Académique Documents
Professionnel Documents
Culture Documents
O R C OH: Diethyl Ether Acetone Chloroform Methylene Chloride
Transféré par
Rhegie PocongTitre original
Copyright
Formats disponibles
Partager ce document
Partager ou intégrer le document
Avez-vous trouvé ce document utile ?
Ce contenu est-il inapproprié ?
Signaler ce documentDroits d'auteur :
Formats disponibles
O R C OH: Diethyl Ether Acetone Chloroform Methylene Chloride
Transféré par
Rhegie PocongDroits d'auteur :
Formats disponibles
LIPIDS • Parent HC + -oic acid
- "lipos" = fats or lard Ex: C18 saturated fatty acid C atoms # of double bonds
- collection of organic molecules of varying chemical compositions = octadecane + -oic acid
- grouped together based on solubility in nonpolar solvents = octadecanoic acid (stearic acid) (18:0)
- Carbon chain length Water solubility Melting point C18 monounsaturated fatty acid
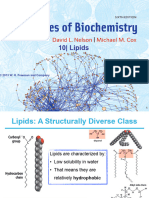
- INSOLUBLE in water = octadecene + -oic acid
- HIGHLY SOLUBLE in aprotic organic solvents such as: = octadecenoic acid (oleic acid) (18:1)
○ diethyl ether ○ acetone
○ chloroform ○ methylene chloride C18 unsaturated fatty acid (2 double bonds)
- Open Chain Forms: = octadecadienoic acid (linoleic acid) (18:2)
• Fatty acids • Triacylglycerols • Phosphoacylglycerols C18 unsaturated fatty acid (3 double bonds)
• Sphingolipids • Glycolipids • Lipid-soluble vitamins = octadecatrienoic acid (linolenic acid) (18:3)
• Prostaglandins • Leukotrienes • Thromboxanes
- Cyclic Forms: • If fatty acid is ionized (COOH part), change to -ate (COO-)
• Cholesterol • Steroid hormones • Bile acids Ex: Palmitic acid palmitate
- BIOLOGIC FUNCTIONS: Oleic acid Oleate
• Energy Source - when oxidized, each gram of fat releases 9 kcal
of energy or more than twice the energy
released by carbohydrates(=4 kcal)
• Energy Storage - in the form of triglycerides (TAG) in adipocytes
• Cell Membrane Structural Compounds
- phospholipid bilayer
- phosphoglycerides, sphingolipids and steroids make up the
basic structure of cell membranes
• Digestion - Bile
• Hormones - steroid hormones - CLASSIFICATION:
- Ex: sex hormones, adrenal hormones (corticoids) • Length of Carbon chain
• Vitamins - lipid-soluble vitamins (vitamin A,D,E and K) - long-chain fatty acids - >12 carbons
• Vitamin Absorption - Dietary fat serves as a carrier of lipid- - medium-chain fatty acids - 6-10 carbons
soluble vitamins - short-chain fatty acids - <6 carbons
• Protection - fats as shock absorber • Degree of Unsaturation
• Insulation - prevention of the release of heat - Saturated Fatty Acids
- subcutaneous fat - Genera Formula: CH3(CH2)nCOOH
- obtained from animal sources (SOLID at room temp.)
- SOLID @ RT due to its linear structure of single bonds,
allowing it to be packed tightly
- higher melting point
- NO DOUBLE BONDS - each carbon is being "saturated" w/ H
- Ex: • Capric Acid (10 carbons)
• Lauric Acid (12 carbons) • Palmitic Acid (16 carbons)
• Myristic Acid (14 carbons) • Stearic Acid (18 carbons)
- Oily or greasy nonpolar molecules stored in adipose tissue of the body
- Heterogenous compounds mainly composed of hydrocarbon chains
- Energy-rich organic molecules - Unsaturated Fatty Acids
• more energy than carbohydrates - obtained from plant sources (LIQUID at room temp.)
• long-term; used when all carbohydrates have been exhausted - LIQUID @ RT due to its bent structure of double bonds,
NOT allowing it to be packed tightly
- lower melting point
• Fatty Acids - Saturated - AT LEAST 1 CARBON-CARBON DOUBLE BOND
- Unsaturated - almost all are in CIS configuration
• Glycerides - Neutral glycerides - 2 TYPES:
- Phosphoglycerides 1. Monounsaturated Fatty Acids (MUFA)
• Complex Lipids - Lipolipids - ONLY 1 carbon-carbon double bond
• Nonglyceride - Steroids
- Waxes
- Sphingolipids - Spingomyelin
- Glycolipids
Fatty Acids 2. Polyunsaturated Fatty Acids (PUFA)
- building blocks of lipids - > 1 carbon-carbon double bonds
- naturally occurring monocarboxylic acid
- an unbranched-chain carboxylic acid 18:3 (Δ9,12,15)
- have EVEN # of carbon atoms, usually between 12-20
*2 Double bonds are separated by at least 1 hydrocarbon*
• Acetyl CoA - adds 2 C atoms as it lengthens lipid chain
- AMPHIPATHIC - has both polar and nonpolar
- hydrocarbon chain + terminal carboxyl group
Nonpolar End
O Polar/Hydrophilic End
Hydrophobic End
(Fat-soluble Tail)
R C OH (ACIDIC PROPERTY)
- NOMENCLATURE: • Location of Double Bonds
- The body CANNOT form C=C before 9th Carbon, making fatty • NO!!!
acids W/ double bonds before 9th Carbon ESSENTIAL - Trans-vaccenic acid - can LOWER risk of heart disease, diabetes
- The body CAN form C=C before 9th Carbon, making fatty & obesity
acids W/O double bonds before 9th Carbon NONESSENTIAL - Omega-3
- in Polyunsaturated Fatty Acids (PUFA): - essential fatty acids
• identified by position of double bond nearest the methyl • body can ONLY make some EPA & DHA from ALA
end (CH3) or omega end (ω) - Eicosopentanoic acid (EPA)
• Δ - shows location of double bonds - Docohexanoic acid (DHA)
- start counting from carboxyl end (COOH as C1) - Alpha-linolenic acid (ALA)
• first = 3 carbons away from methyl end Omega-3 • from flaxseed–most, canola (rapeseed), soybean, walnut,
• first = 6 carbons away from methyl end Omega-6 wheat germ
STEP 1: Find the nearest double bond near omega end - Omega-6
2: Start counting from the omega end - corn, safflower, cottonseed, sesame, sunflower
(Terminal methyl as C1) - Ex: Linoleic acid
OMEGA END (ω) - CHEMICAL REACTIONS OF FATTY ACIDS
/METHYL END • Esterification
- Fatty acids (acid) react with alcohols to form esters and water
• Acid Hydrolysis
- fatty acids are produced from esters
- reverse of esterification
- acid medium
• Saponification
- base-catalyzed hydrolysis of an ester
- alkaline medium
- PRODUCTS: Ionized Salt (SOAP) and Alcohol
- Soaps have a long uncharged hydrocarbon tail and a
negatively-charged terminus (carboxylate terminus)
- form Micelles that dissolve oil and dirt particles
- Omega-3 fatty acid
• unsaturated fatty acid with its endmost double bond 3
carbons away from its methyl end - Reaction at Double bonds: (Unsaturated Fatty Acids)
• Hydrogenation
- food industry converts polyunsaturated vegetable oils into
- Omega-6 fatty acid saturated solid fats
• unsaturated fatty acid with its endmost double bond 6 - Unsaturated fatty acids converted to saturated fatty acids
carbons away from its methyl end - increase degree of saturation as double bonds are converted to
single bonds, melting point
• Partial Hydrogenation
- NOT ALL BONDS are broken
- add H to some but NOT ALL.
- Double bonds in polyunsaturated oil
- CIS-TRANS ISOMERISM
• Cis- Fatty Acid
- Hs are on the same side of double bond
- fold into U-like formation
- naturally occurring
• Trans- Fatty Acid
- Hs are on the opposite sides of double bond
- occur in partially hydrogenated food
- BAD!!! It can lead to CVD by promoting
deposition of fat plaques to blood vessels
= ATHEROSCLEROSIS
- ARE ALL TRANS-FAT BAD?
Vous aimerez peut-être aussi
- Proposed) Declaration of Factual Innocence Under Penal Code Section 851.8 and OrderDocument4 pagesProposed) Declaration of Factual Innocence Under Penal Code Section 851.8 and OrderBobby Dearfield100% (1)
- A Beautiful Mind - Psychology AnalysisDocument15 pagesA Beautiful Mind - Psychology AnalysisFitto Priestaza91% (34)
- Pediatric ECG Survival Guide - 2nd - May 2019Document27 pagesPediatric ECG Survival Guide - 2nd - May 2019Marcos Chusin MontesdeocaPas encore d'évaluation
- Christianity and Mental Health WEB VERSIONDocument64 pagesChristianity and Mental Health WEB VERSIONWorld Religion NewsPas encore d'évaluation
- Lipid Chemistry & Biological Membranes: Functions of LipidsDocument14 pagesLipid Chemistry & Biological Membranes: Functions of LipidsALYSSA SEVEN VELASCOPas encore d'évaluation
- Lipids IIDocument45 pagesLipids IIKlieden LobrigasPas encore d'évaluation
- Lipids: Geromil J. Lara, RMT, MSMTDocument45 pagesLipids: Geromil J. Lara, RMT, MSMTSophia AmpatuanPas encore d'évaluation
- Kimia Lipida 2017 Setya - D3Document44 pagesKimia Lipida 2017 Setya - D3Anggito PrayogaPas encore d'évaluation
- BioChem LEC - M2Document3 pagesBioChem LEC - M2Gianna LingadPas encore d'évaluation
- LipidsDocument26 pagesLipidsZanie CruzPas encore d'évaluation
- Lipid Structure and Function Lecture Dorothy FinalDocument39 pagesLipid Structure and Function Lecture Dorothy FinalMarvin JeaPas encore d'évaluation
- 10 - Lipids: © 2013 W. H. Freeman and CompanyDocument71 pages10 - Lipids: © 2013 W. H. Freeman and Companymyatpwintp880Pas encore d'évaluation
- (Biochem A) Lipid Chemistry-Alcantara (Gradelifting Fairies)Document11 pages(Biochem A) Lipid Chemistry-Alcantara (Gradelifting Fairies)bero beroPas encore d'évaluation
- Xi Chapter 9 BiomoleculesDocument18 pagesXi Chapter 9 BiomoleculesStudy BoiPas encore d'évaluation
- Week 5 - LipidsDocument56 pagesWeek 5 - LipidsPaula LouisePas encore d'évaluation
- Chem123lec MidtermDocument18 pagesChem123lec Midtermlily of the valleyPas encore d'évaluation
- Lipids: Structure and ClassificationDocument83 pagesLipids: Structure and ClassificationGOT7 ł IGOT7 TRUSTPas encore d'évaluation
- Lesson 2-Fats-Water-23-1Document55 pagesLesson 2-Fats-Water-23-1Kri QPas encore d'évaluation
- Capítulo 10Document37 pagesCapítulo 10Adriana VelazquezPas encore d'évaluation
- Biochemistry LipidsDocument14 pagesBiochemistry LipidsCeline Ricci AbrahamPas encore d'évaluation
- Lipid Chemistry 1Document102 pagesLipid Chemistry 1queenPas encore d'évaluation
- L4 LipidDocument23 pagesL4 Lipidsampsonsoo17Pas encore d'évaluation
- (BIOCHEM LAB) LipidsDocument8 pages(BIOCHEM LAB) Lipidsprettyfriends 05Pas encore d'évaluation
- LipidsDocument23 pagesLipidsRukhsar JabbarPas encore d'évaluation
- 03 Lipids StudentsDocument40 pages03 Lipids Studentsmakabigail7Pas encore d'évaluation
- LipidsDocument4 pagesLipidsshane.surigaoPas encore d'évaluation
- LipidsDocument30 pagesLipidsSandy IkbarPas encore d'évaluation
- 3 LipidsDocument29 pages3 LipidsJayashree RajamaniPas encore d'évaluation
- Biochemistry I (SGS 236)Document25 pagesBiochemistry I (SGS 236)Mustafa SaßerPas encore d'évaluation
- LipidDocument118 pagesLipidAnonymous LdOWUwgEmvPas encore d'évaluation
- Lipids - L.L-N.V.Document34 pagesLipids - L.L-N.V.ViragPas encore d'évaluation
- LipidsDocument10 pagesLipidsladyPas encore d'évaluation
- Macromolecules2 1Document51 pagesMacromolecules2 1Allison DudleyPas encore d'évaluation
- 1 LipidDocument30 pages1 LipidAwab MohamedPas encore d'évaluation
- Biochemistry FinalsDocument43 pagesBiochemistry FinalsInday de DiosPas encore d'évaluation
- Unit IiDocument67 pagesUnit Iiprathap somuPas encore d'évaluation
- 1142 L9 LipidsDocument34 pages1142 L9 LipidsjanindujayathmaPas encore d'évaluation
- LipidDocument31 pagesLipidARINAL HIDAYATIPas encore d'évaluation
- New Lipid PowerpointDocument113 pagesNew Lipid PowerpointMadane Jamila Amerol SaminPas encore d'évaluation
- Lipids: Definition, Classification and FunctionsDocument27 pagesLipids: Definition, Classification and Functionssani sundayPas encore d'évaluation
- LipidsDocument31 pagesLipidsMods PadernillaPas encore d'évaluation
- Chapter 3.1 - LipidsDocument35 pagesChapter 3.1 - LipidsDazell VarronPas encore d'évaluation
- Chapter 3 Lipids PDFDocument55 pagesChapter 3 Lipids PDFVinz TombocPas encore d'évaluation
- Lipids 1Document73 pagesLipids 1France Jan First SaplacoPas encore d'évaluation
- LipidsDocument154 pagesLipidsMahnoor KhalilPas encore d'évaluation
- Chapter 11 - Lipids: of All Living Organisms Organic Compounds (Nonpolar) or (Containing Both Nonpolar and Polar Regions)Document19 pagesChapter 11 - Lipids: of All Living Organisms Organic Compounds (Nonpolar) or (Containing Both Nonpolar and Polar Regions)tiaraPas encore d'évaluation
- LipidsDocument24 pagesLipidsRandy AminolaPas encore d'évaluation
- Inbound 9025796000039556153Document41 pagesInbound 9025796000039556153ellaverino6Pas encore d'évaluation
- LipidsDocument29 pagesLipidsdavid gomezPas encore d'évaluation
- Chemistry of Lipids: DefinitionDocument45 pagesChemistry of Lipids: DefinitionLinus BraunPas encore d'évaluation
- Lipid Chemistry Overview PPP 1Document51 pagesLipid Chemistry Overview PPP 1SundarPas encore d'évaluation
- Nonpolar.: Dissolve in Non-Polar Solvents Such As Ether, Chloroform, and Acetone But Not Considerably in WaterDocument31 pagesNonpolar.: Dissolve in Non-Polar Solvents Such As Ether, Chloroform, and Acetone But Not Considerably in WaterNejib ReshadPas encore d'évaluation
- LipidsDocument3 pagesLipidsCamille GrandePas encore d'évaluation
- Chapter 4. LipidsDocument157 pagesChapter 4. Lipidsdakroub1108Pas encore d'évaluation
- Lipids - BCSC-BNDocument25 pagesLipids - BCSC-BNJudithPas encore d'évaluation
- LipidsDocument100 pagesLipidschennielafleurPas encore d'évaluation
- LipidsDocument12 pagesLipidsTheresaPas encore d'évaluation
- 161020190Dr EmanDocument31 pages161020190Dr Emanslmen1269Pas encore d'évaluation
- Unit 8 WebDocument86 pagesUnit 8 WebIris Dinah BacaramanPas encore d'évaluation
- Chemistry of Lipid-IDocument38 pagesChemistry of Lipid-Idj77djcqy7Pas encore d'évaluation
- Cells and Sugars 12-Fats and Oils-Structure-StudentDocument15 pagesCells and Sugars 12-Fats and Oils-Structure-StudenttyhbbhhPas encore d'évaluation
- Chapter 26Document18 pagesChapter 26TimmyNguyenPas encore d'évaluation
- Biochem Transes IIDocument7 pagesBiochem Transes IICienna IganoPas encore d'évaluation
- The Greatest Classics of Russian Literature in One VolumeD'EverandThe Greatest Classics of Russian Literature in One VolumePas encore d'évaluation
- Medicidefamilie 2011Document6 pagesMedicidefamilie 2011Mesaros AlexandruPas encore d'évaluation
- Contoh RPH Ts 25 Engish (Ppki)Document1 pageContoh RPH Ts 25 Engish (Ppki)muhariz78Pas encore d'évaluation
- Discrete Probability Distribution UpdatedDocument44 pagesDiscrete Probability Distribution UpdatedWaylonPas encore d'évaluation
- SHS PE MODULE 1 - RemovedDocument21 pagesSHS PE MODULE 1 - RemovedXypher NPas encore d'évaluation
- Wwe SVR 2006 07 08 09 10 11 IdsDocument10 pagesWwe SVR 2006 07 08 09 10 11 IdsAXELL ENRIQUE CLAUDIO MENDIETAPas encore d'évaluation
- 1986 Elektric M InfoDocument1 page1986 Elektric M InfoDanielDiasPas encore d'évaluation
- Motion Exhibit 4 - Declaration of Kelley Lynch - 03.16.15 FINALDocument157 pagesMotion Exhibit 4 - Declaration of Kelley Lynch - 03.16.15 FINALOdzer ChenmaPas encore d'évaluation
- Soal Bahasa Inggris Sastra Semester Genap KLS Xi 2023Document3 pagesSoal Bahasa Inggris Sastra Semester Genap KLS Xi 2023Ika Endah MadyasariPas encore d'évaluation
- Bootstrap DatepickerDocument31 pagesBootstrap DatepickerdandczdczPas encore d'évaluation
- Thomas Noochan Pokemon Review Final DraftDocument6 pagesThomas Noochan Pokemon Review Final Draftapi-608717016Pas encore d'évaluation
- Florida v. DunnDocument9 pagesFlorida v. DunnJustice2Pas encore d'évaluation
- Research Report On Energy Sector in GujaratDocument48 pagesResearch Report On Energy Sector in Gujaratratilal12Pas encore d'évaluation
- Sarcini: Caiet de PracticaDocument3 pagesSarcini: Caiet de PracticaGeorgian CristinaPas encore d'évaluation
- Electronic ClockDocument50 pagesElectronic Clockwill100% (1)
- Army Public School Recruitment 2017Document9 pagesArmy Public School Recruitment 2017Hiten BansalPas encore d'évaluation
- Athena 60 Installation Manual EN 2022.07.03Document30 pagesAthena 60 Installation Manual EN 2022.07.03joaquin.cadondonPas encore d'évaluation
- Reply To Pieta MR SinoDocument9 pagesReply To Pieta MR SinoBZ RigerPas encore d'évaluation
- GE 110HP DC Trolley MotorDocument10 pagesGE 110HP DC Trolley MotorAnthony PetersPas encore d'évaluation
- LiverDocument6 pagesLiverMiguel Cuevas DolotPas encore d'évaluation
- Rebecca Young Vs CADocument3 pagesRebecca Young Vs CAJay RibsPas encore d'évaluation
- An Etymological Dictionary of The Scottivol 2Document737 pagesAn Etymological Dictionary of The Scottivol 2vstrohmePas encore d'évaluation
- Formal Letter Format Sample To Whom It May ConcernDocument6 pagesFormal Letter Format Sample To Whom It May Concernoyutlormd100% (1)
- Grossman 1972 Health CapitalDocument33 pagesGrossman 1972 Health CapitalLeonardo SimonciniPas encore d'évaluation
- On The Linguistic Turn in Philosophy - Stenlund2002 PDFDocument40 pagesOn The Linguistic Turn in Philosophy - Stenlund2002 PDFPablo BarbosaPas encore d'évaluation
- Conrad John's ResumeDocument1 pageConrad John's ResumeTraining & OD HRODPas encore d'évaluation
- (s5.h) American Bible Society Vs City of ManilaDocument2 pages(s5.h) American Bible Society Vs City of Manilamj lopez100% (1)