Académique Documents
Professionnel Documents
Culture Documents
(Base Name of Oxyanion + Suffix (-Ide) ) + Acid
Transféré par
Divine Angela Serag0 évaluation0% ont trouvé ce document utile (0 vote)
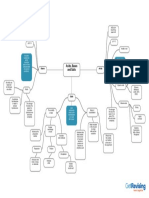
19 vues1 pageThis document provides a decision tree for naming binary compounds, acids, and ionic compounds. It starts with determining if a compound is binary, then provides suffixes and prefixes to apply based on whether it is covalent, ionic, a metal, acid, or other classification. The tree ends by addressing hydrates, polyprotic acids, and potassium salts.
Description originale:
Titre original
Flow-chart (1).docx
Copyright
© © All Rights Reserved
Formats disponibles
DOCX, PDF, TXT ou lisez en ligne sur Scribd
Partager ce document
Partager ou intégrer le document
Avez-vous trouvé ce document utile ?
Ce contenu est-il inapproprié ?
Signaler ce documentThis document provides a decision tree for naming binary compounds, acids, and ionic compounds. It starts with determining if a compound is binary, then provides suffixes and prefixes to apply based on whether it is covalent, ionic, a metal, acid, or other classification. The tree ends by addressing hydrates, polyprotic acids, and potassium salts.
Droits d'auteur :
© All Rights Reserved
Formats disponibles
Téléchargez comme DOCX, PDF, TXT ou lisez en ligne sur Scribd
0 évaluation0% ont trouvé ce document utile (0 vote)
19 vues1 page(Base Name of Oxyanion + Suffix (-Ide) ) + Acid
Transféré par
Divine Angela SeragThis document provides a decision tree for naming binary compounds, acids, and ionic compounds. It starts with determining if a compound is binary, then provides suffixes and prefixes to apply based on whether it is covalent, ionic, a metal, acid, or other classification. The tree ends by addressing hydrates, polyprotic acids, and potassium salts.
Droits d'auteur :
© All Rights Reserved
Formats disponibles
Téléchargez comme DOCX, PDF, TXT ou lisez en ligne sur Scribd
Vous êtes sur la page 1sur 1
Start
Is it a Binary
Compound?
Yes No
Put suffix (-ide) Is it a Binary Acid?
on anion
Yes No
Is it
covalent? Hydro + (base Is it an
name of oxyacid?
element + -ic)
Yes No
+ acid
Yes No
Name using
Is
End
prefixes for Is the
number of it transitional
suffix (-ite)?
metal or semi-
atoms metal? Yes No
End [Base name of
oxyanion + suffix Is the suffix Is it a
Yes No (-ous)] + acid (-ate)? Hydrate?
Polyatomic Ions Ionic Compound
End [Base name of oxyanion
Name of cation + + suffix (-ide)] + acid
No prefixes /
charge of cation name of cation +
+ base name of base name of End
anion with (-ide) anion (-ide)
Yes No
End End
Ionic compound
+ (hydrate with Is it a
Salt of
prefix) Polyprotic
Acids?
End
Yes No
Add prefix to Hydrogen +
cation and [anion(-ide)/
[anion(-ide)/ oxyanion]
oxyanion]
Is it a
End Potassium
Salt?
Change H to K
End
Vous aimerez peut-être aussi
- Stanford-Binet Test Scoring Explained - Stanford-Binet IQ TestDocument3 pagesStanford-Binet Test Scoring Explained - Stanford-Binet IQ TestLM R50% (2)
- Acid Mind MapDocument1 pageAcid Mind MapIndianagrofarms100% (2)
- Trane Air Cooled Scroll Chillers Installation Operation MaintenanceDocument276 pagesTrane Air Cooled Scroll Chillers Installation Operation MaintenanceBay Mưa100% (1)
- Bartos P. J., Glassfibre Reinforced Concrete - Principles, Production, Properties and Applications, 2017Document209 pagesBartos P. J., Glassfibre Reinforced Concrete - Principles, Production, Properties and Applications, 2017Esmerald100% (3)
- Docu Ifps Users Manual LatestDocument488 pagesDocu Ifps Users Manual LatestLazar IvkovicPas encore d'évaluation
- (CHEM) Flowchart NamingDocument1 page(CHEM) Flowchart Namingsodiumboyupinthishoe100% (1)
- Ion Exchange - 1Document33 pagesIon Exchange - 1Bayu KristyonoPas encore d'évaluation
- (Tutorial) MC IiaDocument29 pages(Tutorial) MC IiaamirPas encore d'évaluation
- Inorganic - Acids - and - Bases - Edrolo - Study - NotesDocument15 pagesInorganic - Acids - and - Bases - Edrolo - Study - Notessam12gouldPas encore d'évaluation
- Phase DiagramDocument13 pagesPhase DiagramVishaldatt KohirPas encore d'évaluation
- Katalis HeterogenDocument36 pagesKatalis HeterogenDewisPas encore d'évaluation
- Naming TreeDocument1 pageNaming TreeJerremiah YuPas encore d'évaluation
- Material Selection Guide: 01633 636800 Sales@tomoe - Co.ukDocument35 pagesMaterial Selection Guide: 01633 636800 Sales@tomoe - Co.ukDanang Cliff RizaldiPas encore d'évaluation
- Atomic Structure and The Periodic Table Foundation Revision Activity MatDocument4 pagesAtomic Structure and The Periodic Table Foundation Revision Activity MatHồng Ngọc VõPas encore d'évaluation
- ENG101 Acidizing L N2 - #06a - Acidizing Products Chemical DivertersDocument1 pageENG101 Acidizing L N2 - #06a - Acidizing Products Chemical DivertersdanielmcaePas encore d'évaluation
- Atomic Structure and The Periodic Table Revision Activity MatDocument1 pageAtomic Structure and The Periodic Table Revision Activity Matkareem el barbaryPas encore d'évaluation
- Note Aug 23, 2023Document4 pagesNote Aug 23, 2023Meghan IrishPas encore d'évaluation
- Trimec: Chemical Compatibility ChartDocument12 pagesTrimec: Chemical Compatibility ChartMandiriPas encore d'évaluation
- Adobe Scan 07-Nov-2023Document2 pagesAdobe Scan 07-Nov-2023yuvrajsingh776306Pas encore d'évaluation
- 15 Lecture PPTDocument7 pages15 Lecture PPTDavid Hoktua Siregar siregarPas encore d'évaluation
- Naming CompoundsDocument10 pagesNaming CompoundsIan Miala MorenoPas encore d'évaluation
- Naming Flowchart ChemistryDocument1 pageNaming Flowchart ChemistryamyPas encore d'évaluation
- ASK-M 72cells Monocrystalline Module: Key FeaturesDocument2 pagesASK-M 72cells Monocrystalline Module: Key FeaturesNam Tran HoangPas encore d'évaluation
- Thermit Welding (GB) LTD Process ManualsDocument10 pagesThermit Welding (GB) LTD Process ManualsAntónio AlmeidaPas encore d'évaluation
- Shoulder Joint Position Sense Improves With ElevationDocument10 pagesShoulder Joint Position Sense Improves With ElevationpredragbozicPas encore d'évaluation
- 5000mah Mi Power Bank 2 - PDFDocument6 pages5000mah Mi Power Bank 2 - PDFManuel Jesús Fernández lavadoPas encore d'évaluation
- OM Mannual FOsDocument38 pagesOM Mannual FOsAbdulmuqtadetr AhmadiPas encore d'évaluation
- Nature and Nurture - How They Play A Role in Serial Killers and THDocument40 pagesNature and Nurture - How They Play A Role in Serial Killers and THJ.A.B.APas encore d'évaluation
- EESC 111 Worksheets Module 5Document5 pagesEESC 111 Worksheets Module 5Keira O'HowPas encore d'évaluation
- Ca 2013 39Document40 pagesCa 2013 39singh1699Pas encore d'évaluation
- Sepsis Management UCLA HealthDocument23 pagesSepsis Management UCLA HealthKomang_JananuragaPas encore d'évaluation
- Boli Vertebro MedulareDocument12 pagesBoli Vertebro MedulareHalit DianaPas encore d'évaluation
- Product Term and ConditionsDocument50 pagesProduct Term and Conditionsain2Pas encore d'évaluation
- Broucher Design - 02Document8 pagesBroucher Design - 02ಉಮೇಶ ಸಿ. ಹುಕ್ಕೇರಿ ಹುಕ್ಕೇರಿPas encore d'évaluation
- EE2401 Power System Operation and ControlDocument93 pagesEE2401 Power System Operation and ControlPrasanth GovindarajPas encore d'évaluation
- Ne XT ProtDocument2 pagesNe XT Protwilliam919Pas encore d'évaluation
- Practice Test For Exam 3 Name: Miguel Vivas Score: - /10Document2 pagesPractice Test For Exam 3 Name: Miguel Vivas Score: - /10MIGUEL ANGELPas encore d'évaluation
- Top 6 Beginner Work Out MistakesDocument4 pagesTop 6 Beginner Work Out MistakesMARYAM GULPas encore d'évaluation
- 5: Chemical Trends - Topic Questions: Year Series Paper NumberDocument10 pages5: Chemical Trends - Topic Questions: Year Series Paper NumberSumaira AliPas encore d'évaluation
- Class 7 Work Book Answers Acid Bases and SaltsDocument2 pagesClass 7 Work Book Answers Acid Bases and SaltsGaurav SethiPas encore d'évaluation
- Presentasi Evaluasi Manajemen Risiko - YLYDocument16 pagesPresentasi Evaluasi Manajemen Risiko - YLYOPERASIONALPas encore d'évaluation
- Cuts of BeefDocument4 pagesCuts of BeefChristopher EnriquezPas encore d'évaluation
- 7 LevelDocument5 pages7 LevelACHREF RIHANIPas encore d'évaluation
- The Lion and The AntDocument4 pagesThe Lion and The AntDebby Jean ChavezPas encore d'évaluation
- House of Candy PresentationDocument42 pagesHouse of Candy PresentationRohit JaroudiyaPas encore d'évaluation
- 1 A Finalexam FNH330 June 2015 Final Review QuestionsDocument6 pages1 A Finalexam FNH330 June 2015 Final Review QuestionsChinley HinacayPas encore d'évaluation
- Margarita ForesDocument20 pagesMargarita ForesKlarisse YoungPas encore d'évaluation
- Biology Project Cronary Heart Diseas (CHD)Document7 pagesBiology Project Cronary Heart Diseas (CHD)احمد المغربي50% (2)