Académique Documents
Professionnel Documents
Culture Documents
Adult Focal Epilepsy
Transféré par
zhoujCopyright
Formats disponibles
Partager ce document
Partager ou intégrer le document
Avez-vous trouvé ce document utile ?
Ce contenu est-il inapproprié ?
Signaler ce documentDroits d'auteur :
Formats disponibles
Adult Focal Epilepsy
Transféré par
zhoujDroits d'auteur :
Formats disponibles
Review Article
Adult Focal Epilepsies
Address correspondence to
Dr Christopher T. Skidmore,
Thomas Jefferson University,
901 Walnut Street, 4th Floor,
Philadelphia, PA 19107, Christopher T. Skidmore, MD
christopher.skidmore@
jefferson.edu.
Relationship Disclosure:
Dr Skidmore serves as a ABSTRACT
consultant for Upsher-Smith Purpose of Review: Focal epilepsy is the most common type of epilepsy in
Laboratories, Inc, and has
received research support from adulthood. This article discusses the seizure symptomatology, EEG findings, and
NeuroPace, Inc. imaging findings of the various forms of focal epilepsy. The majority of the article
Unlabeled Use of focuses on temporal and frontal lobe epilepsy as these represent the majority of
Products/Investigational
Use Disclosure:
focal epilepsies.
Dr Skidmore reports Recent Findings: While significant overlap exists in the clinical symptomatology of
no disclosure. the focal epilepsies, detailed seizure descriptions can often provide useful clinical
* 2016 American Academy evidence to help establish an accurate diagnosis. EEG and MRI continue to serve as
of Neurology.
the main diagnostic tools for the diagnosis of focal epilepsy.
Summary: The various forms of focal epilepsy generate seizure presentations that
are dependent on the anatomic structures that are involved in the seizure. By
understanding the symptoms typically generated in each region of the brain, a
better understanding of the possible seizure localizations can be made. Most forms
of epilepsy have clear changes on EEG that permit accurate localization, but several
pitfalls exist, which are discussed in this article. Imaging has revolutionized our
ability to accurately identify lesions associated with epilepsy and increased our
ability to localize seizures in the brain.
Continuum (Minneap Minn) 2016;22(1):94–115.
INTRODUCTION Other acquired causes, such as in-
This article focuses on focal (partial- fections or trauma, can be associated
onset) epilepsy since it is the most with acute or remote onset of epilepsy.
common form of epilepsy to be Obtaining a detailed epilepsy risk
diagnosed during adulthood. Certainly factor history from patients, including
patients with idiopathic generalized a history of pregnancy complica-
epilepsy, especially juvenile myoclonic tions, learning or physical disabilities/
epilepsy, can have onset in early milestone delay, febrile seizures, sig-
adulthood, but the clinical history nificant head trauma, central nervous
and diagnostic workup are the same system infections, and a family history
as for childhood-onset epilepsy and of epilepsy, is important to help guide
are discussed in the article “Infantile, the clinical workup.
Childhood, and Adolescent Epilep- This article outlines the typical
sies” by Elaine Wirrell, MD,1 in this presentation of various forms of focal
issue of Continuum. epilepsy and highlights the typical
Focal epilepsy can develop at any seizure symptomatology, interictal and
point in life, and the etiologies associ- ictal scalp EEG, and potential etiologies.
ated with it vary by age. In general
terms, congenital abnormalities or TEMPORAL LOBE EPILEPSY
Supplemental digital content: genetic triggers are often associated The temporal lobe is one of the most
Direct URL citations appear in with earlier onset, while stroke and common localizations for focal epilepsy,
the printed text and are provided
in the HTML, PDF, and app neurodegenerative disorders are asso- and it is important to recognize the
versions of this article. ciated with an older age of onset. common presentations to ensure that
94 www.ContinuumJournal.com February 2016
Copyright © American Academy of Neurology. Unauthorized reproduction of this article is prohibited.
KEY POINT
an accurate diagnosis is established. The factor for the development of hippo- h Mesial temporal lobe
features of temporal lobe epilepsy can be campal sclerosis,2 which is a common epilepsy is the most
broken down into two broad categories: cause of mesial temporal lobe epi- common form of
mesial and lateral (neocortical) onset. lepsy. Hippocampal sclerosis is char- temporal lobe epilepsy,
acterized by atrophy and gliosis of and the most common
Mesial Temporal Lobe Epilepsy the hippocampus, which may be pathology associated
Mesial temporal lobe epilepsy is one of unilateral or bilateral (Figure 5-1 with mesial temporal
the most well-defined forms of epilepsy and Figure 5-2). Other etiologies that lobe epilepsy is
in the literature. It often begins during may be seen in the mesial temporal hippocampal sclerosis.
late adolescence or early adulthood. lobe include cavernous hemangiomas,
Clinical features. Prolonged febrile gliosis/encephalomalacia secondary to
seizures are believed to be a risk prior injury, and tumors such as
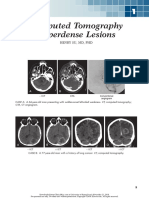
FIGURE 5-1 Right mesial temporal sclerosis. A, B, Coronal T1-weighted MRIs demonstrate a
smaller right hippocampus. C, D, Coronal fluid-attenuated inversion recovery
(FLAIR) MRIs demonstrate increased signal in the right hippocampus consistent
with gliosis.
Continuum (Minneap Minn) 2016;22(1):94–115 www.ContinuumJournal.com 95
Copyright © American Academy of Neurology. Unauthorized reproduction of this article is prohibited.
Adult Focal Epilepsies
FIGURE 5-2 Bilateral mesial temporal sclerosis. Coronal
fluid-attenuated inversion recovery (FLAIR)
MRI demonstrates bilateral increased signal
in the hippocampus consistent with gliosis.
dysembryoplastic neuroepithelial tumors with nondominant temporal lobe sei-
(Case 5-1). zures include ictal spitting, vomiting,
Many patients with mesial temporal and urinary urge.6
lobe epilepsy experience auras, which The seizures typically last 60 to
can include psychic sensations (déjà vu 90 seconds and are often followed by
and jamais vu), a gastric rising sensa- a period of confusion/disorientation,
tion, a sensation of “butterflies” in their more prominent in patients with dom-
stomach, fear, or olfactory symptoms inant temporal lobe seizures. After
(usually an unpleasant chemical or dominant temporal lobe seizures,
burning smell). The aura is typically prominent postictal symptoms may be
followed by loss of consciousness/ attributed to involvement of the lan-
awareness with prominent oral and guage centers in the ictal discharge.
manual automatisms in patients with Seizures from the mesial temporal
dominant temporal lobe seizures, while region rarely secondarily generalize, but
nondominant temporal lobe seizures when they do, it is typically very late
may have preserved awareness with into the seizure in contrast to neo-
oral and manual automatisms (Case 5-1).5 cortical temporal lobe or extratemporal
Dystonic limb posturing during the ictus lobe epilepsy, which frequently second-
has been described and is very predic- arily generalizes earlier in the seizure.
tive of seizure onset starting in the Several clinical features help to lateral-
mesial temporal lobe contralateral to ize the seizure focus to the ipsilateral
the dystonic limb. It is not uncommon temporal lobe regardless of dominance.
for the patient to have manual autom- These include postictal nose wiping,
atisms in the hand ipsilateral to the ictal unilateral eye blinking (Case 5-2),
seizure focus. Symptoms associated and ictal piloerection.6 The hand that
96 www.ContinuumJournal.com February 2016
Copyright © American Academy of Neurology. Unauthorized reproduction of this article is prohibited.
KEY POINT
wipes the nose first after a seizure is very focal fields, and, without the h Anterior temporal
typically ipsilateral to the seizure focus. addition of extra anterior temporal discharges may often
A delay often exists in diagnosing electrodes (T1, T2), an interictal spike have a limited field, and
nondominant temporal lobe seizures may be missed (Supplemental Digital additional electrodes
because of preserved awareness and Content 5-2, links.lww.com/CONT/ (T1 and T2) may aid
minimal postictal symptoms. Often the A158). The interictal epileptiform activ- in the detection of
patient is unaware of the ictal automa- ity is typically bilateral,5 even in pa- epileptiform activity.
tisms, and until an observer witnesses a tients with unilateral seizures, but
seizure, the symptoms are often mini- typically one side has a preponderance
mized or ignored by the patient. How- (Supplemental Digital Content 5-3,
ever, once patients are made aware of links.lww.com/CONT/A159). Sleep
the symptoms, they typically can reli- deprivation can often activate focal
ably report their seizures. epileptiform discharges; therefore,
EEG findings. In mesial temporal an EEG with sleep recording is
lobe epilepsy, the interictal EEG is recommended during the diagnostic
associated with anterior temporal workup. The presence of mid or
spikes/sharp waves that are maximum posterior temporal spikes may suggest
in electrodes T1/F7 or T2/F8 (FT9/FT10 a more widespread epileptogenic net-
in the 10/10 system) (Supplemental work even in the presence of clear mesial
Digital Content 5-1, links.lww.com/ temporal pathology. The ictal EEG is
CONT/A157). Often the spikes can have typically characterized by rhythmic
Case 5-1
A 35-year-old woman presented for evaluation after having a seizure associated with loss of
consciousness that resulted in a minor car accident. She did not sustain any injuries during the accident.
Upon questioning, she stated that she first developed a sense of fear and then a butterfly sensation in
her stomach. Ten seconds later, she lost awareness. She reported that she had experienced the aura
sporadically over the last 5 years, and it had increased in frequency over the last 6 months. She denied
prior spells with loss of awareness. According to her partner, when she reported this sensation she
often had oral automatisms; she was able to converse during the events, but her responses were slightly
delayed. Typically, these spells were 45 seconds in duration.
Based on the history of a single complex partial seizure and multiple simple partial seizures
associated with a fear/abdominal aura and oral automatisms with preserved awareness, a diagnosis
of epilepsy was made. To try to define an etiology, an MRI of the brain with and without contrast was
obtained, which demonstrated findings most consistent with a right temporal dysembryoplastic
neuroepithelial tumor (DNET) (Figure 5-3). A routine EEG was obtained, which revealed right anterior
temporal sharp waves (Figure 5-4A). The patient was treated with lamotrigine 150 mg 2 times a day.
Despite maintaining therapeutic blood levels, she continued to experience seizures and was
transitioned to levetiracetam 500 mg 2 times a day and weaned off lamotrigine. She had an initial
seizure-free period of 3 months on levetiracetam monotherapy, but her seizures recurred and she
continued to have four seizures per month.
She was admitted to an epilepsy monitoring unit and underwent continuous video-EEG
monitoring. Her habitual seizures were recorded, and the EEG revealed a right temporal ictal
discharge (Figure 5-4B, 5-4C, and 5-4D). Based on the concordance of her clinical symptomatology,
interictal and ictal EEG, and imaging, a right anterior temporal lobe resection was recommended and
performed. Pathology of the resected lesion showed a DNET. She remained seizure free after the
surgery and was eventually taken off her antiepileptic drugs.
Continued on page 98
Continuum (Minneap Minn) 2016;22(1):94–115 www.ContinuumJournal.com 97
Copyright © American Academy of Neurology. Unauthorized reproduction of this article is prohibited.
Adult Focal Epilepsies
Continued from page 97
FIGURE 5-3 Imaging of the patient in Case 5-1 showing characteristics consistent with right temporal dysembryoplastic
neuroepithelial tumor (DNET). Coronal T1 (A) and fluid-attenuated inversion recovery (FLAIR) (B) and axial
FLAIR (C) MRIs demonstrate a mass lesion in the region of the right amygdala.
FIGURE 5-4 EEGs of the patient in Case 5-1 demonstrating right temporal lobe epilepsy. A, EEG
demonstrates right anterior to midtemporal spikes. Panels BYD reveal a typical right
temporal ictal EEG discharge. The ictal discharge starts with low-amplitude rhythmic
delta activity that evolves on panel C to high-amplitude rhythmic theta before stopping around
the fourth second of panel D.
Continued on page 99
98 www.ContinuumJournal.com February 2016
Copyright © American Academy of Neurology. Unauthorized reproduction of this article is prohibited.
Continued from page 98
FIGURE 5-4 EEGs of the patient in Case 5-1 demonstrating right temporal lobe epilepsy. A, EEG
demonstrates right anterior to midtemporal spikes. Panels BYD reveal a typical
right temporal ictal EEG discharge. The ictal discharge starts with low-amplitude
rhythmic delta activity that evolves on panel C to high-amplitude rhythmic theta before stopping
around the fourth second of panel D.
Continued on page 100
Continuum (Minneap Minn) 2016;22(1):94–115 www.ContinuumJournal.com 99
Copyright © American Academy of Neurology. Unauthorized reproduction of this article is prohibited.
Adult Focal Epilepsies
Continued from page 99
FIGURE 5-4 EEGs of the patient in Case 5-1 demonstrating right temporal lobe epilepsy. A, EEG
demonstrates right anterior to midtemporal spikes. Panels BYD reveal a typical
right temporal ictal EEG discharge. The ictal discharge starts with low-amplitude
rhythmic delta activity that evolves on panel C to high-amplitude rhythmic theta before stopping
around the fourth second of panel D.
Comment. The clinical symptomatology in this patient was consistent with seizures emanating
from the mesial temporal region, and, based on the clinical history alone, a diagnosis of focal
epilepsy could be established. EEG and MRI confirmed the clinical diagnosis and better defined the
location and potential cause of her epilepsy. In this case, the patient had the typical mesial temporal
EEG patterns and imaging consistent with a common benign tumor associated with temporal lobe
epilepsy. In this patient, trials of two therapeutic antiepileptic drugs had failed, therefore fulfilling the
definition of drug-resistant epilepsy.3 The likelihood of long-term seizure freedom with medical
therapy was low at that point (less than 5%), and she was referred for consideration of epilepsy
surgery, which led to successful seizure control.3,4
alpha or theta activity that evolves into ceded by an initial sharp wave or
higher-amplitude rhythmic delta or suppression of the normal EEG
theta activity that may be sharply patterns or epileptiform activity that
contoured or contain discrete spikes was occurring immediately before the
(Supplemental Digital Content 5-4, ictus. Other patterns at onset include
links.lww.com/CONT/A160). Often, rhythmic delta with or without spikes
the initial ictal discharge may be pre- (Supplemental Digital Content 5-5,
100 www.ContinuumJournal.com February 2016
Copyright © American Academy of Neurology. Unauthorized reproduction of this article is prohibited.
Case 5-2
A 40-year-old man presented for evaluation of persistent seizures. The patient began to have seizures
at age 22. He stated that his seizures always started with a burning sensation in his stomach that often
rose up through his chest. This occurred over 10 to 15 seconds, after which he lost awareness.
Observers had noted that he stared blankly, was unresponsive to questions, and had oral and manual
automatisms and unilateral left eyelid blinking during the seizure. The seizures typically lasted for
60 to 90 seconds and were followed by postictal fatigue and confusion that lasted for up to 1 hour.
He had three seizures per month. He was being treated with zonisamide 300 mg/d, and previous
treatments with levetiracetam, carbamazepine, lamotrigine, phenytoin, and topiramate had failed.
He continued to work as a math teacher in a junior high school, but needed rides to and from work
because of his inability to drive. He admitted to mild depression in the past that had not required
treatment, but he denied ever considering suicide. He wished to consider alternative treatment
options since medical therapy had failed.
An MRI of the brain was obtained (Figure 5-5A and 5-5B) and was consistent with left mesial
temporal sclerosis. The patient was admitted to the epilepsy monitoring unit for video-EEG
monitoring (Figure 5-6). The interictal EEG revealed frequent left anterior temporal delta slowing
(Figure 5-6A) and left anterior temporal sharp waves (Figure 5-6B) that were maximal at the T1
electrode in the transverse chain and F7 in the anterior-posterior chain of electrodes. The ictal EEG
revealed a left temporal seizure pattern (Figure 5-6C, 5-6D, 5-6E, and 5-6F). Neuropsychological
evaluation revealed normal visual memory and reduced, but still average, verbal memory.
FIGURE 5-5 Imaging of the patient in Case 5-2 showing left mesial temporal lobe sclerosis. Coronal fluid-attenuated inversion
recovery (FLAIR) (A) and T1 (B) MRIs demonstrate increased signal on T2-weighted imaging and atrophy on
T1-weighted imaging of the hippocampus. Coronal T2 MRI demonstrates postoperative encephalomalacia in the
mesial temporal lobe that was created using a minimally invasive laser thermal ablation technique (C).
Comment. Based on the patient’s seizure symptomatology, interictal and ictal EEG, and imaging, he
was diagnosed with mesial temporal lobe epilepsy secondary to hippocampal sclerosis. He was felt to be a
very good candidate for a left anterior temporal lobectomy, but his reduced verbal memory and need to
function as a math teacher were concerns. The patient underwent a new minimally invasive approach
utilizing stereotactic thermal ablation for the treatment of mesial temporal sclerosis7,8 and has remained
seizure free for 2 years since the operation (Figure 5-5C). He remained on zonisamide 300 mg/d and was
back working as a math teacher 2 weeks after the surgery. Follow-up neuropsychological testing
demonstrated no decline when compared to his preoperative data, and he reported no subjective
cognitive concerns.
Continued on page 102
Continuum (Minneap Minn) 2016;22(1):94–115 www.ContinuumJournal.com 101
Copyright © American Academy of Neurology. Unauthorized reproduction of this article is prohibited.
Adult Focal Epilepsies
Continued from page 101
FIGURE 5-6 EEGs of the patient in Case 5-2 demonstrating left mesial temporal lobe
epilepsy. A, Interictal focal slowing in the left anterior temporal region. B,
Frequent left anterior temporal spikes. Panels CYF reveal a left temporal lobe
seizure that begins with moderate-amplitude rhythmic delta and evolves to higher-amplitude
delta rhythmic spiking, which ceases 12 seconds into panel F.
Continued on page 103
102 www.ContinuumJournal.com February 2016
Copyright © American Academy of Neurology. Unauthorized reproduction of this article is prohibited.
Continued from page 102
FIGURE 5-6 EEGs of the patient in Case 5-2 demonstrating left mesial temporal lobe epilepsy.
A, Interictal focal slowing in the left anterior temporal region. B, Frequent left
anterior temporal spikes. Panels CYF reveal a left temporal lobe seizure that
begins with moderate-amplitude rhythmic delta and evolves to higher-amplitude delta rhythmic
spiking, which ceases 12 seconds into panel F.
Continued on page 104
Continuum (Minneap Minn) 2016;22(1):94–115 www.ContinuumJournal.com 103
Copyright © American Academy of Neurology. Unauthorized reproduction of this article is prohibited.
Adult Focal Epilepsies
Continued from page 103
FIGURE 5-6 EEGs of the patient in Case 5-2 demonstrating left mesial temporal lobe epilepsy.
A, Interictal focal slowing in the left anterior temporal region. B, Frequent left
anterior temporal spikes. Panels CYF reveal a left temporal lobe seizure that begins
with moderate-amplitude rhythmic delta and evolves to higher-amplitude delta rhythmic spiking,
which ceases 12 seconds into panel F.
104 www.ContinuumJournal.com February 2016
Copyright © American Academy of Neurology. Unauthorized reproduction of this article is prohibited.
KEY POINTS
links.lww.com/CONT/A161 and Supple- are normal, and the disease course is h Imaging of the mesial
mental Digital Content 5-6, links.lww. benign for most patients. The exact temporal lobe requires
com/CONT/A162). genetic mutation is not known.10 thin-cut coronal
The seizure discharge may remain imaging with T1- and
limited to the mesial temporal struc- Lateral/Neocortical Temporal T2-weighted images to
tures, but often spreads to the remain- Lobe Epilepsy properly visualize the
der of the ipsilateral hemisphere and Lateral/neocortical temporal lobe epi- key structures.
contralateral temporal lobe. During lepsy is less common than mesial tem- h The symptomatology
the postictal state, unilateral temporal poral lobe epilepsy and less well-defined. associated with
lobe slowing that is typically ipsilateral Because of the rich connections to other neocortical temporal
to the seizure onset may be seen. regions of the brain and the larger total lobe epilepsy is quite
Imaging. An MRI of the brain with- cortical area available to generate seizures, varied and dependent
out contrast is recommended in the the symptomatology, EEG findings, and on the location of the
evaluation of a patient with suspected associated pathologies are varied. seizure onset.
mesial temporal lobe epilepsy. An Clinical features. Seizures originat- h Auditory auras and
enhanced MRI may be used dependent ing from the temporal pole or anterior aphasic seizures are
on local imaging protocols and basal temporal surface often display very suggestive of
suspected etiologies. It is preferable auras and other clinical features that neocortical temporal
to include coronal T1- and T2-weighted lobe epilepsy.
are similar to mesial temporal lobe
images with thin cuts (less than 3 mm) epilepsy as described earlier. This is
through the temporal lobes to properly most likely secondary to early spread
evaluate the hippocampus. Key imag- of the ictal electrographic discharge to
ing characteristics to evaluate for hip- the mesial temporal lobe structures.
pocampal sclerosis are asymmetric size Lateral and perisylvian seizure foci
(smaller on the affected side) and often have features that involve the
increased T2 signal in the hippocam- eloquent cortex in that region, such as
pus consistent with gliosis. Inclusion of aphasic seizures in the dominant tem-
gradient echo imaging may assist in the poral lobe and auditory auras due to
identification of vascular etiologies. involvement of the primary and second-
Fluorodeoxyglucose positron emission ary auditory cortex.6 Seizure onsets near
tomography (FDG-PET) scan imaging the temporooccipital junction may be
has been extensively studied in pa- associated with vertiginous symptoms
tients undergoing presurgical evalua- or visual auras at the onset of the sei-
tion, and unilateral temporal lobe zures. The auras often evolve to bland
hypometabolism ipsilateral to the sei- staring and unresponsiveness followed
zure focus is a positive predictor of a by rapid secondary generalization. As
good outcome after epilepsy surgery.9 the seizures begin to generalize and
FDG-PET scanning is reserved only activate the motor cortex, it is not
for patients who are being consi- uncommon to have unilateral (contra-
dered for epilepsy surgery since the lateral to the seizure focus) clonic
presence or absence of temporal lobe jerking of the face or upper extremity.11
hypometabolism does not impact EEG findings. The interictal and
medical treatment. ictal EEG patterns in lateral/neocortical
Genetic considerations. Familial lobe epilepsy can be similar to those
forms of mesial temporal lobe epi- seen in mesial temporal lobe epilepsy;
lepsy have been described. The ma- however, patients with lateral or
jority of reports describe families with perisylvian foci may have more mid to
psychic auras and simple or complex posterior temporal foci involving elec-
partial seizures. The imaging studies trodes T4/T3 and T6/T5. The activity
Continuum (Minneap Minn) 2016;22(1):94–115 www.ContinuumJournal.com 105
Copyright © American Academy of Neurology. Unauthorized reproduction of this article is prohibited.
Adult Focal Epilepsies
the same MRI techniques described
for mesial temporal lobe epilepsy. The
pathologies associated with this form
of epilepsy can include, but are not
limited to, malformations of cor-
tical development, vascular malfor-
mations, encephalomalacia secondary
to trauma, and tumors (Figure 5-7,
Figure 5-8, and Figure 5-9).11 FDG-PET
scans may show hypometabolism
in the lateral temporal neocortex,
but again, this imaging technique is
reserved for patients undergoing a
presurgical evaluation.
Genetic considerations. Two forms
of genetic lateral temporal lobe epilepsy
have been identified. The familial
form is referred to as autosomal dom-
inant lateral temporal lobe epilepsy or
autosomal dominant partial epilepsy
with auditory features. The sporadic
form is referred to as idiopathic partial
epilepsy with auditory features. Both
FIGURE 5-7 Temporal malformation of cortical forms are associated with partial-onset
development. Coronal T1-weighted MRI
demonstrating a transmantle cortical dysplasia seizures, often with auditory auras, but
in the left mesial temporal region (black arrow) and a they may also have other sensory auras,
periventricular nodular heterotopia in the right mesial
temporal region (white arrow). as described earlier, and frequent sec-
ondary generalization. Imaging is typi-
cally negative, and the EEG findings are
KEY POINT often rapidly propagates to other supportive of a lateral temporal neo-
h Two genetic forms of lobes, and spread to the contralateral cortical focus. The most common
lateral temporal lobe hemisphere typically occurs earlier mutation found in patients with the
epilepsy have been when compared to mesial temporal familial form of the disorder is in
identified: autosomal
lobe epilepsy.11 Foldvary and col- the leucine-rich, glioma inactivated
dominant partial
epilepsy with auditory
leagues12 described a lateralized poly- 1 (LGI1) gene. The familial form has a
features and idiopathic morphic delta pattern in patients with penetrance of approximately 70% to
partial epilepsy with neocortical temporal lobe epilepsy, 80%. How this mutation causes
auditory features. and paroxysmal fast/beta onset activity epilepsy remains unclear at this time.
has also been reported (Supplemental The medical treatment of these pa-
Digital Content 5-7, links.lww.com/ tients is no different than that of
CONT/A163 and Supplemental Digital patients with other forms of tem-
Content 5-8, links.lww.com/CONT/ poral lobe epilepsy, but they typi-
A164). Occasionally the onset is poorly cally have a more benign prognosis
defined but eventually evolves into a and their seizures can be controlled
lateralized temporal lobe ictal dis- with medication.13
charge (Supplemental Digital Content
5-9, links.lww.com/CONT/A165). FRONTAL LOBE EPILEPSY
Imaging. Imaging in lateral/neocortical Frontal lobe epilepsy is the second most
epilepsy should be obtained with common type of focal epilepsy. The
106 www.ContinuumJournal.com February 2016
Copyright © American Academy of Neurology. Unauthorized reproduction of this article is prohibited.
KEY POINT
h Frontal lobe seizures are
typically brief and
associated with various
motor symptoms.
FIGURE 5-8 Posttraumatic encephalomalacia. Coronal fluid-attenuated inversion recovery
(FLAIR) (A) and T1-weighted (B) MRIs demonstrating extensive areas of
encephalomalacia within the right frontal and temporal lobe (blue arrows).
A smaller region of encephalomalacia and gliosis is also noted in the left lateral temporal
lobe (red arrows).
seizure symptomatology associated with volve motor symptomatology, and oc-
frontal lobe epilepsy varies depending cur more often from sleep. The
on the location of the seizure onset zone postictal period is often characterized
within the frontal lobe.14 Compared to by a quick return to normal cognition
temporal lobe seizures, frontal lobe but is occasionally associated with sub-
seizures are often shorter, usually in- tle paresis or frank paralysis of the
FIGURE 5-9 Right temporal cavernous hemangioma. Coronal fluid-attenuated inversion
recovery (FLAIR) (A) and T1-weighted (B) MRIs demonstrate a mass lesion in the
anterior temporal pole, consistent with a cavernous hemangioma.
Continuum (Minneap Minn) 2016;22(1):94–115 www.ContinuumJournal.com 107
Copyright © American Academy of Neurology. Unauthorized reproduction of this article is prohibited.
Adult Focal Epilepsies
involved motor region that resolves frontal lobe regions. Several of the
over time. Frontal lobe seizures can more common presentations are
often appear unusual or bizarre to the discussed later in the article.
untrained observer, and many patients Focal clonic or tonic seizures affecting
are misdiagnosed with psychogenic a limb or region of the trunk without loss
nonepileptic seizures or parasomnias. of consciousness or awareness are highly
suggestive of a seizure focus within the
Clinical Features perirolandic cortex or dorsolateral cor-
With respect to seizures, the frontal tex. Seizures of the perirolandic cortex
lobe can be divided into primary are typically clonic movements limited
motor, supplementary sensorimotor, to one region of the motor homunculus
orbitofrontal, dorsolateral, frontopolar, (eg, face or hand) but can spread to
and opercular regions. Most frontal adjacent regions; this spreading is re-
regions are associated with prominent ferred to as the jacksonian march.
motor phenomena because of the Bilateral asymmetric tonic seizures,
presence of premotor and motor cor- which are described as tonic flexion of
tices within the frontal lobe. However, one arm and extension of the other with
frontopolar and orbitofrontal seizures or without tonic leg involvement, are
can have bland symptomatology, with associated with activation of the supple-
staring, unresponsiveness, and late mentary sensorimotor area located in
motor manifestations (complex autom- Brodmann area 6 on the mesial or dorsal
atisms or versive head or eye move- aspect of the frontal lobe. These seizures
ment) that can be confused with are also referred to as “fencing seizures”
temporal lobe epilepsy. Table 5-1 out- because of the position of the arms.
lines the clinical features that can be They are considered simple partial sei-
seen with seizures from the various zures since patients retain awareness
a
TABLE 5-1 Clinical Manifestations of Frontal Lobe Syndromes
Location Ictal Behavior
Perirolandic or Focal motor seizures with or without jacksonian march,
primary motor speech arrest or dysphasia, vocalization
Supplementary Focal asymmetric tonic posturing, versive movements of head
sensorimotor and eyes, speech arrest, vocalization
Dorsolateral Focal tonic or clonic activity, versive movements of head and
eyes, speech arrest or dysphasia
Orbitofrontal Complex motor automatisms, olfactory hallucinations
and illusions, autonomic features
Anterior Versive movements of head and eyes, forced thinking, initial
frontopolar loss of contact or “absencelike,” speech or motor arrest
Opercular Mastication, salivation, swallowing, laryngeal symptoms;
speech arrest or dysphasia; epigastric aura, fear; autonomic
features; facial clonic activity; gustatory hallucinations
Cingulate Fear, vocalization, emotional or mood changes, complex
motor automatisms, autonomic features
a
Reprinted with permission from Bagla R, Skidmore CT, Neurologist.14 journals.lww.com/
theneurologist/Abstract/2011/05000/Frontal_Lobe_Seizures.1.aspx. B 2011 Lippincott Williams
& Wilkins, Inc.
108 www.ContinuumJournal.com February 2016
Copyright © American Academy of Neurology. Unauthorized reproduction of this article is prohibited.
KEY POINT
despite the bilateral motor manifesta- movement, contralateral to the sei- h The supplementary
tions (Case 5-3). The presence of this zure focus. Occasionally, the entire sensorimotor area has
seizure type is often associated with a body will turn with the head and eyes, bilateral sensory and
mesial frontal or dorsolateral seizure and the patient may spin around the motor representation,
focus that preferentially spreads to long axis of his or her body several and seizures generated
activate the supplementary sensorimo- times. Involvement of the Broca re- from this region have
tor area. Seizures associated with large gion can result in an aphasic seizure, bilateral motor
vigorous proximal muscle movements, and activation of the negative motor symptoms with
such as bicycling movements of the legs cortex, located on the posterior aspect preserved awareness.
or windmilling of the arms, are often of the inferior frontal gyrus or near the
associated with activation of the supplementary sensorimotor area, can
frontopolar or orbitofrontal region. These produce seizures during which the
seizures have also been described in patient is unable to move a limb or
the literature as hypermotor or hyper- body part.14,15
kinetic seizures. Activation of other
specific structures in the frontal lobe EEG Findings
can produce very specific seizure symp- The interictal EEG associated with fron-
toms. For example, activation of the tal lobe epilepsy can vary depending
frontal eye field located in the premotor on the location of the seizure focus.
cortex results in forced head and eye Overall, in a large surgical series, in-
version, unnatural upward and lateral terictal epileptiform activity was within
Case 5-3
A 25-year-old man presented for evaluation after having a generalized tonic-clonic seizure at home. He was
previously diagnosed with psychogenic nonepileptic seizures (PNES) at age 23. When asked to describe his
events, he stated that he often woke up from sleep and had tonic posturing of his arms and legs. He
specifically stated that his right arm was extended and his left arm was flexed toward his chest. At the end of
the seizure, he had several clonic jerks of his entire body. He stated that he is fully aware throughout the
events but cannot speak because he cannot control his mouth. A typical seizure lasted for approximately
20 seconds, and he often had clusters with 5 to 10 seizures per cluster. Immediately following the event, he
felt weak, but within 30 to 60 seconds, he felt normal. He had been evaluated at a local hospital and had a
normal EEG during one of these events. Based on the fact that he recalled the events despite having
bilateral motor manifestations and a negative EEG during one of the events, a diagnosis of PNES had been
made, and he was treated by a behavioral psychologist after that diagnosis, without relief.
Comment. The symptomatology described in the case is consistent with a bilateral asymmetric tonic
(fencing) seizure. These seizures are common for mesial frontal lobe epilepsy with activation of the
supplementary motor cortex. The supplementary motor cortex has bilateral motor and sensory
representation that has somatotopic organization. Therefore, a unilateral ictal discharge could cause
bilateral motor manifestations without impairing consciousness. Because of the limitations of scalp EEG, it is
not uncommon for patients, such as this patient, with mesial frontal lobe epilepsy to have normal EEGs.
A montage that included electrodes Fz, Cz, and Pz could have increased the diagnostic yield of the EEG. Even
in the face of a normal EEG, the stereotyped brief events are not typical of PNES, and PNES should
always remain a diagnosis of exclusion.
This patient was admitted to an epilepsy monitoring unit for continuous EEG monitoring, and
had an MRI of the brain that revealed a small cortical dysplasia in the left mesial frontal region
(Figure 5-10). During his admission, 15 stereotyped seizures were captured as described earlier. The
EEG revealed the onset of rhythmic beta in the central region, maximum at electrode Cz (Figure 5-11).
The patient was started on carbamazepine and was seizure free at last follow-up.
Continued on page 110
Continuum (Minneap Minn) 2016;22(1):94–115 www.ContinuumJournal.com 109
Copyright © American Academy of Neurology. Unauthorized reproduction of this article is prohibited.
Adult Focal Epilepsies
Continued from page 109
FIGURE 5-10 Imaging of the patient in Case 5-3 showing left frontal cortical dysplasia.
Coronal fluid-attenuated inversion recovery (FLAIR) MRIs demonstrate an area
of increased cortical thickness in the mesial aspect of the superior frontal gyrus
(A, red arrow) and an associated tail of abnormality that extends toward the ventricle (B, blue
arrow). These findings are consistent with a cortical migrational abnormality.
FIGURE 5-11 EEG of the patient in Case 5-3 demonstrating mesial frontal seizure. During the third second of the EEG, a
paroxysmal fast discharge develops at electrode Cz (arrow). This finding was reproducible with all seizures
and always preceded clinical onset.
110 www.ContinuumJournal.com February 2016
Copyright © American Academy of Neurology. Unauthorized reproduction of this article is prohibited.
KEY POINT
the frontal lobe in 65% of patients, unilaterally before the “generalized” h The interictal and ictal
outside the frontal lobe in 37%, contra- discharge, which, if stereotyped, pro- EEG associated with
lateral to the seizure focus in 17%, and vides a clue to the lateralization of the frontal lobe epilepsy can
generalized in 24%.16 Dorsolateral and epileptiform activity. Paroxysmal fast be challenging from a
frontopolar epilepsies are most likely to (beta frequency) activity is also a technical perspective
show focal epileptiform activity since the feature of the interictal EEG that has because of movement
typical scalp EEG electrodes are imme- been associated with frontal lobe epi- artifact and the limited
diately over these locations. However, lepsy (Supplemental Digital Content ability of scalp
even in these regions, epileptiform 5-10, links.lww.com/CONT/A166).18 electrodes to record
activity may not be recorded with scalp The ictal EEG is also challenging from seizure locations
(basal frontal,
electrodes because of the size of the in frontal lobe epilepsy. In the sur-
interhemispheric, and
cortical region involved. Based on gical series published by Jeha and
opercular regions) and
simultaneous scalp and intracranial re- colleagues, 16 the ictal discharge in cases with limited
cordings, an area of at least 6 cm2 must was localized to the frontal lobe in cortical involvement.
fire simultaneously to generate a depo- 51% of cases, unilateral in 14%, and
larization that can be recorded reliably generalized in 21%. Because of the
with scalp electrodes.17 This technical prominent motor manifestations asso-
limitation is further exacerbated when ciated with frontal lobe seizures, ictal
the distance between the generator and recordings are often obscured by
the recording electrode is increased, muscle artifact (EMG). This makes it
such as with foci involving the inter- difficult to see the initial ictal onset
hemispheric region, orbitofrontal cortex, and spread even after the digital EEG
or frontal opercular cortex. The addi- is filtered. Other challenges include
tion of extra 10-10 electrodes, in partic- the technical limitations mentioned
ular over the midline region, may earlier with respect to recording
increase the likelihood of recording ictal epileptiform activity from deeper
interictal epileptiform discharges and foci and simple partial seizures that
ictal EEG changes. Because of the rich may involve a limited amount of
connections to and location near the cortex. These limitations are not
temporal lobe, orbitofrontal epilepsy unique to frontal lobe epilepsy and
can be associated with epileptiform apply to all forms of focal epilepsy.
activity in the anterior temporal elec- Despite these technical limitations,
trodes. The overlap between the clinical several patterns have been associated
symptomatology and interictal EEG with frontal lobe epilepsy, includ-
between orbitofrontal lobe epilepsy ing diffuse electrodecrement, focal
and temporal lobe epilepsy must be rhythmic fast activity, and bilateral
considered, especially in patients with hypersynchrony.18 An example of a
nonlesional epilepsy. frontal ictal discharge can be seen in
Bilateral secondary hypersynchrony Supplemental Digital Content 5-11,
has been described in patients with links.lww.com/CONT/A167.
frontal lobe epilepsy and is character-
ized by the rapid spread of the interictal Imaging
spike between the two hemispheres. MRI of the brain without contrast
These epileptiform discharges often should be obtained in the coronal and
appear as generalized spikes, poly- axial plane and should include thin
spikes, or spike and wave, and it is slices (less than 3 mm). As described
important not to misdiagnose a patient for mesial temporal lobe epilepsy, the
with generalized epilepsy. Often an MRI should include T1- and T2-
initial spike/sharp wave can be seen weighted images, and the use of
Continuum (Minneap Minn) 2016;22(1):94–115 www.ContinuumJournal.com 111
Copyright © American Academy of Neurology. Unauthorized reproduction of this article is prohibited.
Adult Focal Epilepsies
KEY POINTS
h MRI scans should contrast will vary depending on poten- lobe epilepsy had regions of hypo-
include thin-cut T1- and tial etiology and local protocols. In two metabolism identified. This compared
T2-weighted images surgical series, malformations of corti- to 67% for temporal lobe epilepsy and
in the axial and cal development were the most com- 61% for nontemporal and nonfrontal
coronal planes. mon etiology associated with frontal lobe epilepsies. In the frontal lobe cases with
h Malformations of epilepsy (40% to 58%) (Figure 5-12), hypometabolism, the findings were on-
cortical development compared to tumor (10% to 19%), ly helpful in making a surgical decision
are the most common vascular (3% to 7%), and encephalo- in 38% of cases.20 In the frontal lobe
etiology associated with malacia (10% to 12%) (Figure 5-8). surgical series mentioned earlier, when
frontal lobe seizures in Based on imaging or pathology, 10% PET was performed, the results were
large surgical series. to 31% of patients had no identifiable abnormal in 25% of the patients in the
h Autosomal dominant etiology. MRI scans were nonlesional in Lazow and colleagues series19 and in
nocturnal frontal lobe 26% to 45% of patients.16,19 Use of 76% of the patients in the Jeha and
epilepsy is associated FDG-PET imaging for presurgical plan- colleagues series.16
with a genetic mutation ning is less likely to reveal a deficit in
in the neuronal nicotinic patients with frontal lobe epilepsy com- Genetic Considerations
acetylcholine receptor. pared to patients with temporal lobe Autosomal dominant nocturnal frontal
epilepsy. However, in a 2012 series of lobe epilepsy is a well-described genetic
194 patients (34% with frontal lobe frontal lobe epilepsy syndrome. It is
epilepsy), 51% of patients with frontal characterized by brief motor seizures that
are stereotyped and occur multiple times
per night and is associated with normal
intelligence and a normal neurologic
examination between attacks. A family
history of similar seizures often exists,
although patients are often misdiagnosed
as having parasomnias, and a detailed
history regarding family members is
needed. The most commonly associated
abnormality with this disorder is in the
neuronal nicotinic acetylcholine recep-
tor, and the penetrance is variable, 29%
to 100% in the literature.21
PARIETAL AND OCCIPITAL
LOBE EPILEPSY
Epilepsy associated with the parietal and
occipital lobes is much less common
than temporal or frontal lobe epilepsy.
Due to the rich connections between
the parietal and occipital lobes and the
frontal and temporal lobes, it can often
be challenging to reliably identify these
patients without the use of invasive
FIGURE 5-12 Left frontal malformation of cortical
development. Coronal T1-weighted MRI
techniques. Visual auras are more com-
demonstrates a large malformation of mon in occipital lobe epilepsy and are
cortical development in the left frontal region typically elemental auras. Other symp-
(arrow). Associated enlargement of the left lateral
horn is seen. toms may include ictal blindness,
blinking, and ocular movement.22 More
112 www.ContinuumJournal.com February 2016
Copyright © American Academy of Neurology. Unauthorized reproduction of this article is prohibited.
KEY POINTS
complex visual scenes or objects may lepsy. In studies that reviewed occipital h Occipital lobe epilepsy is
be seen in occipital lobe epilepsy and and parietal lobe epilepsy, malforma- often associated with a
suggest spread of the seizure discharge tions of cortical development and tu- visual aura as the first
to the temporooccipital region. Sei- mors were more common in occipital clinical sign.
zure foci below the calcarine sulcus will lobe epilepsy,25 while tumors were the h Parietal and occipital
preferentially spread to the temporal most common etiology for parietal lobe epilepsies
region and may generate complex lobe epilepsy.24,26 frequently spread to
partial seizures with symptomatology adjacent cortical regions
and ictal EEG typical for temporal lobe INSULAR AND CINGULATE and may have features
epilepsy.23 Foci above the calcarine LOBE EPILEPSY of seizures commonly
sulcus preferentially spread to the Focal epilepsy involving the insular associated with temporal
parietal and frontal lobes. cortex and cingulate is uncommon. or frontal seizures.
Parietal lobe epilepsy is often associ- Insular seizures should be suspected h Parietal lobe epilepsy is
ated with somatosensory auras that are in patients who present with seizure often associated with a
typically contralateral to the seizure symptoms involving contraction or a somatosensory aura as
focus but can be bilateral.24 Often choking sensation of the oropharynx/ the first clinical sign.
patients with parietal lobe epilepsy will larynx or a painful somatosensory h Insular and cingulate
display symptoms associated with aura.27,28 Cingulate epilepsy is very lobe epilepsy are rare
spread to the motor cortex, premotor difficult to differentiate clinically from and should be carefully
regions, or temporooccipital region. the other focal epilepsies. Because of considered in patients
Because of the rapid spread of the ictal the rich anatomic connections to the with nonlesional
discharge to distant regions, it is very frontal and temporal regions, cingulate epilepsy since the
symptomatology often
important to take a detailed history to seizures often mimic seizures originat-
overlaps with other
try to identify if the first symptom is a ing from these structures.13 In the
types of focal epilepsy.
somatosensory aura. absence of a clear lesion on MRI, these
The EEG findings in occipital and cases often require invasive EEG mon-
parietal lobe epilepsy can be challeng- itoring to confirm an ictal onset in the
ing. It is possible to see focal interictal cingulate or insular region.
spikes and ictal onsets, but it is also
common to see interictal and ictal CONCLUSION
patterns localizing to the temporal The focal epilepsies are very common
and frontal regions because of the rich in adulthood, and, while overlap in
synaptic connections. With midline clinical presentations often exists, each
parietal or occipital foci, the possibility region of the cortex often has several
exists for false localization to the distinct clinical presentations, EEG
wrong hemisphere because of the findings, or imaging findings that may
orientation of the electrical dipole aid in establishing an accurate diagno-
pointing to the contralateral hemi- sis. The clinical history of the seizure
sphere. In addition, the possibility of event either as reported by the patient
bilateral hypersynchrony, described or observers, or through watching a
earlier for frontal lobe epilepsy, also video of the event, is extremely impor-
exists. An example of the onset of a tant. The terms complex partial and
left parietal seizure that was associated simple partial are certainly helpful in
with a cortical dysplasia is shown in defining whether a patient has lost
Supplemental Digital Content 5-12, awareness or consciousness or not,
links.lww.com/CONT/A168. but do not reflect the details that
The causes of parietal and occipital permit a more accurate diagnosis.
lobe epilepsies are similar to those Imaging and EEG are critical in
mentioned earlier for frontal lobe epi- evaluating patients with focal epilepsy,
Continuum (Minneap Minn) 2016;22(1):94–115 www.ContinuumJournal.com 113
Copyright © American Academy of Neurology. Unauthorized reproduction of this article is prohibited.
Adult Focal Epilepsies
but one must be cautious to ensure American Epilepsy Society 68th Annual Meeting;
December 5Y9. 2014; Seattle, Washington.
that the study is properly designed to
ensure that common pathologies or 9. LoPinto-Khoury C, Sperling MR, Skidmore C,
et al. Surgical outcome in PET-positive,
EEG findings are not missed inadver- MRI-negative patients with temporal lobe
tently. This includes properly imaging epilepsy. Epilepsia 2012;53(2):342Y348.
the temporal lobes with coronal views doi:10.1111/j.1528-1167.2011.03359.x.
and obtaining a thin-cut MRI for all 10. Crompton DE, Scheffer IE, Taylor I, et al.
patients with focal epilepsy. The stan- Familial mesial temporal lobe epilepsy: a
benign epilepsy syndrome showing complex
dard MRI used for stroke imaging is inheritance. Brain 2010;133(11):3221Y3231.
not sufficient since epileptogenic le- doi:10.1093/brain/awq251.
sions may often be subtle and missed 11. Kennedy JD, Schuele SU. Neocortical
on an MRI with thicker cuts. The temporal lobe epilepsy. J Clin Neurophysiol
addition of key midline or temporal 2012;29(5):366Y370. doi:10.1097/
WNP.0b013e31826bd78b.
electrodes in the proper clinical con-
12. Foldvary N, Lee N, Thwaites G, et al. Clinical
text will also assist in making an and electrographic manifestations of lesional
accurate diagnosis. neocortical temporal lobe epilepsy. Neurology
1997;49(3):757Y763.
13. Michelucci R, Pasini E, Nobile C. Lateral
REFERENCES temporal lobe epilepsies: clinical and genetic
1. Wirrell E. Infantile, childhood, and adolescent features. Epilepsia 2009;50(suppl 5):52Y54.
epilepsies. Continuum (Minneap Minn) doi:10.1111/j.1528-1167.2009.02122.x.
2016;22(1 Epilepsy):60Y93.
14. Bagla R, Skidmore CT. Frontal lobe seizures.
2. Scott RC, King MD, Gadian DG, et al. Neurologist 2011;17(3):125Y135.
Hippocampal abnormalities after prolonged doi:10.1097/NRL.0b013e31821733db.
febrile convulsion: a longitudinal MRI study.
Brain 2003;126(pt 11):2551Y2557. 15. Lee RW, Worrell GA. Dorsolateral frontal
lobe epilepsy. J Clin Neurophysiol 2012;29
3. Kwan P, Schachter SC, Brodie MJ. Drug-resistant (5):379Y384. doi:10.1097/
epilepsy. N Engl J Med 2011;365(10):919Y926. WNP.0b013e31826b3c7c.
doi:10.1056/NEJMra1004418.
16. Jeha LE, Najm I, Bingaman W, et al. Surgical
4. Engel J Jr, Wiebe S, French J, et al. Practice outcome and prognostic factors of frontal
parameter: temporal lobe and localized lobe epilepsy surgery. Brain 2007;130(pt 2):
neocortical resections for epilepsy: report of 574Y584.
the Quality Standards Subcommittee of the
17. Tao JX, Baldwin M, Hawes-Ebersole S,
American Academy of Neurology, in Association
Ebersole JS. Cortical substrates of scalp EEG
with the American Epilepsy Society and the
epileptiform discharges. J Clin Neurophysiol
American Association of Neurological
2007;24(2):96Y100.
Surgeons. Neurology 2003;60(4):538Y547.
18. Kutsy RL. Focal extratemporal epilepsy:
5. Tatum WO 4th. Mesial temporal lobe
clinical features, EEG patterns, and surgical
epilepsy. J Clin Neurophysiol 2012;29
approach. J Neurol Sci 1999;166(1):1Y15.
(5):356Y365. doi:10.1097/
WNP.0b013e31826b3ab7. 19. Lazow SP, Thadani VM, Gilbert KL, et al.
6. Loddenkemper T, Kotagal P. Lateralizing Outcome of frontal lobe epilepsy surgery.
signs during seizures in focal epilepsy. Epilepsia 2012;53(10):1746Y1755.
Epilepsy Behav 2005;7(1):1Y17. doi:10.1111/j.1528-1167.2012.03582.x.
7. Gross RE, Mahmoudi B, Riley JP. Less is more: 20. Rathore C, Dickson JC, Teoto nio R, et al.
novel less-invasive surgical techniques for The utility of 18F-fluorodeoxyglucose PET
mesial temporal lobe epilepsy that minimize (FDG PET) in epilepsy surgery. Epilepsy Res
cognitive impairment. Curr Opin Neurol 2014;108(8):1306Y1314. doi:10.1016/j.
2015;28(2):182Y191. doi:10.1097/ eplepsyres.2014.06.012.
WCO.0000000000000176.
21. Ferini-Strambi L, Sansoni V, Combi R.
8. Ashwini S, Chengyuan W, Michael S, et al. Nocturnal frontal lobe epilepsy and the
Stereotactic laser ablation: how much acetylcholine receptor. Neurologist 2012;18
hippocampus and amygdala ablation (6):343Y349. doi:10.1097/
volume optimizes seizure freedom [abstract]. NRL.0b013e31826a99b8.
114 www.ContinuumJournal.com February 2016
Copyright © American Academy of Neurology. Unauthorized reproduction of this article is prohibited.
22. Salanova V, Andermann F, Olivier A, et al. 25. Aykut-Bingol C, Bronen RA, Kim JH, et al.
Occipital lobe epilepsy: electroclinical Surgical outcome in occipital lobe epilepsy:
manifestations, electrocorticography, implications for pathophysiology. Ann Neurol
cortical stimulation and outcome in 42 1998;44(1):60Y69.
patients treated between 1930 and 1991. 26. Salanova V, Andermann F, Rasmussen T, et al.
Surgery of occipital lobe epilepsy. Brain Tumoural parietal lobe epilepsy. Clinical
1992;115(pt 6):1655Y1680. manifestations and outcome in 34 patients
23. Adcock JE, Panayiotopoulos CP. Occipital lobe treated between 1934 and 1988. Brain 1995;
seizures and epilepsies. J Clin Neurophysiol 118(pt 5):1289Y1304.
2012;29(5):397Y407. doi:10.1097/WNP. 27. Kriegel MF, Roberts DW, Jobst BC. Orbitofrontal
0b013e31826c98fe. and insular epilepsy. J Clin Neurophysiol 2012;29
24. Salanova V, Andermann F, Rasmussen T, et al. (5):385Y391. doi:10.1097/WNP.
Parietal lobe epilepsy. Clinical manifestations 0b013e31826bd82e.
and outcome in 82 patients treated surgically 28. Montavont A, Mauguière F, Mazzola L, et al.
between 1929 and 1988. Brain 1995;118(pt 3): On the origin of painful somatosensory
607Y627. seizures. Neurology 2015;84(6):594Y601.
Continuum (Minneap Minn) 2016;22(1):94–115 www.ContinuumJournal.com 115
Copyright © American Academy of Neurology. Unauthorized reproduction of this article is prohibited.
Vous aimerez peut-être aussi
- Back To Basics: The Pathophysiology of Epileptic Seizures: A Primer For PediatriciansDocument12 pagesBack To Basics: The Pathophysiology of Epileptic Seizures: A Primer For Pediatriciansgerson altamiranoPas encore d'évaluation
- The Diagnosis and Management of Epilepsy: Journal of Tropical Pediatrics January 2002Document9 pagesThe Diagnosis and Management of Epilepsy: Journal of Tropical Pediatrics January 2002Roy MarechaPas encore d'évaluation
- Bianda Axanditya 22010110130181 Bab2ktiDocument10 pagesBianda Axanditya 22010110130181 Bab2ktimeiutaPas encore d'évaluation
- Re Ex Myoclonic Epilepsy of Infancy: Seizures Induced by Tactile StimulationDocument8 pagesRe Ex Myoclonic Epilepsy of Infancy: Seizures Induced by Tactile StimulationOziPas encore d'évaluation
- Focal Epilepsy Causes and Clinical Features - UpToDateDocument44 pagesFocal Epilepsy Causes and Clinical Features - UpToDateBenjamínGalvanPas encore d'évaluation
- Quero L Pascual 2007Document8 pagesQuero L Pascual 2007Donald CabreraPas encore d'évaluation
- Frontal Lobe SeizuresDocument11 pagesFrontal Lobe Seizuresjhonny churata huarachiPas encore d'évaluation
- Classification: Epilepsy (Sometimes Referred To As A Seizure Disorder) Is A Common ChronicDocument11 pagesClassification: Epilepsy (Sometimes Referred To As A Seizure Disorder) Is A Common ChronicChristian BartolomePas encore d'évaluation
- Aroxysmal Isorders: Approach To Paroxysmal DisordersDocument46 pagesAroxysmal Isorders: Approach To Paroxysmal DisordersblwPas encore d'évaluation
- Seizure: Alberto Verrotti, Pasquale Striano, Vincenzo Belcastro, Sara Matricardi, Maria Pia Villa, Pasquale ParisiDocument5 pagesSeizure: Alberto Verrotti, Pasquale Striano, Vincenzo Belcastro, Sara Matricardi, Maria Pia Villa, Pasquale ParisiMirachel AugustPas encore d'évaluation
- Epilepsia - 2022 - Specchio - International League Against Epilepsy Classification and Definition of Epilepsy SyndromesDocument45 pagesEpilepsia - 2022 - Specchio - International League Against Epilepsy Classification and Definition of Epilepsy SyndromesIngrid RivadeneiraPas encore d'évaluation
- Seizure and Epilepsy July 2014Document28 pagesSeizure and Epilepsy July 2014ashuPas encore d'évaluation
- L Kleffner InglesDocument11 pagesL Kleffner InglesBombinTallerdeTeatroPas encore d'évaluation
- C R C S: Urrent Eview in Linical CienceDocument7 pagesC R C S: Urrent Eview in Linical CienceAdriel Chandra AngPas encore d'évaluation
- Epilepsia focalDocument17 pagesEpilepsia focalVíctor CalzadaPas encore d'évaluation
- Epilepsy - Definition, Classification, Pathophysiology, and EpidemiologyDocument7 pagesEpilepsy - Definition, Classification, Pathophysiology, and EpidemiologyMartín FleiPas encore d'évaluation
- Epilepsy Syndromes inDocument24 pagesEpilepsy Syndromes inEdwin AlvarezPas encore d'évaluation
- Orofacial Reflex Myocloni. Definition, Relation To Epilepsy Syndromes, Nosological and Prognosis Significance. A Focused ReviewDocument5 pagesOrofacial Reflex Myocloni. Definition, Relation To Epilepsy Syndromes, Nosological and Prognosis Significance. A Focused ReviewJuliana AparcanaPas encore d'évaluation
- Fneur 12 775781Document8 pagesFneur 12 775781ximena sanchezPas encore d'évaluation
- The Neurobiology of EpilepsyDocument11 pagesThe Neurobiology of EpilepsyCharpapathPas encore d'évaluation
- Infantile, Childhood, and Adolescent Epilepsies.9Document34 pagesInfantile, Childhood, and Adolescent Epilepsies.9Silviana IliePas encore d'évaluation
- Biology: Investigatory ProjectDocument22 pagesBiology: Investigatory ProjectNarayana BaipadithayaPas encore d'évaluation
- Epilepsy: Ever-Changing States of Cortical Excitability: R. A. B. Badawy, D. R. Freestone, A. Lai AND M. J. CookDocument11 pagesEpilepsy: Ever-Changing States of Cortical Excitability: R. A. B. Badawy, D. R. Freestone, A. Lai AND M. J. CooknurhikmahPas encore d'évaluation
- Sleep and Epilepsy Syndromes: Bernhard SchmittDocument9 pagesSleep and Epilepsy Syndromes: Bernhard Schmittmarcoteixa22Pas encore d'évaluation
- NCBI Infantil Spasme PDFDocument5 pagesNCBI Infantil Spasme PDFRiri Dwi AnggaraniPas encore d'évaluation
- Neonatal Seizures: Causes, Clinical Features and PrognosisDocument5 pagesNeonatal Seizures: Causes, Clinical Features and PrognosiscainzkeinzPas encore d'évaluation
- 2017 Helmstaedter Epilepsy and Cognition A Biderctional RelationshipDocument7 pages2017 Helmstaedter Epilepsy and Cognition A Biderctional RelationshipManuel CarreonPas encore d'évaluation
- Neonatal Seizure OriginalDocument47 pagesNeonatal Seizure Originalsreekala100% (1)
- Genetic and Therapeutic Review of Progressive Myoclonic EpilepsiesDocument10 pagesGenetic and Therapeutic Review of Progressive Myoclonic EpilepsieschintanPas encore d'évaluation
- Epilepsi 1Document12 pagesEpilepsi 1Ima SoniaPas encore d'évaluation
- Epileptic Disorders - 2023 - Nascimento - Focal Epilepsies Update On Diagnosis and ClassificationDocument17 pagesEpileptic Disorders - 2023 - Nascimento - Focal Epilepsies Update On Diagnosis and ClassificationIvan MihailovicPas encore d'évaluation
- 25 Patofisiologi EpilepsiDocument13 pages25 Patofisiologi EpilepsiWisnuBaskoroPas encore d'évaluation
- Jurnal Reading Epilepsi (Jenifer Johana Paath 201383019)Document28 pagesJurnal Reading Epilepsi (Jenifer Johana Paath 201383019)jenifer paathPas encore d'évaluation
- Sleep and Epilepsy: Sejal V. Jain, MD, and Sanjeev V. Kothare, MDDocument7 pagesSleep and Epilepsy: Sejal V. Jain, MD, and Sanjeev V. Kothare, MDRezky 'kiki' Oktarianti SyahputriPas encore d'évaluation
- Sleep and epilepsy relationship explored through EEG studiesDocument26 pagesSleep and epilepsy relationship explored through EEG studiesrifki irsyadPas encore d'évaluation
- Ref Ref 2Document5 pagesRef Ref 2melly adityaPas encore d'évaluation
- Epidemiology of Epilepsy: DefinitionsDocument13 pagesEpidemiology of Epilepsy: DefinitionsdyaksaniPas encore d'évaluation
- Benign and Severe Early Life Seizures A Round in The First Year of LifeDocument11 pagesBenign and Severe Early Life Seizures A Round in The First Year of LifeClemente PoncePas encore d'évaluation
- Myoclonus and Myoclonic Seizures: Hirokazu OGUNIDocument2 pagesMyoclonus and Myoclonic Seizures: Hirokazu OGUNIPeby M. RamdhanPas encore d'évaluation
- Epilepsy, Cognition, and BehaviorDocument8 pagesEpilepsy, Cognition, and BehaviorliliPas encore d'évaluation
- Dravet Syndrome and SCN1A Disorders - PaediatricFOAMDocument14 pagesDravet Syndrome and SCN1A Disorders - PaediatricFOAMshaik saifuddinPas encore d'évaluation
- Epi Gral 2018 1Document92 pagesEpi Gral 2018 1Anonimo DesconocidoPas encore d'évaluation
- 7b4c PDFDocument5 pages7b4c PDFWinsen HaryonoPas encore d'évaluation
- The Best Evidence For Progressive Myoclonic Epilepsy - A Pathway To Precision TherapyDocument11 pagesThe Best Evidence For Progressive Myoclonic Epilepsy - A Pathway To Precision TherapyHabib G. Moutran BarrosoPas encore d'évaluation
- Patophysiology of EpilepsyDocument14 pagesPatophysiology of EpilepsyMurni Sri HastutiPas encore d'évaluation
- Mesial Temporal Lobe Epilepsy: Nvited EviewDocument10 pagesMesial Temporal Lobe Epilepsy: Nvited EviewDonald CabreraPas encore d'évaluation
- Bell's Palsy Guide: Causes, Symptoms and RecoveryDocument4 pagesBell's Palsy Guide: Causes, Symptoms and RecoveryAyuPas encore d'évaluation
- NEUROLOGY SYMPOSIUMDocument6 pagesNEUROLOGY SYMPOSIUMSantiago Chávez BolañosPas encore d'évaluation
- 753 FullDocument17 pages753 FullPhilip Takato TengPas encore d'évaluation
- J Child Neurol 2001 Fennell 58 63Document6 pagesJ Child Neurol 2001 Fennell 58 63Elena GatoslocosPas encore d'évaluation
- Hypsarrythmia in IsDocument20 pagesHypsarrythmia in IsAndrew SantosoPas encore d'évaluation
- Understanding Epileptic SeizuresDocument19 pagesUnderstanding Epileptic SeizuresRiki AntoPas encore d'évaluation
- Abdulsalam Mohammed Yakasai Physiotherapy Intervention in Children With EpiDocument17 pagesAbdulsalam Mohammed Yakasai Physiotherapy Intervention in Children With Epinaveen chaudharyPas encore d'évaluation
- Ambulatory Seminar.... Epilepsy..KhalidDocument68 pagesAmbulatory Seminar.... Epilepsy..KhalidAliyi MuktarPas encore d'évaluation
- What Is Lennox-Gastaut Syndrome in The Modern Era?: Hirokazu OguniDocument2 pagesWhat Is Lennox-Gastaut Syndrome in The Modern Era?: Hirokazu OguniWinsen HaryonoPas encore d'évaluation
- Biology Investigatory ProjectDocument27 pagesBiology Investigatory ProjectRahul Choudhary66% (38)
- E148 FullDocument17 pagesE148 Fullpatricx10100% (1)
- Neurology Multiple Choice Questions With Explanations: Volume ID'EverandNeurology Multiple Choice Questions With Explanations: Volume IÉvaluation : 4 sur 5 étoiles4/5 (7)
- Childhood Seized: Linguistic Considerations of the Effects of Epilepsy on Cognition in Children with Benign Childhood EpilepsyD'EverandChildhood Seized: Linguistic Considerations of the Effects of Epilepsy on Cognition in Children with Benign Childhood EpilepsyPas encore d'évaluation
- Combined Riluzole and Sodium Phenylbutyrate Therapy in Transgenic Amyotrophic Lateral Sclerosis MiceDocument11 pagesCombined Riluzole and Sodium Phenylbutyrate Therapy in Transgenic Amyotrophic Lateral Sclerosis MicezhoujPas encore d'évaluation
- Design, Power, and Interpretation of Studies in The Standard Murine Model of ALSDocument13 pagesDesign, Power, and Interpretation of Studies in The Standard Murine Model of ALSzhoujPas encore d'évaluation
- Trial of Sodium Phenylbutyrate-Taurursodiol For Amyotrophic Lateral SclerosisDocument12 pagesTrial of Sodium Phenylbutyrate-Taurursodiol For Amyotrophic Lateral SclerosiszhoujPas encore d'évaluation
- Retinal and Optic Nerve Ischemia PDFDocument19 pagesRetinal and Optic Nerve Ischemia PDFzhoujPas encore d'évaluation
- Nejmoa1916945 ProtocolDocument282 pagesNejmoa1916945 ProtocolzhoujPas encore d'évaluation
- Nejmoa1916945 AppendixDocument34 pagesNejmoa1916945 AppendixzhoujPas encore d'évaluation
- Incremental Gains in The Battle Against Als: Michael Benatar, M.D., PH.D., and Michael P. Mcdermott, PH.DDocument2 pagesIncremental Gains in The Battle Against Als: Michael Benatar, M.D., PH.D., and Michael P. Mcdermott, PH.DzhoujPas encore d'évaluation
- Incremental Gains in The Battle Against Als: Michael Benatar, M.D., PH.D., and Michael P. Mcdermott, PH.DDocument2 pagesIncremental Gains in The Battle Against Als: Michael Benatar, M.D., PH.D., and Michael P. Mcdermott, PH.DzhoujPas encore d'évaluation
- Ah 1 N 1Document9 pagesAh 1 N 1DareRaymondPas encore d'évaluation
- Nfluenza-Associated Encephalitis Encephalopathy Identified by The Australian Childhood Encephalitis Study 2013-2015.Document6 pagesNfluenza-Associated Encephalitis Encephalopathy Identified by The Australian Childhood Encephalitis Study 2013-2015.zhoujPas encore d'évaluation
- Episodic Syndromes That May Be Associated With Migraine PDFDocument7 pagesEpisodic Syndromes That May Be Associated With Migraine PDFzhoujPas encore d'évaluation
- Influenza-Associated Neurological Complications During 2014â "2017 in TaiwanDocument8 pagesInfluenza-Associated Neurological Complications During 2014â "2017 in TaiwanzhoujPas encore d'évaluation
- Sidmans Neuroanatomy CurriculumDocument1 pageSidmans Neuroanatomy CurriculumzhoujPas encore d'évaluation
- Autoimmune Neurology of The Central Nervous System PDFDocument27 pagesAutoimmune Neurology of The Central Nervous System PDFzhoujPas encore d'évaluation
- Acute Encephalopathy, InfluenzaDocument7 pagesAcute Encephalopathy, InfluenzazhoujPas encore d'évaluation
- A Review On The Clinical Spectrum and Natural History of Human InfluenzaDocument10 pagesA Review On The Clinical Spectrum and Natural History of Human InfluenzazhoujPas encore d'évaluation
- GbsDocument8 pagesGbszhoujPas encore d'évaluation
- Chronic Axonal PolyneuropathyDocument19 pagesChronic Axonal PolyneuropathyzhoujPas encore d'évaluation
- A Paediatric Influenza Update 100 Years PDFDocument10 pagesA Paediatric Influenza Update 100 Years PDFzhoujPas encore d'évaluation
- 1 - Paroxysmal DisordersDocument48 pages1 - Paroxysmal DisorderszhoujPas encore d'évaluation
- Chapter 18 - Cerebrovascular Disease PDFDocument14 pagesChapter 18 - Cerebrovascular Disease PDFzhoujPas encore d'évaluation
- Panayiotopoulos Syndrome A Benign Childhood Autonomic Epilepsy Frequently Imitating Encephalitis, Syncope, Migraine Sleep Disorder, or GastroenteritisDocument9 pagesPanayiotopoulos Syndrome A Benign Childhood Autonomic Epilepsy Frequently Imitating Encephalitis, Syncope, Migraine Sleep Disorder, or GastroenteritiszhoujPas encore d'évaluation
- Polyneuropathy AssessmentDocument9 pagesPolyneuropathy AssessmentzhoujPas encore d'évaluation
- A Paediatric Influenza Update 100 Years PDFDocument10 pagesA Paediatric Influenza Update 100 Years PDFzhoujPas encore d'évaluation
- 199 FullDocument11 pages199 FullzhoujPas encore d'évaluation
- 199 FullDocument11 pages199 FullzhoujPas encore d'évaluation
- Chapter 1 CT Hypertensity LesionDocument5 pagesChapter 1 CT Hypertensity LesionzhoujPas encore d'évaluation
- 00008Document15 pages00008zhoujPas encore d'évaluation
- Braddom RheumatologyDocument22 pagesBraddom RheumatologynesPas encore d'évaluation
- Pulsenotes - Chronic Kidney Disease NotesDocument12 pagesPulsenotes - Chronic Kidney Disease NotesElay PedrosoPas encore d'évaluation
- Prof BSP-Akinetic Rigid Synd - PDDocument139 pagesProf BSP-Akinetic Rigid Synd - PDSavitha BasriPas encore d'évaluation
- Kofarest LS SyrupDocument3 pagesKofarest LS SyrupGAUTAMPas encore d'évaluation
- Maternal Sepsis 2022Document13 pagesMaternal Sepsis 2022Erika RosasPas encore d'évaluation
- Age-Related Macular DegenerationDocument386 pagesAge-Related Macular Degenerationcsalvig100% (1)
- MERRF Syndrome: Rare Mitochondrial DiseaseDocument4 pagesMERRF Syndrome: Rare Mitochondrial DiseaseQuezon D. Lerog Jr.Pas encore d'évaluation
- Cramming Facts For MRCP Part 2 Exam (UK) MRCP Part 2Document7 pagesCramming Facts For MRCP Part 2 Exam (UK) MRCP Part 2siddique022Pas encore d'évaluation
- Jurnal Abses PeritonsilarDocument12 pagesJurnal Abses PeritonsilarGita KurniasariPas encore d'évaluation
- Hepatobiliary and Pancreas Surgery: Liver Disease Management & Transplant ProgramDocument4 pagesHepatobiliary and Pancreas Surgery: Liver Disease Management & Transplant ProgramDeviselvam100% (1)
- Med History & PE GuideDocument7 pagesMed History & PE GuideStephanie GaerlanPas encore d'évaluation
- Diagnosa PRBDocument12 pagesDiagnosa PRBSAEHANPas encore d'évaluation
- Approach To Diagnosis of Haemolytic AnaemiasDocument2 pagesApproach To Diagnosis of Haemolytic AnaemiasGerardLumPas encore d'évaluation
- Respiratory AssessmentDocument25 pagesRespiratory AssessmentchristyPas encore d'évaluation
- Abd Pain 2019Document66 pagesAbd Pain 2019mohammed alrubaiaanPas encore d'évaluation
- Chapter 1 - Pharmacotherapy of The Ophthalmic - 2008 - Clinical Ocular PharmacoDocument13 pagesChapter 1 - Pharmacotherapy of The Ophthalmic - 2008 - Clinical Ocular PharmacosPas encore d'évaluation
- E-Book Ophthalmology Referral Guide For GPsDocument11 pagesE-Book Ophthalmology Referral Guide For GPsnaravichandran3662Pas encore d'évaluation
- Hypomagnesemic Tetany in Ruminants: B.Gowrishankar BVN 05013Document34 pagesHypomagnesemic Tetany in Ruminants: B.Gowrishankar BVN 05013vetgowriPas encore d'évaluation
- Co AmoxyclavDocument2 pagesCo AmoxyclavHaneulPas encore d'évaluation
- مخططات ذهنية ENDOCRINE فارما - 2022 - يونس الحماديDocument17 pagesمخططات ذهنية ENDOCRINE فارما - 2022 - يونس الحماديMohned AlhababiPas encore d'évaluation
- Isglt2 Nefirits LupicaDocument2 pagesIsglt2 Nefirits LupicaAldo Amed Montaño SalinasPas encore d'évaluation
- Acute Febrile IllnessesDocument54 pagesAcute Febrile IllnessesfraolPas encore d'évaluation
- HEMASAMPLEDocument81 pagesHEMASAMPLEangela roperezPas encore d'évaluation
- Treatment of Electrolyte Disorders in Adult Patients in The Intensive Care UnitDocument21 pagesTreatment of Electrolyte Disorders in Adult Patients in The Intensive Care UnitBuat TugasPas encore d'évaluation
- Erythema Multiforme and Drug EruptionDocument5 pagesErythema Multiforme and Drug EruptionAdparizby FachryPas encore d'évaluation
- DR Lakshmi Saleem 7thPSAAP Conference - Cosmetic Surgery Clinic HyderabadDocument32 pagesDR Lakshmi Saleem 7thPSAAP Conference - Cosmetic Surgery Clinic Hyderabadcosmeticsurgeryclinic_in9534Pas encore d'évaluation
- Update Guideline For Intravenous Contrast Used of CT and MRIDocument12 pagesUpdate Guideline For Intravenous Contrast Used of CT and MRITenta Hartian HendyatamaPas encore d'évaluation
- GastroenterologyDocument30 pagesGastroenterologyMohammad MohyeddienPas encore d'évaluation
- Gastrinomas Medical or Surgical TreatmentDocument25 pagesGastrinomas Medical or Surgical TreatmentTony Miguel Saba SabaPas encore d'évaluation
- Test Bank For Medical Terminology An Accelerated Approach by Jones Full DownloadDocument67 pagesTest Bank For Medical Terminology An Accelerated Approach by Jones Full DownloadandrewlopezetosfzigkjPas encore d'évaluation