmanPas encore d'évaluation
- Curva de Difusion MolecularDocument2 pagesCurva de Difusion MolecularCarlos AlbertoPas encore d'évaluation
- Natural Gas Density CalculatorDocument3 pagesNatural Gas Density CalculatorVaibhav KuralePas encore d'évaluation
- PDES2 Unit ConversionDocument23 pagesPDES2 Unit ConversionAnnisa DwiyantiPas encore d'évaluation
- Nur Fadillah (21613293) - Exercise 2 (Farmakokinetik Dasar)Document5 pagesNur Fadillah (21613293) - Exercise 2 (Farmakokinetik Dasar)NUR FADILLAH -Pas encore d'évaluation
- Absorption of CO Unit: Assumption: CO2 Removal in CO2 Absorber 97% Assumption: CO2 Removal in Flash Separtor 99%Document7 pagesAbsorption of CO Unit: Assumption: CO2 Removal in CO2 Absorber 97% Assumption: CO2 Removal in Flash Separtor 99%Muhammad Umer RanaPas encore d'évaluation
- H2C204 Reaction Rate AnalysisDocument2 pagesH2C204 Reaction Rate AnalysisAdindaRezaOctaviaPas encore d'évaluation
- Inf BombasDocument10 pagesInf BombasWara Martha Pacheco HuancaPas encore d'évaluation
- WoodDocument6 pagesWoodLeswPas encore d'évaluation
- UntitledDocument6 pagesUntitledMartin BishwasPas encore d'évaluation
- Seminar2 McCabe ThieleDocument24 pagesSeminar2 McCabe ThieleTifano KhristiyantoPas encore d'évaluation
- Assignment 3 AdsorptionDocument12 pagesAssignment 3 Adsorptionshivam patelPas encore d'évaluation
- Residence of Separator (Min)Document4 pagesResidence of Separator (Min)marykongPas encore d'évaluation
- I General Loading (Damping 0%) Simple Summation: Tugas 05Document10 pagesI General Loading (Damping 0%) Simple Summation: Tugas 05kason huarPas encore d'évaluation
- Hasil Pengamatan Laprak 3 Dan 4Document4 pagesHasil Pengamatan Laprak 3 Dan 4Reynand ThoriqPas encore d'évaluation
- Crane Beam DesignDocument23 pagesCrane Beam DesignBijoy KPas encore d'évaluation
- Ecuaciones y Graficas-Practica Cerveza-CineticaDocument2 pagesEcuaciones y Graficas-Practica Cerveza-CineticaJuan CadPas encore d'évaluation
- A Robert Evaporator Is Used To Concentrate A 10Document8 pagesA Robert Evaporator Is Used To Concentrate A 10Scrappy WellPas encore d'évaluation
- Amylase EnzymeDocument6 pagesAmylase EnzymeDianne Faye ManabatPas encore d'évaluation
- Calculation Exp FinalDocument16 pagesCalculation Exp FinalSadia HasanPas encore d'évaluation
- Optimized Levenspiel Plot AnalysisDocument14 pagesOptimized Levenspiel Plot AnalysisAqib LatifPas encore d'évaluation
- Gas Natural1Document12 pagesGas Natural1ISRAEL RODRIGUEZPas encore d'évaluation
- Jawaban SlideDocument3 pagesJawaban SlideCitra SalbellaPas encore d'évaluation
- Tugas II Fenomena Transport LanjutDocument3 pagesTugas II Fenomena Transport Lanjutزينل الغزاليPas encore d'évaluation
- Lgtrung - MO - HW#1Document3 pagesLgtrung - MO - HW#1Trung SnowboyPas encore d'évaluation
- Rawabuntu 2019 FixDocument361 pagesRawabuntu 2019 FixSepti MutPas encore d'évaluation
- Tobit Output ExampleDocument3 pagesTobit Output Examplereigeki garfieldPas encore d'évaluation
- Kurva Kalibrasi Parasetamol λ maks 243 nmDocument2 pagesKurva Kalibrasi Parasetamol λ maks 243 nmGelisaPas encore d'évaluation
- Id Ekc316Document2 pagesId Ekc316Viveka RamuPas encore d'évaluation
- Analysis of Drug Concentration Levels in Infusion Over TimeDocument9 pagesAnalysis of Drug Concentration Levels in Infusion Over TimeRizqita Atikah SPas encore d'évaluation
- Laminar water and mercury flow gradientsDocument9 pagesLaminar water and mercury flow gradientsLaura PaipaPas encore d'évaluation
- Zentiva DataDocument19 pagesZentiva DataJay Sheth9Pas encore d'évaluation
- FisicaDocument4 pagesFisicaSantiago GutierrezPas encore d'évaluation
- Lab Report 3Document9 pagesLab Report 3mali.abbas555110Pas encore d'évaluation
- Water Quality in ThailandDocument5 pagesWater Quality in ThailandpsaayoPas encore d'évaluation
- Ideal Gas ConversionDocument5 pagesIdeal Gas ConversionpsaayoPas encore d'évaluation
- OTK - Ekstraksi Cair-Cair Multi StageDocument9 pagesOTK - Ekstraksi Cair-Cair Multi StageArganesa Erniko PPas encore d'évaluation
- Bondocch4810 InversionDocument5 pagesBondocch4810 InversionKristopher Jayson BondocPas encore d'évaluation
- Conversion Table 2Document1 pageConversion Table 2Cj cruzPas encore d'évaluation
- Alfa 3.6 Function Values TableDocument2 pagesAlfa 3.6 Function Values TableHafizh PrashantyoPas encore d'évaluation
- Kelas H Kel 8 - Soal 4.7Document4 pagesKelas H Kel 8 - Soal 4.7Hafizh PrashantyoPas encore d'évaluation
- Simulation of A Mine Plants PresentationDocument58 pagesSimulation of A Mine Plants PresentationKevin DrummPas encore d'évaluation
- Presentation 5Document4 pagesPresentation 5Hafizh PrashantyoPas encore d'évaluation
- ResultsDocument9 pagesResultsHafizh PrashantyoPas encore d'évaluation
- AGA 5 Calculation Spreadsheet for Natural Gas CompositionDocument21 pagesAGA 5 Calculation Spreadsheet for Natural Gas CompositionHafizh Prashantyo100% (1)
- ResultsDocument9 pagesResultsHafizh PrashantyoPas encore d'évaluation
- Asam Basa Ok PDFDocument43 pagesAsam Basa Ok PDFHafizh PrashantyoPas encore d'évaluation
- Counting Chamber (Hemacytometer) GuideDocument7 pagesCounting Chamber (Hemacytometer) GuideJames McguirePas encore d'évaluation
- 10.kumpulan Diagram Fasa PDFDocument70 pages10.kumpulan Diagram Fasa PDFHafizh PrashantyoPas encore d'évaluation
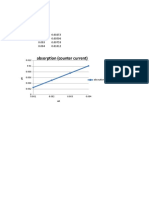
- Absorption (Counter Current) : No1 X y 0.001 0.00253 0.002 0.00506 0.003 0.00759 0.004 0.01012Document1 pageAbsorption (Counter Current) : No1 X y 0.001 0.00253 0.002 0.00506 0.003 0.00759 0.004 0.01012Hafizh PrashantyoPas encore d'évaluation
- Cement IndustryDocument12 pagesCement IndustryHafizh PrashantyoPas encore d'évaluation
- Pulp and Paper IndustryDocument17 pagesPulp and Paper IndustryHafizh PrashantyoPas encore d'évaluation
- Agitated VesselDocument41 pagesAgitated VesselHafizh PrashantyoPas encore d'évaluation
- Palm OilDocument20 pagesPalm OilHafizh PrashantyoPas encore d'évaluation
- Chocolate and CheeseDocument10 pagesChocolate and CheeseHafizh PrashantyoPas encore d'évaluation
- BoilersDocument38 pagesBoilersAshlin Augusty92% (24)
- Industrial BoilersDocument22 pagesIndustrial BoilersAbdul Razzaq MughalPas encore d'évaluation
- Christoper Pizzaro E P Muh Fadil Muh Adrian Budianto: Industrial GasesDocument16 pagesChristoper Pizzaro E P Muh Fadil Muh Adrian Budianto: Industrial GasesHafizh PrashantyoPas encore d'évaluation
- Materials - PlasticsDocument20 pagesMaterials - PlasticsHafizh PrashantyoPas encore d'évaluation