Académique Documents
Professionnel Documents
Culture Documents
3'-Thiol-Modifier C6 S-S CPG
Transféré par
AlleleBiotechDescription originale:
Titre original
Copyright
Formats disponibles
Partager ce document
Partager ou intégrer le document
Avez-vous trouvé ce document utile ?
Ce contenu est-il inapproprié ?
Signaler ce documentDroits d'auteur :
Formats disponibles
3'-Thiol-Modifier C6 S-S CPG
Transféré par
AlleleBiotechDroits d'auteur :
Formats disponibles
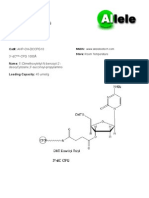
3’ Thiol, 3’-Thiol-modifier C6
S-S CPG
Box 1 | Product List
Beaucage SL. Strategies in the preparation of DNA oligonucleotide
3’ Thiol-Modifier C6 S-S CPG arrays for diagnostic applications. Curr Medicinal Chem, 8, 1213-1244.
(2001)
Cat. No. Amount
AHP-CH-TC60100 100 mg Gorodetsky AA, Barton JK. Electrochemistry using self-assembled DNA
monolayers on highly oriented pyrolytic graphite. Langmuir, 22, 7917-
AHP-CH-TC61000 1000 mg
7922. (2006)
Liu RH, Coty WA, Reed MW, Gust G. Electrochemical detection-based
DNA microarrays, IVD Tech, 31. (2008)
Name: 3’-Thiol-modifier C6 S-S CPG
Chemical Name: 1-O-Dimethoxytrityl-hexyl-disulfide,1’-succi- Umek RM, Lin SW, Vielmetter J, Terbrueggen RH, Yu CJ, Kayyem JF,
noyl-long chain alkylamino-CPG Irvine B, Yowanto H, Blackburn GF, Farkas DH, Chen YP. Electronic
Formula: N/A detection of nucleic acids: a versatile platform for molecular diagnostics.
J Mol Diagn, 3, 74-84. (2001)
Use: 3’-Thiol-modifier C6 S-S CPG is used to label a thiol group Lockett, MR, Phillips, MF, Jarecki, JL, Peelen, D, Smith LM. A tetrafluo-
at the 3’ end of oligonucleotides. rophenol activated ester self-assembled monolayer for the immobiliza-
Purity: 95%+ tion of amine-modified oligonucleotides. Langmuir, 24 (1), 69-75. (2008)
Diluent: Anhydrous Acetonitrile
Stability in Solution: N/A Wei F, Wang J, Liao W, Zimmermann BG, Wong DT, Ho CM. Electro-
chemical detection of low-copy number salivary RNA based on specific
Background: signal amplification with a hairpin probe. Nucleic Acids Res, 36(11), e65.
(2008)
Thiol modifications are important in the production of diagnos-
tic probes. They are use in DNA synthesizers to functionalize Frutos AG, Brockman JM, Corn RM. Reversible protection and reactive
oligonucleotides. The terminal thiol group allows attachment of patterning of amine and hydroxyl-terminated self-assembled monolay-
an oligo to various ligands, such as biotin, alkaline phosphatase ers on gold surfaces for the fabrication of biopolymer arrays. Langmuir,
or fluorescent dyes. Via an iodoacetamide or maleimide link- 16, 2192-2197. (2000)
age, it allows for the attachment of an oligo to a solid surface.
Thiol modification can be placed at either the 5’ or 3’ end of the Liepold P, Kratzmüller T, Persike N, Bandilla M, Hinz M, Wieder H,
oligonucleotide. For introduction of a thiol group at the 3’-end, Hillebrandt H, Ferrer E, Hartwich G. Electrically detected displacement
thiol-modifier C6 S-S CPG is available. To introduce a thiol assay (EDDA): a practical approach to nucleic acid testing in clinical or
group at the 5’ end, either 5’-thiol-modifier-C6 or 5’-thiol modi- medical diagnosis. Anal Bioanal Chem, 391, 1759-72. (2009)
fier C6 S-S may be used.
Li Z, Jin R, Mirkin CA, Letsinger RL. Multiple thiol-anchor capped DNA-
Coupling: All oxidation steps should use 0.02M Iodine solution gold nanoparticle conjugates. Nucleic Acids Res., 30, 1558-62. (2002)
to avoid oxidative cleavage of the disulfide linkage.
Demers LM, Mirkin CA, Mucic RC, Reynolds RA III, Letsinger RL.
Deprotection: After proceeding with normal deprotection, the Fluorescence-based method for determining the surface coverage and
disulfide can be cleaved at room temperature in 30 minutes with hybridization efficiency of thiol-capped oligonucleotides bound to gold
a buffered 100 mM DTT pH 8.3 - 8.5 solution. It is imporant to thin films and nanoparticles. Anal Chem, 72, 5535–5541. (2000)
completely remove the reducing agent before using the oligo in
a coupling reaction. Letsinger RL, Elghanian R, Viswanadham G, Mirkin CA. Use of a ster-
oid cyclic disulfide anchor in constructing gold nanoparticle-oligonucleo-
Storage: Freezer storage, -10 to -30°C, dry, dark area tide conjugates,” Bioconjugate Chem, 11(2), 289–291. (2000)
Labeled Oligo Storage: Store labeled oligo in the dark, either
dry or in a neutral aqueous media at -20°C. Do not store crude
fluorescently labeled oligonucleotides using ammonia.
MSDS: www.allelebiotech.com
Reference:
Phillips MF, Lockett MR, Rodesch MJ, Shortreed MR, Cerrina F, Smith
LM. In situ oligonucleotide synthesis on carbon materials: stable sub-
strates for microarray fabrication. Nucleic Acids Res, 36, e7. (2008)
Vous aimerez peut-être aussi
- Gel Electrophoresis of ProteinsD'EverandGel Electrophoresis of ProteinsMichael J DunnPas encore d'évaluation
- Articulo Quimica Medicinal 3Document16 pagesArticulo Quimica Medicinal 3Jose Antonio Espinosa TorresPas encore d'évaluation
- J. Biol. Chem.-1988-Hodges-11768-75Document8 pagesJ. Biol. Chem.-1988-Hodges-11768-75Inggrid MadaniPas encore d'évaluation
- MBP 2 DimageDocument7 pagesMBP 2 DimageDolphingPas encore d'évaluation
- NIH Public Access: Author ManuscriptDocument27 pagesNIH Public Access: Author ManuscriptChristian AlvisPas encore d'évaluation
- Biopolym - Cell 2018 34 5 374 enDocument13 pagesBiopolym - Cell 2018 34 5 374 enАнна ШаповаловаPas encore d'évaluation
- 7265Document8 pages7265DiogomussumPas encore d'évaluation
- 1985 Biochem Molecular Structure of The b3 Adrenergic ReceptorDocument7 pages1985 Biochem Molecular Structure of The b3 Adrenergic Receptorjames mellaleievPas encore d'évaluation
- FA and TAGS NamesDocument19 pagesFA and TAGS NamesIryna BonPas encore d'évaluation
- Inhibition of Peptidoglycan Biosynthesis in Gram-Positive Bacteria by LY146032Document7 pagesInhibition of Peptidoglycan Biosynthesis in Gram-Positive Bacteria by LY146032SachithPas encore d'évaluation
- Novel Phthalocyanines Containing SubstitDocument10 pagesNovel Phthalocyanines Containing SubstitPopusoi AnaPas encore d'évaluation
- Oxygen Free Radical Modified Dna: Implications in The Etiopathogenesis of Systemic Lupus ErythematosusDocument8 pagesOxygen Free Radical Modified Dna: Implications in The Etiopathogenesis of Systemic Lupus ErythematosusfilianiePas encore d'évaluation
- Giaouris Et Al POSTER CBL2007 (Rennes 13-15 Nov 2007)Document1 pageGiaouris Et Al POSTER CBL2007 (Rennes 13-15 Nov 2007)Stathis GiaourisPas encore d'évaluation
- Deshmukh 2000Document24 pagesDeshmukh 2000Tecno QB7Pas encore d'évaluation
- An 21093 LC DNAPac PA200 RS Oligonucleotides AN21093 enDocument7 pagesAn 21093 LC DNAPac PA200 RS Oligonucleotides AN21093 enRajat DasPas encore d'évaluation
- IJPPS NBAC November2010Document4 pagesIJPPS NBAC November2010Anantha LakshmiPas encore d'évaluation
- 2016 HarmalolDocument11 pages2016 Harmaloltaoufik akabliPas encore d'évaluation
- Hammud2008 PDFDocument13 pagesHammud2008 PDFgringoPas encore d'évaluation
- G.S. Suresh Kumar, A. Antony Muthu Prabhu, P.G. Seethalaksmi, N. Bhuvanesh, S. KumaresanDocument8 pagesG.S. Suresh Kumar, A. Antony Muthu Prabhu, P.G. Seethalaksmi, N. Bhuvanesh, S. KumaresanAfdhonil Qomarul PPas encore d'évaluation
- GKL 430Document8 pagesGKL 430Anindya RoyPas encore d'évaluation
- T 7024 PisDocument2 pagesT 7024 PisMadhuri PatelPas encore d'évaluation
- POLGDocument11 pagesPOLGİzem DevecioğluPas encore d'évaluation
- Dionysios A. Papaioannou Professor of Organic Chemistry Department of Chemistry University of PatrasDocument90 pagesDionysios A. Papaioannou Professor of Organic Chemistry Department of Chemistry University of PatrasDiomedes DPas encore d'évaluation
- List of References: Fractogel EMD Tentacle-PhasesDocument54 pagesList of References: Fractogel EMD Tentacle-PhasesTuyền KimPas encore d'évaluation
- Electrochemical Transduction of DNA Hybridization at Modified Electrodes by Using An Electroactive Pyridoacridone IntercalatorDocument10 pagesElectrochemical Transduction of DNA Hybridization at Modified Electrodes by Using An Electroactive Pyridoacridone IntercalatorferPas encore d'évaluation
- Application of Sodium Aluminate As A Heterogeneous Base Catalyst For Biodiesel Production From Soybean OilDocument4 pagesApplication of Sodium Aluminate As A Heterogeneous Base Catalyst For Biodiesel Production From Soybean Oillakhya07Pas encore d'évaluation
- بحث رقم 50Document23 pagesبحث رقم 50ahmed eldesokyPas encore d'évaluation
- V. Sanz-Nebot Et Al. J. Chromatogr. B 798 2003Document7 pagesV. Sanz-Nebot Et Al. J. Chromatogr. B 798 2003bookmoonPas encore d'évaluation
- Iridium-Catalyzed Asymmetric Ring Opening of Azabicyclic Alkenes by AminesDocument9 pagesIridium-Catalyzed Asymmetric Ring Opening of Azabicyclic Alkenes by AminesDiogomussumPas encore d'évaluation
- Heterogeneous Asymmetric Diels-Alder Reactions Using A Copper-Chiral Bis (Oxazoline) Complex Immobilized On Mesoporous SilicaDocument5 pagesHeterogeneous Asymmetric Diels-Alder Reactions Using A Copper-Chiral Bis (Oxazoline) Complex Immobilized On Mesoporous SilicaJC Jane BarnesPas encore d'évaluation
- 1 s2.0 S0162013420301938 MainDocument13 pages1 s2.0 S0162013420301938 Mainstanleytiu63Pas encore d'évaluation
- Assessment of Heavy Metal Bioavailability Using Escherichia Coli Zntap::Lux and Copap::Lux-Based BiosensorsDocument5 pagesAssessment of Heavy Metal Bioavailability Using Escherichia Coli Zntap::Lux and Copap::Lux-Based BiosensorsShahida ZimiPas encore d'évaluation
- 2a-Basic of Genetic Engineering PDFDocument42 pages2a-Basic of Genetic Engineering PDFWomen 68Pas encore d'évaluation
- Synthesis of Novel Quaternary Chitosan Derivatives ViaDocument4 pagesSynthesis of Novel Quaternary Chitosan Derivatives ViaAxel MéndezPas encore d'évaluation
- 2005 OL OlefinesDocument3 pages2005 OL OlefinesCarmen SimalPas encore d'évaluation
- 2005 Synthesis of DinitrochalconesDocument4 pages2005 Synthesis of DinitrochalconesJESUS DAVID BOLA‹O JIMENEZPas encore d'évaluation
- 12 Ane Metal Ion ComplexesDocument9 pages12 Ane Metal Ion ComplexesSabrina MilanoPas encore d'évaluation
- Ahmed 2009Document7 pagesAhmed 2009shaksganeshanPas encore d'évaluation
- J. Agric. Food Chem. 2012, 60, 7204 7210Document7 pagesJ. Agric. Food Chem. 2012, 60, 7204 7210Duong Pham QuangPas encore d'évaluation
- 17Document9 pages17'Licenza AdagioPas encore d'évaluation
- C7ef PDFDocument3 pagesC7ef PDFVishak VsPas encore d'évaluation
- Tween Influence DNA HybridizationDocument7 pagesTween Influence DNA Hybridizationsimone.bassini1996Pas encore d'évaluation
- Fluoresscent DNA NanotagsDocument12 pagesFluoresscent DNA NanotagsreoloxPas encore d'évaluation
- DAN Bertozzi PDFDocument40 pagesDAN Bertozzi PDFMark PainterPas encore d'évaluation
- Lu 2017Document6 pagesLu 2017MikePas encore d'évaluation
- 2009 JOC AziridinesDocument8 pages2009 JOC AziridinesCarmen SimalPas encore d'évaluation
- Dye-Ligand Affinity SystemsDocument26 pagesDye-Ligand Affinity SystemsnekojcoPas encore d'évaluation
- (99mTc) HYNIC-RGD for Imaging Integrin Αvβ3 ExpressionDocument8 pages(99mTc) HYNIC-RGD for Imaging Integrin Αvβ3 ExpressionacbgdvPas encore d'évaluation
- Melting Behavior of Pseudoknots Containing Adjacent GC and AT Rich DomainsDocument7 pagesMelting Behavior of Pseudoknots Containing Adjacent GC and AT Rich DomainsMiftahul KautsariPas encore d'évaluation
- Ic401607underscoring The Influence of Inorganic Chemistry On Nuclear Imaging With RadiometalszDocument20 pagesIc401607underscoring The Influence of Inorganic Chemistry On Nuclear Imaging With RadiometalszkawtherahmedPas encore d'évaluation
- Electronic Detection of Nucleic Acids: A Versatile Platform For Molecular DiagnosticsDocument16 pagesElectronic Detection of Nucleic Acids: A Versatile Platform For Molecular DiagnosticsLinggonilus MasturandaPas encore d'évaluation
- Organotin Compound Derived From 3 Hydroxy 2 Formylpyridine Semicarbazone Synthesis Crystal Structure and Antiproliferative Activity PDFDocument8 pagesOrganotin Compound Derived From 3 Hydroxy 2 Formylpyridine Semicarbazone Synthesis Crystal Structure and Antiproliferative Activity PDFIT InventoryPas encore d'évaluation
- DNA SequencingDocument23 pagesDNA Sequencingrr48843100% (1)
- A Stereoselective Approach To Nucleosides andDocument4 pagesA Stereoselective Approach To Nucleosides andcarloscampanherPas encore d'évaluation
- MTT - Sigma AldrichDocument2 pagesMTT - Sigma AldrichFellicia RachmadianaPas encore d'évaluation
- TBC InternasionalDocument9 pagesTBC InternasionalDebbyZulfitrie CahyuniPas encore d'évaluation
- Antimetabolites in Biochemistry, Biology and Medicine: Proceedings of a Symposium on Antimetabolites in Biochemistry, Biology and Medicine, Held in Prague, Czechoslovakia, July 10-12, 1978D'EverandAntimetabolites in Biochemistry, Biology and Medicine: Proceedings of a Symposium on Antimetabolites in Biochemistry, Biology and Medicine, Held in Prague, Czechoslovakia, July 10-12, 1978J. SkodaÉvaluation : 5 sur 5 étoiles5/5 (1)
- 2018 ArriortuaDocument10 pages2018 Arriortuajesus ibarraPas encore d'évaluation
- KLOMPOKDocument8 pagesKLOMPOKRikardoPas encore d'évaluation
- Zero-Valent Metals Accelerate The Neopentylglycolborylation of Aryl Halides Catalyzed by Nicl - Based Mixed-Ligand SystemsDocument7 pagesZero-Valent Metals Accelerate The Neopentylglycolborylation of Aryl Halides Catalyzed by Nicl - Based Mixed-Ligand SystemsDiogomussumPas encore d'évaluation
- Modified UTPDocument1 pageModified UTPAlleleBiotechPas encore d'évaluation
- High Quality, Standard RGTP Suitable For Large Scale IVT.: DescriptionDocument1 pageHigh Quality, Standard RGTP Suitable For Large Scale IVT.: DescriptionAlleleBiotechPas encore d'évaluation
- M13KO7 Helper PhageDocument1 pageM13KO7 Helper PhageAlleleBiotechPas encore d'évaluation
- Modified CTPDocument1 pageModified CTPAlleleBiotechPas encore d'évaluation
- High Quality, Standard rATP Suitable For Large Scale IVT.: DescriptionDocument1 pageHigh Quality, Standard rATP Suitable For Large Scale IVT.: DescriptionAlleleBiotechPas encore d'évaluation
- Anti-RFP (5F8) Monoclonal AntibodyDocument1 pageAnti-RFP (5F8) Monoclonal AntibodyAlleleBiotechPas encore d'évaluation
- PNCS dLanYFPDocument1 pagePNCS dLanYFPAlleleBiotechPas encore d'évaluation
- mClavGR2 Fusion VectorsDocument3 pagesmClavGR2 Fusion VectorsAlleleBiotechPas encore d'évaluation
- Anti-RFP (3F5) Monoclonal AntibodyDocument1 pageAnti-RFP (3F5) Monoclonal AntibodyAlleleBiotechPas encore d'évaluation
- Custom Sub Cloning ServiceDocument1 pageCustom Sub Cloning ServiceAlleleBiotechPas encore d'évaluation
- Mwasabi PCR Template For IVTDocument1 pageMwasabi PCR Template For IVTAlleleBiotechPas encore d'évaluation
- Luciferase PCR Template For IVTDocument1 pageLuciferase PCR Template For IVTAlleleBiotechPas encore d'évaluation
- Allele AgaroseDocument1 pageAllele AgaroseAlleleBiotechPas encore d'évaluation
- pmTFP1 ClathrinDocument4 pagespmTFP1 ClathrinAlleleBiotechPas encore d'évaluation
- ChromoTek RFP BoosterDocument1 pageChromoTek RFP BoosterAlleleBiotechPas encore d'évaluation
- 3' DT-CPGDocument1 page3' DT-CPGAlleleBiotechPas encore d'évaluation
- 3' DG-CPGDocument1 page3' DG-CPGAlleleBiotechPas encore d'évaluation
- 3' DC-CPGDocument1 page3' DC-CPGAlleleBiotechPas encore d'évaluation
- 3'-Amino-Modifier C7 CPGDocument1 page3'-Amino-Modifier C7 CPGAlleleBiotechPas encore d'évaluation
- Gryphon Selection MediumDocument1 pageGryphon Selection MediumAlleleBiotechPas encore d'évaluation
- PyridineDocument16 pagesPyridineMuhammad AliAliPas encore d'évaluation
- Extraction and Purification of Plasmid Dna PDFDocument2 pagesExtraction and Purification of Plasmid Dna PDFCourtney0% (2)
- Riasorb Uv-1577 TDSDocument1 pageRiasorb Uv-1577 TDSparijat patelPas encore d'évaluation
- Ether and AldehydeDocument112 pagesEther and Aldehydejhapindra adhikariPas encore d'évaluation
- v3.3 - Cut-Off - Activated Carbon Production Granular From Hard Coal - Rer - Activated Carbon Granular 1Document14 pagesv3.3 - Cut-Off - Activated Carbon Production Granular From Hard Coal - Rer - Activated Carbon Granular 1jimPas encore d'évaluation
- Q2Week-2 PhotosynthesisDocument36 pagesQ2Week-2 Photosynthesisjustin charles jerimy raymundoPas encore d'évaluation
- Chapter 14 CDocument8 pagesChapter 14 CAnonymous T02GVGzBPas encore d'évaluation
- A Survey Report On PreservativesDocument46 pagesA Survey Report On Preservatives0921pyPas encore d'évaluation
- Chem Academy: Daily Assignment Iit-Jam Home Work DPPDocument4 pagesChem Academy: Daily Assignment Iit-Jam Home Work DPPVkumar100% (1)
- D 03 Man Aspirin Syn AnalysisDocument12 pagesD 03 Man Aspirin Syn AnalysisWahyu RedfieldPas encore d'évaluation
- Project Report On: "Variation in Intrinsic Viscosity by Ostwald Method - A StudyDocument44 pagesProject Report On: "Variation in Intrinsic Viscosity by Ostwald Method - A Study03 AFTAB MANSURIPas encore d'évaluation
- 3.lactic Acid Fermentation of Potato Pulp by The FungusDocument4 pages3.lactic Acid Fermentation of Potato Pulp by The FungusGizemPas encore d'évaluation
- HTC Summary and Discussion of Chemical Mechanisms For Process EngineeringDocument18 pagesHTC Summary and Discussion of Chemical Mechanisms For Process EngineeringJoseCuevasMartinezPas encore d'évaluation
- Biochemistry Practical WorkbookDocument130 pagesBiochemistry Practical WorkbookNaomiKerryaThompsonPas encore d'évaluation
- Lupin Seminar 1Document40 pagesLupin Seminar 1Abraha Abadi100% (1)
- Biochemistry Review Booklet 1Document28 pagesBiochemistry Review Booklet 1vaegmundigPas encore d'évaluation
- pET-38b (+) Vector: Developed Through Collaboration Between Novagen and CBD Technologies, IncDocument2 pagespET-38b (+) Vector: Developed Through Collaboration Between Novagen and CBD Technologies, Incjacobo urbinaPas encore d'évaluation
- ADEKA Surfactants-productsList 1111 PDFDocument16 pagesADEKA Surfactants-productsList 1111 PDFichsanPas encore d'évaluation
- Molecular Biology Fourth Edition Chapt01 LectureDocument18 pagesMolecular Biology Fourth Edition Chapt01 LecturePandit JiPas encore d'évaluation
- Nonessential Aminoacid SynthesisDocument46 pagesNonessential Aminoacid SynthesisJenny JosePas encore d'évaluation
- Post UTME Past QuestionsDocument162 pagesPost UTME Past Questionslee min ho fantasyPas encore d'évaluation
- Preparation of Urea Formaldehyde ResinDocument3 pagesPreparation of Urea Formaldehyde ResinMUHAMMAD AKRAM100% (1)
- D4547 1208627-1Document15 pagesD4547 1208627-1Jailson SilvaPas encore d'évaluation
- Dye Bath LubricantsDocument23 pagesDye Bath Lubricantshuerta_antonio1181100% (1)
- PC 20166Document10 pagesPC 20166chopin.wlive.cnPas encore d'évaluation
- Cosmetics Products Development Hair TreatmentsDocument8 pagesCosmetics Products Development Hair TreatmentsmasorPas encore d'évaluation
- 07-073 Colorants FinalDocument5 pages07-073 Colorants Finalex_infinity0% (1)
- J.M. Cowie, Polymers SynthesisDocument25 pagesJ.M. Cowie, Polymers SynthesisKoNi ChiWaPas encore d'évaluation
- Alkanes and HalogenoalkanesDocument67 pagesAlkanes and HalogenoalkanesRAFIDPas encore d'évaluation
- The United States Pharmacopeial (USP 37) Monographs, Vitamina D PDFDocument2 pagesThe United States Pharmacopeial (USP 37) Monographs, Vitamina D PDFMeli AppelhansPas encore d'évaluation