Académique Documents
Professionnel Documents
Culture Documents
Topic 1 Exercise 2 - Ram RMM and Mass Spectra
Transféré par
Salman ZaidiTitre original
Copyright
Formats disponibles
Partager ce document
Partager ou intégrer le document
Avez-vous trouvé ce document utile ?
Ce contenu est-il inapproprié ?
Signaler ce documentDroits d'auteur :
Formats disponibles
Topic 1 Exercise 2 - Ram RMM and Mass Spectra
Transféré par
Salman ZaidiDroits d'auteur :
Formats disponibles
Topic 1 Exercise 2 – ram, rmm and mass spectra
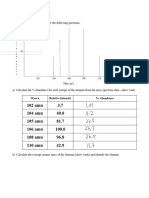
Deduce the relative atomic masses of the following elements.
1. Silicon (28Si 92.21%, 29Si 4.70%, 30Si 3.09%)
2. Silver (107Ag 51.88%, 109Ag 48.12%)
3. Boron (10B 19.7%, 11B 80.7%)
4. Gallium (69Ga 60.2%, 71Ga 39.8%)
5. Zirconium:
Assume in all cases the relative isotopic mass is equal to the mass number.
--------
6. Bromine has two isotopes, with mass numbers 79 and 81. Its relative atomic mass is often given as 80. What does that tell you
about the relative abundance of the two isotopes?
7. Most argon atoms have a mass number 40. How many neutrons does this isotope have? The relative isotopic mass of this isotope
is 39.961, but the relative atomic mass of argon is 39.948. What can you deduce about the other isotopes of argon?
----------
8. For each of the following stages in a mass spectrometer, state which part of the spectrometer is responsible for it and how it
works:
a) electrospray ionization
b) acceleration
c) ion drift
d) detection
--------------------
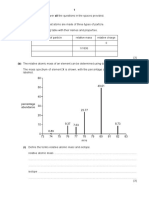
9. Deduce, giving reasons, the relative molecular mass of compound A, which has the following mass spectrum:
100
80
relative 60
abundance
29
40 57
15 43
20
72
Vous aimerez peut-être aussi
- 1.1 EXERCISE 2 - Ram, RMM and Mass SpectraDocument2 pages1.1 EXERCISE 2 - Ram, RMM and Mass Spectrathc8477Pas encore d'évaluation
- 1.1 Exercise 2 - Ram, RMM and Mass Spectra. MASS SPECTROMETRYDocument2 pages1.1 Exercise 2 - Ram, RMM and Mass Spectra. MASS SPECTROMETRYDr.AndrewMatovuPas encore d'évaluation
- Section A QP MSDocument5 pagesSection A QP MSDaniel ConwayPas encore d'évaluation
- Section A QP MSDocument5 pagesSection A QP MSDaniel ConwayPas encore d'évaluation
- Nuclear and Particle Physics QuestionsDocument41 pagesNuclear and Particle Physics QuestionsSid KPas encore d'évaluation
- Calculating Atomic MassesDocument3 pagesCalculating Atomic MassesMaddalenaPas encore d'évaluation
- CHEM 101 LECTURE EXCERCISE 2 On Mass Spectrometer and Quantum Theory - E. N DIM-1-1Document4 pagesCHEM 101 LECTURE EXCERCISE 2 On Mass Spectrometer and Quantum Theory - E. N DIM-1-1Ekene Agwu100% (1)
- Moodia - Lnmit.ac - In/moodle/mod/quiz/review - Phip?attempt-1407a8cmid - 282#question-16274-2Document8 pagesMoodia - Lnmit.ac - In/moodle/mod/quiz/review - Phip?attempt-1407a8cmid - 282#question-16274-2happyhomie6969Pas encore d'évaluation
- Physics Atomic Structure QuestionsDocument22 pagesPhysics Atomic Structure QuestionsAbigail BPas encore d'évaluation
- Mass Spectra Worksheet 1Document5 pagesMass Spectra Worksheet 13estherPas encore d'évaluation
- Worksheet of Atomic Structure (IX)Document5 pagesWorksheet of Atomic Structure (IX)sabeehaPas encore d'évaluation
- Combines Spectra ExerciseDocument76 pagesCombines Spectra ExerciseIbtihal noorPas encore d'évaluation
- Mass Spectra WSDocument3 pagesMass Spectra WS776pmsfq2fPas encore d'évaluation
- A Level Pracice - Atomic StructureDocument8 pagesA Level Pracice - Atomic StructureXiyue ChenPas encore d'évaluation
- Dokumen PDF 26Document1 pageDokumen PDF 26Farhan FarhanPas encore d'évaluation
- Chem 86 Nuclear ChemistryDocument1 pageChem 86 Nuclear ChemistryHernan SalvadorPas encore d'évaluation
- Particle Physics 2 QPDocument10 pagesParticle Physics 2 QPRichard Davidson12Pas encore d'évaluation
- Year 1 Tutorial 1Document3 pagesYear 1 Tutorial 1Mel ManningPas encore d'évaluation
- The Modern View of Atomic Structure Atomic Weights (Sections 2.3 and 2.4)Document2 pagesThe Modern View of Atomic Structure Atomic Weights (Sections 2.3 and 2.4)CRISTINA MUÑOZ CASTAÑOPas encore d'évaluation
- Uni/Tersity The Pui/Jab: Llo :'J ' 'J:'JDocument21 pagesUni/Tersity The Pui/Jab: Llo :'J ' 'J:'JJhon EstwoodPas encore d'évaluation
- Exercise 3.3 - Nuclear StabilityDocument1 pageExercise 3.3 - Nuclear StabilityRinalyn AsuncionPas encore d'évaluation
- Atom - Ws Avg Atomic MassDocument2 pagesAtom - Ws Avg Atomic MassTaylor DixonPas encore d'évaluation
- Calculating Average Atomic MassDocument4 pagesCalculating Average Atomic MassBrenda SchroederPas encore d'évaluation
- WWW - Manaresults.co - In: II B. Tech II Semester Regular/Supplementary Examinations, April/May - 2019 Electrical Machines-IiDocument8 pagesWWW - Manaresults.co - In: II B. Tech II Semester Regular/Supplementary Examinations, April/May - 2019 Electrical Machines-IiJyosthna pathuriPas encore d'évaluation
- Exp03-Gamma-Ray Spectroscopy Using NaI (TL)Document10 pagesExp03-Gamma-Ray Spectroscopy Using NaI (TL)Muhammad ToharohPas encore d'évaluation
- Jorge Willis Guide Practice Problems 1 (Atomic Number)Document4 pagesJorge Willis Guide Practice Problems 1 (Atomic Number)jogerwillisPas encore d'évaluation
- Basic ElectronicsDocument18 pagesBasic ElectronicsGrace jerePas encore d'évaluation
- Elec2091st Semester Exam Papr20221 - 1705355554568Document7 pagesElec2091st Semester Exam Papr20221 - 1705355554568family7482pleasePas encore d'évaluation
- Y12Document5 pagesY12Kissiedu YirenkyiPas encore d'évaluation
- X Sem - CHEMISTRY.Quantum, Nuclear & Radiation Chem - Old CBCS - MSC Ed May 2019Document3 pagesX Sem - CHEMISTRY.Quantum, Nuclear & Radiation Chem - Old CBCS - MSC Ed May 2019Raghavendra BPas encore d'évaluation
- BCTN RelayDocument57 pagesBCTN RelayThư Phạm Hoàng MinhPas encore d'évaluation
- MSC II SEM III Natural Product and Spectroscopy MOCK TESTDocument3 pagesMSC II SEM III Natural Product and Spectroscopy MOCK TESTEvgenia MakantasiPas encore d'évaluation
- Roots Classes: Physics TestDocument3 pagesRoots Classes: Physics TestAnupam DubeyPas encore d'évaluation
- Gamma Spectroscopy: Pre-Lab: Physics 160 Principles of Modern Physics Laboratory Session #9Document5 pagesGamma Spectroscopy: Pre-Lab: Physics 160 Principles of Modern Physics Laboratory Session #9Mohammad AlmistarehiPas encore d'évaluation
- Exercises - ReciprocalSpaces and XRDDocument2 pagesExercises - ReciprocalSpaces and XRDtackyjc0% (1)
- EEE105 2007 Question PDFDocument9 pagesEEE105 2007 Question PDFShobanraj LetchumananPas encore d'évaluation
- β -: 3.92 max (7%), 3.10 max (5%), 1.60 max (88%)Document1 pageβ -: 3.92 max (7%), 3.10 max (5%), 1.60 max (88%)Chris LovedPas encore d'évaluation
- Structure of Atom.Document2 pagesStructure of Atom.Antony KonikaraPas encore d'évaluation
- Atomic Structure Questions Relative Atomic Mass, Relative Molecular Mass & Mass SpectrosDocument2 pagesAtomic Structure Questions Relative Atomic Mass, Relative Molecular Mass & Mass SpectrosAndrei FloreaPas encore d'évaluation
- Assignment Booklet: Elements of Modern PhysicsDocument4 pagesAssignment Booklet: Elements of Modern PhysicsQwertyuPas encore d'évaluation
- CAPE Physics 2007 U2 P2 PDFDocument18 pagesCAPE Physics 2007 U2 P2 PDFJadon ThomasPas encore d'évaluation
- Problems 44Document12 pagesProblems 44Alvien HidayatPas encore d'évaluation
- A Calibration Routine For Full Matrix Capture (FMC)Document10 pagesA Calibration Routine For Full Matrix Capture (FMC)PetrPas encore d'évaluation
- POWER SYSTEMS - 1 - QuestionsDocument7 pagesPOWER SYSTEMS - 1 - QuestionsB.Neelakanteshwar RaoPas encore d'évaluation
- 2016 Physics Board PaperDocument16 pages2016 Physics Board Papershivkarnwal2Pas encore d'évaluation
- Stoichiometry Atomic and Molecular Mass Worksheet - AnswerDocument3 pagesStoichiometry Atomic and Molecular Mass Worksheet - AnswerFoxy world 152Pas encore d'évaluation
- Supplementary Material For Multi-Faceted Distillation of Base-Novel Commonality For Few-Shot Object DetectionDocument6 pagesSupplementary Material For Multi-Faceted Distillation of Base-Novel Commonality For Few-Shot Object DetectionAli Eren KaradağPas encore d'évaluation
- Chemistry 2 - Atoms, Molecules, and Ions 2.3 - Atomic Structure and Symbolism Student ManualDocument2 pagesChemistry 2 - Atoms, Molecules, and Ions 2.3 - Atomic Structure and Symbolism Student ManualsiewyonglimPas encore d'évaluation
- STAT 497 - Old ExamsDocument71 pagesSTAT 497 - Old ExamsAnisa Anisa100% (1)
- Final ExamDocument11 pagesFinal Exammalak hilalPas encore d'évaluation
- 4541.3 End Year 2016Document8 pages4541.3 End Year 2016amin_zamanPas encore d'évaluation
- AP Unit1 WorksheetDocument4 pagesAP Unit1 Worksheetburcak gecPas encore d'évaluation
- Altarelli 1982Document129 pagesAltarelli 1982Jonatan PaschoalPas encore d'évaluation
- (PDF) Race 2 Nurture Atomic Structure 2021Document2 pages(PDF) Race 2 Nurture Atomic Structure 2021Sab MajedarPas encore d'évaluation
- Towards The Rehabilitation of The Leuckart Reductive Amination Reaction Using Microwave TechnologyDocument4 pagesTowards The Rehabilitation of The Leuckart Reductive Amination Reaction Using Microwave TechnologyRomanPas encore d'évaluation
- HL1-Chapter 2 Review SheetDocument5 pagesHL1-Chapter 2 Review SheetShafika AnuarPas encore d'évaluation
- AnalysisDocument14 pagesAnalysisBINDU100% (1)
- Network Analysis Aug 2022Document12 pagesNetwork Analysis Aug 2022Anand TPas encore d'évaluation
- B.Tech. Degree Examination Computer Science & EngineeringDocument1 pageB.Tech. Degree Examination Computer Science & EngineeringDurga KPas encore d'évaluation
- Determination of Rate Equation - 2 QPDocument12 pagesDetermination of Rate Equation - 2 QPSalman ZaidiPas encore d'évaluation
- Determination of Rate Equation - 1 QPDocument13 pagesDetermination of Rate Equation - 1 QPSalman ZaidiPas encore d'évaluation
- Rate Equations - 1 QPDocument15 pagesRate Equations - 1 QPSalman ZaidiPas encore d'évaluation
- Topic 2 Exercise 2 - SolutionsDocument2 pagesTopic 2 Exercise 2 - SolutionsSalman ZaidiPas encore d'évaluation
- Rockford Cambridge School: Play Group To A Level Test Topics For SEPTEMBER - 2019Document3 pagesRockford Cambridge School: Play Group To A Level Test Topics For SEPTEMBER - 2019Salman ZaidiPas encore d'évaluation
- What Is The Ratio of The Volume of 2 G of Hydrogen To The Volume of 16 G of Methane, Both Volumes at R.T.P.? A. 1 To 1 B. 1 To 2 C. 1 To 8 D. 2 To 1Document2 pagesWhat Is The Ratio of The Volume of 2 G of Hydrogen To The Volume of 16 G of Methane, Both Volumes at R.T.P.? A. 1 To 1 B. 1 To 2 C. 1 To 8 D. 2 To 1Salman ZaidiPas encore d'évaluation
- Topic 1 Exercise 1 - Atomic SymbolsDocument1 pageTopic 1 Exercise 1 - Atomic SymbolsSalman ZaidiPas encore d'évaluation