Académique Documents
Professionnel Documents
Culture Documents
SCL9. UV-Vis Spectroscopy - Zamir Sarvari 180410101
Transféré par
ZamirCopyright
Formats disponibles
Partager ce document
Partager ou intégrer le document
Avez-vous trouvé ce document utile ?
Ce contenu est-il inapproprié ?
Signaler ce documentDroits d'auteur :
Formats disponibles
SCL9. UV-Vis Spectroscopy - Zamir Sarvari 180410101
Transféré par
ZamirDroits d'auteur :
Formats disponibles
SCL9.
UV-vis spectroscopy
The purpose of this experiment was to use ultraviolet-visible spectroscopy to identify the spectrums which
belonged to the different known concentrations of rhodamine B solution, and then use this information to
find the extinction coefficient of rhodamine B and thus the concentration of an unknown solution.
Ultraviolet-visible spectroscopy (UV-vis) is a technique which uses radiation to promote bonding electrons
into higher energy levels. Based on the energy level, when an electron is promoted, the radiation absorbed
by that electron will vary at different wavelengths. This can be used in UV-vis spectroscopy to derive a
spectrum for a solution which can then be used to identify specific properties, such as concentration for
this experiment (Convention, 2018).
Under certain conditions, we can derive 𝐴 = 𝜖𝑐𝑑 from the Beer-Lambert law, where A is the absorbance at
a wavelength, ϵ is the molar absorptivity constant, c is the concentration and d is the path length through
the solution (Zhang, 2018). The law states that the absorbance is proportional to the concentration,
therefore the linear relationship can be used to determine molar absorptivity. Furthermore, the molar
absorptivity can be used to determine the concentration of an unknown solution.
Higher concentration solutions will have more bonding electrons to absorb the radiation, therefore more
radiation can be absorbed overall. This means the height of the peaks at a given wavelength can be used to
identify the concentrations of the solutions.
Procedure
• A 100cm³ stock solution of rhodamine B with concentration 62.6µmol/dm3 was prepared. This was
achieved by measuring 0.003g of rhodamine B powder, transferring it to a 25cm³ graduated
cylinder, washing off any remaining powder and then making it up to 25cm³ using deionised water.
The solution was transferred to a separate beaker, and then the graduated cylinder was made up
to 25cm³ and transferred to the beaker three further times, to make up the 100cm³ stock solution.
• Using a graduated pipette with the stock solution and deionised water, 20cm³ of each of the
diluted solutions (5.00µmol/dm³, 1.25µmol/dm³, 0.31µmol/dm³, 0.078µmol/dm³ and
0.019µmol/dm³) were prepared in the graduated cylinder and transferred to separate labelled
containers.
• The solutions in the containers were transferred to separate cuvettes, along with a sample of
deionised water for use as reference. The cuvettes were placed in the spectrometer for analysis,
starting with the deionised water, followed by the solution with the next lowest concentration. The
spectra produced were saved.
• A solution with an unknown concentration which was pre-prepared was also placed in the
spectrometer, so that the concentration can be determined from the spectrum produced. The
cuvettes were then placed in the spectrometer two more times for reliable results.
The spectrometer recorded spectra between a wavelength of 300-700nm, which means that the range of
the cuvettes had to be within that region for the results to be valid. Therefore, 200-800nm cuvettes were
used. The mass scale was not accurate enough for the original stock solution with a concentration of
66µmol/dm³, thus the mass required was rounded and the actual concentration of solution was calculated.
The uncertainty of the mass scale was ±0.0005g, and ±0.1cm³ for the 25cm³ graduated cylinder and
±0.0002cm³ for the graduated pipette. Using this, the maximum and minimum concentrations can be
calculated, thus, the error for each concentration is:
• Stock: 62.6 ± 10.7µmol/dm3
• 5.00 ± 0.84µmol/dm3
• 1.25 ± 0.21µmol/dm3
• 0.31 ± 0.052µmol/dm3
• 0.078 ± 0.014µmol/dm3
• 0.019 ± 0.0040µmol/dm3
Zamir Sarvari 180410101
SCL9. UV-vis spectroscopy
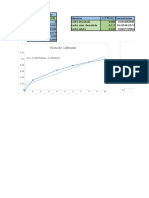
The UV-vis spectroscopy spectrum of the different concentrations
of rhodamine solution
0.7 5±0.84, 554
0.6 1.25±0.21, 554
0.5 Unknown, 555
Absorbance
0.4 0.31±0.052, 555
0.3 0.078±0.014, 555
0.2
0.019±0.0040, 554
0.1
0
300 350 400 450 500 550 600 650 700
Wavelength (𝜆) / nm
5 1.25 0.31 0.078 0.019 Unknown
Knowing the greater peaks belong to solutions of greater concentration, each spectrum can be associated.
The peak wavelength for each concentration is labelled, thus it can be seen that the peak lies at 554-
555nm. The absorbance values from these wavelengths will be averaged and used to find the molar
absorptivity. The spectrum for the unknown solution can be seen between the spectra for 1.25µmol/dm³
and 0.31µmol/dm³, therefore we already know the concentration must lie between those values.
Absorbance
Concentration / µmol/dm³
Wavelength (L) / nm 5 1.25 0.31 0.078 0.019 Unknown
554 0.648 0.154 0.036 0.01 0.003 0.059
555 0.648 0.153 0.036 0.011 0.003 0.059
avg 0.648 0.1535 0.036 0.0105 0.003 0.059
520 0.228 0.055 0.014 0.004 0.001 0.021
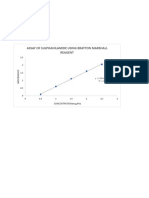
A graph of absorbance against concentration to determine the
molar absorptivity
0.7
0.6
0.5
y = 0.1298x - 0.0026
0.4
Absrobance
0.3 554-555
520
0.2
y = 0.0456x - 0.0003
0.1
0
0 0.5 1 1.5 2 2.5 3 3.5 4 4.5 5
-0.1
Concentration / µmol/dm³
Zamir Sarvari 180410101
SCL9. UV-vis spectroscopy
It can be seen from the graph that the A/c value at 554-555nm is 0.1298dm³/µmol (129811.29dm³/mol)
and 0.0456dm³/µmol (45575.89dm³/mol) at 520nm. Using this, the molar absorptivity can be identified by
dividing by the path length, which in this case is 1.25cm. Therefore, the molar absorptivity is
103849.03dm³/mol/cm between 554-555nm, and 36460.72dm³/mol/cm at 520nm.
To find the concentration of the unknown sample, this molar absorptivity value must be substituted into
𝐴
the Beer-Lambert law. Rearranging the Beer-Lambert law to give concentration: 𝑐 = 𝜖𝑑, and substituting
the absorbance value given in the table, along with the molar absorptivity and path length, gives a
concentration of 0.45µmol/dm³, which fits within our range specified from before.
Percentage error:
106000 − 103849.03
( ) 𝑥100 = 2.02%
106000
(Prahl, 1998)
Conclusion
To conclude, our molar absorptivity constant value is very accurate, however it cannot be certain whether
this is due to random error. The conclusion is rather strong, since many of the errors are accounted for,
however there were many systematic errors out of our control during the experiment. The graph shows
that as the concentration increases, the intensity of the peak also increases.
The procedure had multiple errors, such as reading the apparatus using the eye, which could lead to many
potential human errors. The mass scale did not have enough accuracy for an accurate reading and it was
sensitive, and the amount of powder was very small, therefore making it difficult to get an accurate value.
It also was very difficult to maintain a stable value. This may be due to the ventilation in the room at the
time. To avoid this next time, the air conditioning must be kept off during the experiment. Although repeats
were done for the spectrometer were done, the whole experiment was only done once, which means that
our results may not be reliable.
References
Convention, T. U. S. P., 2018. Qualification of UV-VIS Spectrophotometers. Food Chemical Codex, p. 39.
Prahl, S., 1998. Rhodamine B. [Online]
Available at: https://omlc.org/spectra/PhotochemCAD/html/009.html
[Accessed 25 November 2018].
Zhang, H., 2018. SCL8-11. Nano-engineered Composites with Multi-functionalities, p. 6.
The United States Pharmacopeial Convention. (2018). Food Chemicals Codex (11th Edition) -
Qualification of UV-VIS Spectrophotometers. (pp. 39). The United States Pharmacopeial Convention.
Zamir Sarvari 180410101
Vous aimerez peut-être aussi
- Environmental Engineering Spectrophotometry Part 2Document4 pagesEnvironmental Engineering Spectrophotometry Part 2Sherald AgustinPas encore d'évaluation
- Experiment 1: The Visible Spectra of Soft Drinks: A. Pre-Laboratory QuestionsDocument5 pagesExperiment 1: The Visible Spectra of Soft Drinks: A. Pre-Laboratory QuestionsMuhd Mirza HizamiPas encore d'évaluation
- 103EXP3 SpectrophotometerDocument5 pages103EXP3 SpectrophotometeralperlengerPas encore d'évaluation
- Investigation 31 Determine The Concentration of CuSO4 Using SpectrometerDocument5 pagesInvestigation 31 Determine The Concentration of CuSO4 Using SpectrometerliaojingPas encore d'évaluation
- Bear LambartDocument21 pagesBear Lambartizarul islamPas encore d'évaluation
- Acid Base Indicators Lab ReportDocument6 pagesAcid Base Indicators Lab Reportmuskaan0% (2)
- Spectrophotometric Analysis of KMnO4 SolutionsDocument5 pagesSpectrophotometric Analysis of KMnO4 SolutionsHassan Ali Samoo100% (5)
- Practicum Molecular Biophysics: Protocol For The Week No.3Document11 pagesPracticum Molecular Biophysics: Protocol For The Week No.3Katarina Kaca GacevicPas encore d'évaluation
- Physical Lab ExperimentDocument5 pagesPhysical Lab ExperimentSamanPas encore d'évaluation
- Kurva Kalibrasi Larutan Standar C-Organik: 1. Penentuan LinieritasDocument4 pagesKurva Kalibrasi Larutan Standar C-Organik: 1. Penentuan Linieritasaprilia kurnia putriPas encore d'évaluation
- Experiment.4 Uv/Vis Spectrophotometry: Department of Chemistry University of Bahrain CHEMY310Document6 pagesExperiment.4 Uv/Vis Spectrophotometry: Department of Chemistry University of Bahrain CHEMY310Zahra Al-BasriPas encore d'évaluation
- Additive AravinthDocument15 pagesAdditive AravinthAravinth subramanianPas encore d'évaluation
- Group 9 Assignment Determination of KMnO4Document6 pagesGroup 9 Assignment Determination of KMnO4UsmanPas encore d'évaluation
- Cloruro FerricoDocument8 pagesCloruro FerricoMario GutiérrezPas encore d'évaluation
- Enzyme ActivityDocument3 pagesEnzyme ActivitySyarmine Aqila IsaPas encore d'évaluation
- V V C C: Mass Ion ConcentratDocument2 pagesV V C C: Mass Ion ConcentratJoshua RuizPas encore d'évaluation
- CUSO4 PostlabDocument8 pagesCUSO4 PostlabRuwanthika Fernando100% (1)
- Foster Cole 101230199 Malaïka Zarrouki 2021-01-29Document7 pagesFoster Cole 101230199 Malaïka Zarrouki 2021-01-29Cole FosterPas encore d'évaluation
- Lab Report Beer Lambert'S Law Experiment: Determing The Concentration of Unknown Asa SolutionsDocument5 pagesLab Report Beer Lambert'S Law Experiment: Determing The Concentration of Unknown Asa Solutionsumair saleemPas encore d'évaluation
- Uv Spectro PracDocument11 pagesUv Spectro PracLungeloPas encore d'évaluation
- Experiment 1 SkaDocument12 pagesExperiment 1 SkaSiti Aisyah binti Sha'ari100% (1)
- Quantitative Chromaticity and Turbidity by UV-VIsible Spectroscopy - JASCODocument3 pagesQuantitative Chromaticity and Turbidity by UV-VIsible Spectroscopy - JASCOvrkbioPas encore d'évaluation
- Kurva Standar Metode Biuret Dengan Panjang Gelombang 520 NM: Konsentrasi Albumin (MG/ ML)Document1 pageKurva Standar Metode Biuret Dengan Panjang Gelombang 520 NM: Konsentrasi Albumin (MG/ ML)Teti HungkulPas encore d'évaluation
- Nor Iftiha Binti Abdul Aziz - 2019217292 - Ras1205eDocument26 pagesNor Iftiha Binti Abdul Aziz - 2019217292 - Ras1205eNor Iftiha AzizPas encore d'évaluation
- Chm260 Exp 1Document6 pagesChm260 Exp 1Ilya ZafirahPas encore d'évaluation
- Lab AnalysisDocument4 pagesLab AnalysisErnestasBlaževičPas encore d'évaluation
- SpecDocument8 pagesSpecJirapat ThonglekpechPas encore d'évaluation
- Spectrophotometric Analysis of KMnO4 SolutionsDocument4 pagesSpectrophotometric Analysis of KMnO4 Solutionssidra tariq89% (35)
- Diffraction GratingDocument8 pagesDiffraction GratingClintDoesMusicPas encore d'évaluation
- Glucose Concentr Ation (MG/L) Absorba Nce (A) : Glucose Concentration Vs AbsorbanceDocument6 pagesGlucose Concentr Ation (MG/L) Absorba Nce (A) : Glucose Concentration Vs Absorbancesyahirah shamsudinPas encore d'évaluation
- Absorbance To Transmittance ConverterDocument3 pagesAbsorbance To Transmittance ConverterNadra DanwerPas encore d'évaluation
- Affinity of LDH LabDocument5 pagesAffinity of LDH LabZoe A GarwoodPas encore d'évaluation
- pw1 PDFDocument10 pagespw1 PDFUlvi AgayevPas encore d'évaluation
- pw1 PDFDocument10 pagespw1 PDFUlvi AgayevPas encore d'évaluation
- Experiment 5 - Data TreatmentDocument6 pagesExperiment 5 - Data TreatmentShawn Ann SilanPas encore d'évaluation
- Kami Export - Beers - Law - Lab - GuidedInquiry - StudentHandoutDocument4 pagesKami Export - Beers - Law - Lab - GuidedInquiry - StudentHandoutAlberto RiveraPas encore d'évaluation
- Exepermint 1Document4 pagesExepermint 1Jhone SaaimonPas encore d'évaluation
- Lab Report (Atomic Absorption Spectroscopy)Document8 pagesLab Report (Atomic Absorption Spectroscopy)Shirley Cheong67% (6)
- Concentration Report: Instrument ParametersDocument2 pagesConcentration Report: Instrument ParametersIsabellaNoreñaPas encore d'évaluation
- Section 12 Method DevelopmentDocument21 pagesSection 12 Method DevelopmentNtombizodwa VincenthPas encore d'évaluation
- Copia de BiomasaDocument3 pagesCopia de BiomasaVeronica Lucia Arteaga CorchoPas encore d'évaluation
- KRIBIOLISA Anti-Trastuzumab ELISA Validation - Ver 1 0 Validation FileDocument3 pagesKRIBIOLISA Anti-Trastuzumab ELISA Validation - Ver 1 0 Validation FileKRISHGEN BIOSYSTEMSPas encore d'évaluation
- Physical Organic 1 Post-LabDocument7 pagesPhysical Organic 1 Post-LabsamPas encore d'évaluation
- Group 7 - Data SheetDocument9 pagesGroup 7 - Data SheetJeremy Kyle Edson AustriaPas encore d'évaluation
- Reporte SedimentscionDocument13 pagesReporte SedimentscionANDREA CAMARGOPas encore d'évaluation
- UntitledDocument6 pagesUntitledTokaewa HarusitoPas encore d'évaluation
- Scatter Graph Showing Current Affecting Force On A Current Carrying WireDocument3 pagesScatter Graph Showing Current Affecting Force On A Current Carrying WirePatrick MuñozPas encore d'évaluation
- Iron Calibration Curve For PGDocument6 pagesIron Calibration Curve For PG510418106 ARITRYAGHOSHPas encore d'évaluation
- Contoh Val-Ver Ortofosfat-OkDocument15 pagesContoh Val-Ver Ortofosfat-OkRANY RAMADHANIPas encore d'évaluation
- 01.ex Name Spectrophotometric Determination of Iron.Document4 pages01.ex Name Spectrophotometric Determination of Iron.Md Sohel RanaPas encore d'évaluation
- Calculos Lab 3Document6 pagesCalculos Lab 3Esquivel Bocanegra Pablo HosmarPas encore d'évaluation
- Recta de Calibrado A (595nm)Document4 pagesRecta de Calibrado A (595nm)AlanaPas encore d'évaluation
- Recta de Calibrado A (595nm)Document4 pagesRecta de Calibrado A (595nm)AlanaPas encore d'évaluation
- Residence Time Distribution Analysis of A Continuous Stirred Tank ReactorDocument10 pagesResidence Time Distribution Analysis of A Continuous Stirred Tank ReactorNurul IzzahPas encore d'évaluation
- Graphs - 1Document11 pagesGraphs - 1mr copy xeroxPas encore d'évaluation
- Concentration vs. Absorbance: 1. Standard CurveDocument2 pagesConcentration vs. Absorbance: 1. Standard CurveHee MinPas encore d'évaluation
- Experiment 12: Determination of SO As Baso Using Gravimetry and by "Scattering"Document9 pagesExperiment 12: Determination of SO As Baso Using Gravimetry and by "Scattering"mandayiPas encore d'évaluation
- Spectroscopy Lab ReportDocument7 pagesSpectroscopy Lab ReportSarah B - she herPas encore d'évaluation
- Standard CurveDocument2 pagesStandard CurveNor SyuhailaPas encore d'évaluation
- Math Practice Simplified: Decimals & Percents (Book H): Practicing the Concepts of Decimals and PercentagesD'EverandMath Practice Simplified: Decimals & Percents (Book H): Practicing the Concepts of Decimals and PercentagesÉvaluation : 5 sur 5 étoiles5/5 (3)
- Co5 - Hypothesis Testing (One Sample)Document88 pagesCo5 - Hypothesis Testing (One Sample)RFSPas encore d'évaluation
- Tugas Sistem Pengendali Otomatis: Nama: Bagus Pram Adi Wicaksono Kelas: MS - 4C/07 NIM: 4.21.18.8.07Document10 pagesTugas Sistem Pengendali Otomatis: Nama: Bagus Pram Adi Wicaksono Kelas: MS - 4C/07 NIM: 4.21.18.8.07baguspPas encore d'évaluation
- Syllabus - Transportation AnalyticsDocument3 pagesSyllabus - Transportation AnalyticsRead LeadPas encore d'évaluation
- Meyer PDFDocument42 pagesMeyer PDFVishnuvardhanPas encore d'évaluation
- 6 - Calibrations - Standardizations and Blank Corrections - RevDocument43 pages6 - Calibrations - Standardizations and Blank Corrections - RevKezia CaratorPas encore d'évaluation
- Micro Prelim SolutionsDocument32 pagesMicro Prelim SolutionsMegan JohnstonPas encore d'évaluation
- 05 MAS Distributed Constraint OptimizationDocument124 pages05 MAS Distributed Constraint Optimizationhai zhangPas encore d'évaluation
- Chapter 11 Waiting LineDocument17 pagesChapter 11 Waiting LineJega KanagasabaiPas encore d'évaluation
- Application of Optimization Techniques To Weight Reduction of Automobile BodiesDocument6 pagesApplication of Optimization Techniques To Weight Reduction of Automobile BodiesTibebu MeridePas encore d'évaluation
- Directional HypothesesDocument2 pagesDirectional HypothesesBarby Angel100% (3)
- Nikola Obreshkov - His Life and Mathematical Achievements - HMTM 2012 SarospatakDocument4 pagesNikola Obreshkov - His Life and Mathematical Achievements - HMTM 2012 SarospatakSnezhana Georgieva Gocheva-IlievaPas encore d'évaluation
- Vector CalculusDocument37 pagesVector CalculuserPas encore d'évaluation
- Sin (1/x), Fplot Command - y Sin (X), Area (X, Sin (X) ) - y Exp (-X. X), Barh (X, Exp (-X. X) )Document26 pagesSin (1/x), Fplot Command - y Sin (X), Area (X, Sin (X) ) - y Exp (-X. X), Barh (X, Exp (-X. X) )ayman hammadPas encore d'évaluation
- Gante Iris PPT Pe p006Document20 pagesGante Iris PPT Pe p006Donna Ville GantePas encore d'évaluation
- 2004 - Quality of Life in Romania I MargineanDocument206 pages2004 - Quality of Life in Romania I Margineandale_titiPas encore d'évaluation
- Reliability EngineeringDocument71 pagesReliability Engineeringkjain_9269% (16)
- Principle of Minimum Potential EnergyDocument5 pagesPrinciple of Minimum Potential EnergySaran Jl100% (1)
- Armor.x.paper - Unk.the Numerical Simulation of Warheads Impact and Blast Phenomena Using Autodyn 2d and Autodyn 3d.grady Hayhurst FairlieDocument12 pagesArmor.x.paper - Unk.the Numerical Simulation of Warheads Impact and Blast Phenomena Using Autodyn 2d and Autodyn 3d.grady Hayhurst Fairliegordon827Pas encore d'évaluation
- Lecture 9 The Lag OperatorDocument10 pagesLecture 9 The Lag Operatorz_k_j_vPas encore d'évaluation
- Analyzing Missing DataDocument49 pagesAnalyzing Missing Datasana mehmoodPas encore d'évaluation
- M.SC Maths 1st Sem SyllabusDocument5 pagesM.SC Maths 1st Sem SyllabusSathish KumarPas encore d'évaluation
- Stochastic Calculus Notes 1/5Document25 pagesStochastic Calculus Notes 1/5tburi100% (3)
- Lakens2013-Calculating and Reporting Effect Sizes To Facilitate Cumulative Science PDFDocument12 pagesLakens2013-Calculating and Reporting Effect Sizes To Facilitate Cumulative Science PDFDanielPas encore d'évaluation
- PID ControllerDocument17 pagesPID Controllermothaffar ahmad100% (1)
- Template Weighing Scale - ManualDocument4 pagesTemplate Weighing Scale - ManualMohd Ezan MohammadiahPas encore d'évaluation
- Ass 2021Document7 pagesAss 2021gogajauharPas encore d'évaluation
- Lie BracketDocument2 pagesLie BracketMogaime BuendiaPas encore d'évaluation
- Graph of A RelationDocument48 pagesGraph of A RelationHamza KahemelaPas encore d'évaluation
- Addi Tional Mathematics Sba Mark Scheme - Project B: Caribbean Secondary Education CertificateDocument4 pagesAddi Tional Mathematics Sba Mark Scheme - Project B: Caribbean Secondary Education CertificateDarrion BrucePas encore d'évaluation
- Entire Course Material All Files Combined For 2011 FinalDocument191 pagesEntire Course Material All Files Combined For 2011 FinalFinian MassaPas encore d'évaluation