Académique Documents
Professionnel Documents
Culture Documents
SG2 N79 R11
Transféré par
David OngDescription originale:
Titre original
Copyright
Formats disponibles
Partager ce document
Partager ou intégrer le document
Avez-vous trouvé ce document utile ?
Ce contenu est-il inapproprié ?
Signaler ce documentDroits d'auteur :
Formats disponibles
SG2 N79 R11
Transféré par
David OngDroits d'auteur :
Formats disponibles
GHTF/SG2/N79R11:2009
FINAL DOCUMENT
Title: Medical Devices: Post Market Surveillance: National Competent Authority Report Exchange Criteria and Report Form Authoring Group: Study Group 2 Endorsed by: The Global Harmonization Task Force Date: February 17, 2009
Roland Rotter, GHTF Chair
The document herein was produced by the Global Harmonization Task Force, which is comprised of representatives from medical device regulatory agencies and the regulated industry. The document is intended to provide non-binding guidance for use in the regulation of medical devices, and has been subject to consultation throughout its development. There are no restrictions on the reproduction, distribution or use of this document; however, incorporation of this document, in part or in whole, into any other document, or its translation into languages other than English, does not convey or represent an endorsement of any kind by the Global Harmonization Task Force. Copyright 2000 by the Global Harmonization Task Force
Medical Devices: Post Market Surveillance: National Competent Authority Report Exchange Criteria and Report Form: GHTF/SG2/N79R11:2009 Study Group 2 Final Document
___________________________________________________________________________
Table of Contents
1. 2. 3. 4. 5. 6. 7. Scope .................................................................................................................................4 References .........................................................................................................................4 Definitions .........................................................................................................................4 Reporting Guidance ...........................................................................................................6 National Competent Authority Report (NCAR ..................................................................7 Instructions for Filling in National Competent Authority Report .....................................9 Report Exchange Method ...............................................................................................12
__________________________________________________________________________________________ February 2009 2 / 13
Medical Devices: Post Market Surveillance: National Competent Authority Report Exchange Criteria and Report Form: GHTF/SG2/N79R11:2009 Study Group 2 Final Document
___________________________________________________________________________
Preface
The document herein was produced by the Global Harmonization Task Force, a voluntary group of representatives from medical device regulatory agencies and the regulated industry. The document is intended to provide non-binding guidance for use in the regulation of medical devices, and has been subject to consultation throughout its development. There are no restrictions on the reproduction, distribution or use of this document; however, incorporation of this document, in part or in whole, into any other document, or its translation into languages other than English, does not convey or represent an endorsement of any kind by the Global Harmonization Task Force.
__________________________________________________________________________________________ February 2009 3 / 13
Medical Devices: Post Market Surveillance: National Competent Authority Report Exchange Criteria and Report Form: GHTF/SG2/N79R11:2009 Study Group 2 Final Document
___________________________________________________________________________
Introduction
This document was developed by Study Group 2 of the GHTF, to provide guidance, procedures and forms for the exchange of reports concerning the safety of medical devices between National Competent Authorities (NCA) and other participants of the GHTF National Competent Authority Report (NCAR) exchange program.
1. Scope
This document provides guidance on: the criteria to be used for deciding when to exchange information with other national competent authorities and other NCAR participants the procedures to follow when exchanging information the forms to use for exchanging the information
Countries participating in the exchange of NCARs are encouraged to use this guidance. Requirements for participating in the NCAR exchange program are contained in a supplementary document SG2 N38. SG2 document N8 provides general guidance on the public release of information.
2. References
The latest revisions of GHTF SG2 N8, Guidance on How to Handle Information Concerning Vigilance Reporting Related to Medical Devices GHTF SG2 N38, Application Requirements for Participation in the GHTF National Competent Authority Report Exchange Program
3. Definitions
NCAR Secretariat The organisation which receives NCARs from reporting NCAs and distributes them to other NCAR participants in accordance with this guidance and with GHTF SG2 N38 is known as the Secretariat. Active exchange Active Exchange is a pro-active exchange of information involving direct notification to nominated contact addresses. This is achieved via e-mail and through the NCAR Secretariat. Active exchange is the method of choice for high risk issues. Passive exchange: Passive Exchange is the exchange of information via the use of a database, website or other means for exchange participants to view at their discretion
__________________________________________________________________________________________ February 2009 4 / 13
Medical Devices: Post Market Surveillance: National Competent Authority Report Exchange Criteria and Report Form: GHTF/SG2/N79R11:2009 Study Group 2 Final Document
___________________________________________________________________________ Field Safety Corrective Action - FSCA A field safety corrective action (SG2 N57) is an action taken by a manufacturer to reduce a risk of death or serious deterioration in the state of health associated with the use of a medical device. Such actions should be notified via a field safety notice (see SG2 N57). In assessing the need of the FSCA the manufacturer may use the methodology described in the harmonised standard EN ISO 14971. This may include: - return of a medical device to the manufacturer or its representative; - device modification1; - device exchange; - device destruction; - advice given by manufacturer regarding the use of the device (e.g. where the device is no longer on the market or has been withdrawn but could still possibly be in use e.g. implants) Device modifications may include: - retrofit in accordance with the manufacturer's modification or design change; - permanent or temporary changes to the labeling or instructions for use; - software upgrades including those carried out by remote access; - modification to the clinical management of patients to address a risk of serious injury or death related specifically to the characteristics of the device. For example: - For implantable devices it is often clinically unjustifiable to explant the device. Corrective action taking the form of special patient follow-up, irrespective of whether any affected un-implanted devices remain available for return. - For any diagnostic device (e.g. IVD, imaging equipment or devices) the retesting of affected patients, samples or the review of previous results. - advice on a change in the way the device is used (e.g. IVD manufacturer advises revised quality control procedure -use of third party controls or more frequent calibration). Confidential Information: Information that due to its nature may be prejudicial to one or more persons, or that may be deemed as such by regional confidentiality acts and regulations, and that, for this reason, has been marked by the information provider as being confidential or not for general release. Public Information For the purposes of this document, public information is regarded to be non-confidential. This information may not necessarily be widely or easily available. For example, information contained in recall notifications, safety alerts, hazard alerts, product notifications and other product advisories is considered to be public information.
______________________________ 1 Note: device modifications may need to be approved by the NCA or third parties.
__________________________________________________________________________________________ February 2009 5 / 13
Medical Devices: Post Market Surveillance: National Competent Authority Report Exchange Criteria and Report Form: GHTF/SG2/N79R11:2009 Study Group 2 Final Document
_________________________________________________________________________________ Serious Public Health Threat or Concern Any event type, which results in imminent risk of death, serious injury, or serious illness that may require prompt remedial action. Safeguard Action This describes the action taken by an EU Member State to withdraw, prohibit or otherwise restrict a device from the market or from being put into service, in accordance with EU Community Law on medical devices. (e.g. Article 8 of the Medical Device Directive 93/42/EEC).
4. Reporting Guidance
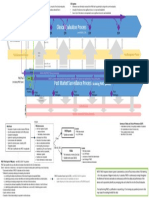
E-mail is the active exchange method currently used by NCAs to exchange information on high-risk issues. Figure 1 (Appendix A) describes the information exchange process and the appropriate route for information exchange. The criteria given below should be considered to determine whether an NCAR should be sent. The greater the number of criteria fulfilled, the greater the justification for exchanging the NCAR.
Seriousness Unexpectedness of the incident/event Population Vulnerable (pediatric/elderly) Preventability (can useful recommendations be made?) Public Concern / Outrage (ex: lead aprons containing radioactive material) Benefit/Risk - State of the art? Alternatives? Lack of Scientific Data (especially long term effects) Repeated device problems that re-surface (ex: heating pads, O.R. fires) Written notifications by the NCA to the public (hospitals, physicians, etc.) Will active exchange help protect public health or have the manufacturers actions been sufficient?
An NCAR should only be sent if the issue is considered to be serious. For example, an NCAR relating to an unexpected but non-serious event is unlikely to be exchanged. Each NCAR participant has the right to choose NOT to exchange information actively through the NCAR Secretariat if it believes the data has already been made public or if the exchange criteria are not met. Each NCAR participant should consider the extent of the global distribution of affected devices, for their proposed NCAR. If distribution is regional, rather than global (eg. Europe alone, rather than Europe and one of the other regions), then consideration should be given to NOT sending an NCAR. If distribution of affected devices includes 2 or more regions (of Europe, North America, Australia, Japan, or any additional NCAR partners), then consideration (in addition to the criteria) should be given to sending the NCAR.
__________________________________________________________________________________________ February 2009 6 / 13
Medical Devices: Post Market Surveillance: National Competent Authority Report Exchange Criteria and Report Form: GHTF/SG2/N79R11:2009 Study Group 2 Final Document
___________________________________________________________________________ a) If the investigation is complete: If the investigation is complete, and a decision has been made by the NCA or manufacturer that action is required, and the issue fulfils the reporting criteria, then a report should be sent in a normal manner. Such reports should normally be considered Public Information. NCAs should involve the manufacturer in the investigation of incidents and resolution of issues or actions and consult with the manufacturer before sending out notices to other NCAR participants. In the case that part of the information is considered to be confidential, then this should be clearly identified in the report. b) If the investigation is not complete: If the investigation is not complete but a decision has been made to take action or action is likely, the public health threat or concern must be assessed and if high, a report should be sent. Such reports are normally considered to be Confidential Information and may contain requests for feedback from the receiving NCAs. If such reports are exchanged, questions to the manufacturer from other NCAs should be directed to the NCA who is the originator of the report whenever possible. c) If no action required. If the investigation is complete and no action is required, then the report should not be exchanged actively. Passive exchange should then be considered, but it should only be used where a link between the device and adverse event concerned has been established.
5. National Competent Authority Report (NCAR)
The National Competent Authority Report (NCAR) form, below, should be used for active exchange. Guidance on its completion is contained in Section 7.
__________________________________________________________________________________________ February 2009 7 / 13
Medical Devices: Post Market Surveillance: National Competent Authority Report Exchange Criteria and Report Form: GHTF/SG2/N79R11:2009 Study Group 2 Final Document
________________________________________________________________________________
NATIONAL COMPETENT AUTHORITY REPORT This form should be used for the exchange of medical device information between NCAR participants only. Completed forms should not be released to the public. 1. Is this report confidential? Yes [ ] No [ ] Reference and Reporter Data 2. NCA report ref. no.: 3. Local NCA reference no.: 4. Related NCA report nos.: (if any) 5. Manufacturer Ref/Recall no.: 8. Tel: Device Data 11. Generic name/kind of device: 12. Nomenclature id: 14. Trade Name and Model: 15. Software version: 16. Serial no.: 18. Manufacturer: Country: Full Address: Contact: Tel: Fax: E-mail: 6. Sent by: (Name and Organization) 9. Fax: 7. Contact person: (if different from 6) 10. E-mail:
13. No.:
17. Lot/batch no.: 19. Authorized rep: Country: Full Address: Contact: Tel: Fax: E-mail:
Event Data 23a. Background information and reason for this report: 23b. Is the investigation of the report complete? [ ]Yes [ ] No 24a. Conclusions: 24b. Have the manufacturers actions been made public? [ ]Yes [ ]No 24c. The originator of this NCAR will take the lead and co-ordinate the investigation [ ]Yes [ ]No 25a. Recommendation to receivers of this report: 25b. Device known to be in the market in (include copy of manufacturers letter): 25c. Device also marketed as (trade name): Report Distribution 26a. This report is being distributed to: [ ] The NCAR Secretariat for further distribution to FULL NCAR PARTICIPANTS. [ ] The NCAR Secretariat for further distribution to ALL NCAR PARTICIPANTS. [ ] EEA states, EC, and EFTA [ ] The following targeted NCAs: [ ] The manufacturer / authorized rep.: 26b. The last NCAR distributed by this NCA was (>>>>>>>>>>>>>)
__________________________________________________________________________________________ February 2009 8 / 13
Medical Devices: Post Market Surveillance: National Competent Authority Report Exchange Criteria and Report Form : GHTF/SG2/N79R11:2009 Study Group 2 Final Document
___________________________________________________________________________
6. Instructions for Filling in National Competent Authority Report
The form should be completed in English The NCAR participant filling in and sending the NCAR is responsible for the quality of the content as well as the appropriateness of sending such a message and the scope of its distribution. Guidance on which issues should be selected for exchange between NCAR participants is given in Section 5 above. Before releasing any information, careful note should be taken of the SG2 N8 (Guidance on How to Handle Information Concerning Vigilance Reporting Related to Medical Devices). This form should be completed by NCAR participants only, when exchanging safety information about relevant measures and/or recommendations relating to the prevention of adverse incidents concerning medical devices. This form is designed for exchanging information between NCAR participants; it should not be passed directly on to patients, users, third persons or the public instead, if there is a need to communicate to this audience another form of notice should be used. It is not to be used for advising of single incidents, unless those incidents have a clear implication for public health. In such cases, the implied recommendation is for other NCAs to be aware and take such local actions they find appropriate. If the NCAR report concerns a specific manufacturers device, then the manufacturer or authorized representative should be consulted about the NCARs content and distribution prior to it being sent preferably by providing a copy for the manufacturer or authorized representative to comment on. This will help to ensure the accuracy of the NCAR. An appropriate time frame for receiving manufacturers comments should be communicated. However, this process should not be allowed to cause unnecessary delay. If an NCAR concerns a range of devices from different manufacturers then the NCA should make efforts to contact and obtain comment from all relevant manufacturers or authorized representatives known to be on their markets. There are differing reporting obligations for various NCAR participants. In general, NCAR participants shall send reports directly to the NCAR Secretariat for appropriate global distribution. The NCAR secretariat will include the originator of the NCA report as confirmation of distribution. The EEA States must exchange reports with each other in accordance with current European Directives for medical device. They should also send the report to the NCAR Secretariat for further distribution to all other (non EEA) NCAR participants. There are instances where reports are sent only to EEA participants of the NCAR program. This may cause a discontinuity in the numbering of reports received from EEA participants. When an NCAR is not to be distributed to all NCAR participants a note of this should be made on the next NCAR that is issued by the originating NCA to all NCAR participants (see Notes on Field 26b).
__________________________________________________________________________________________ February 2009 9 / 13
Medical Devices: Post Market Surveillance: National Competent Authority Report Exchange Criteria and Report Form : GHTF/SG2/N79R11:2009 Study Group 2 Final Document __________________________________________________________________________________________
On the rare occasions-when there are time critical issues of significant public health threat or concern-in addition to sending the report to the NCAR Secretariat, NCAs may send reports directly to countries participating in the NCAR exchange who are known to have the subject device in national distribution. In such circumstances, the issuing NCA should ensure that the form is completed fully and contains the correct sequential reference, preferably by contacting the NCAR Secretariat. Field: 1Please be sure to check Yes or No for confidentiality. This tells the recipient NCA if the information provided can be released publicly or must be held strictly confidential. 2Use the rules for numbering NCARs,(* use the ISO 3166 for country codes) which incorporates a two-letter code of the issuing country to fill in this item. For example: CA-2004-10-19-004 is a report from Canada sent 19 October 2004 and is the 4th report for 2004. Each NCAR should be given a new, unique NCAR number. If an NCAR relates to a previously exchanged NCAR (i.e : is an update), the original NCAR number should be specified in field 4. 3Insert any local reference number used by your NCA relevant to this report, here. 4If there have been previous NCARs exchanged relating to this one, regardless of source, insert their NCA exchange numbers here. 5Insert the manufacturers reference/recall number here, if applicable. 6Identify person and organization sending the NCAR. This should be the single point of contact, previously identified to the NCAR Secretariat. 7Identify contact person for any information / technical discussion of the topic. 8-10 - Telephone, Fax and e-mail of person in (7) above. 11 - Kind of device or generic descriptor. 12 - Identify the nomenclature system used (e.g. GMDN Global medicaldevice nomenclature, etc) . 13 - Number or code to identify the device based on the nomenclature system identified in (12). 14 - Trade name / Brand name AND Model number 15 - Self Explanatory 16-17 If there are many lot/batch numbers or serial numbers (ie: more than 3 or 4), a detailed list should be appended to the bottom of the report. 18 - Manufacturer of device - full address, including country, fax, phone numbers and email. 19 - Identify the authorized representative in reporting country (who is legally responsible for placing the subject device on the market where the incidents occurred), full address, including country, fax, phone numbers and e-mail. 20 - Indicate name or code number of Conformity Assessment Body/Notified Body involved, if applicable. 21 - a.) Identify approval status of the device in the region where the report originates. For example: CE-marking, approval number or licence number b.) Device risk class according to the jurisdiction of the issuing NCA can also be included. 22 - Identify any regulatory, legal or company-initiated action taken in advance of sending out the report. This could, for instance, refer to a Recall or the use of Safeguard action.
__________________________________________________________________________________________ February 2009 10 / 13
Medical Devices: Post Market Surveillance: National Competent Authority Report Exchange Criteria and Report Form: GHTF/SG2/N79R11:2009 Study Group 2 Final Document
___________________________________________________________________________ 23a - .Provide a description of what has happened, including consequences to patients or users. With reference to the criteria for reporting (SECTION 5 ABOVE), describe the reason for the report and why you want to inform other NCAs about these events. Such information will lead to a better understanding by the recipient on what is considered to be appropriate follow-up. 23b - Indicate if the investigation of the report is complete or not. 24a - Describe the outcome or conclusion of the investigation, to date. If useful, include a copy of any manufacturer or NCA advisory notice(s) associated with the NCAR and make reference to them within the NCAR. 24b - Indicate whether the manufacturers actions have been made public. 24c - Indicate whether originating NCA is willing to take the lead in co-ordination of the investigation. 25a - Recommendations to receivers of this report. 25b - List all countries known to have received the device. Put considerable care and effort into obtaining accurate information from the manufacturer for this field. 25c - List the marketed trade name(s) in other countries, if different. 26a - Indicate to whom the report has been sent. Care should be taken to indicate the correct distribution for the NCAR. Confidential NCARs should only be sent via the NCAR Secretariat to full NCAR participants not all NCAR participants. EEA European Economic Area , EC European commission and EFTA European federation trade associates NCAR participants should indicate direct distribution of the NCAR to EEA states, EC and EFTA NCAs in accordance European Medical Device Directives. NCAs outside the exchange program that are being sent the NCAR by the originating NCAR participant should also be listed. 26b - Where NCAR report numbers are not sequential, participants should include the number of the last report issued to all NCAR participants
__________________________________________________________________________________________ February 2009 11 / 13
Medical Devices: Post Market Surveillance: National Competent Authority Report Exchange Criteria and Report Form : GHTF/SG2/N79R11:2009 Study Group 2 Final Document
___________________________________________________________________________
7. Report Exchange Method
1) 2) 3) 4) Send the completed NCAR form to the NCAR Secretariat by email. A note indicating receipt of the NCAR form will be returned to the sender. The National Competent Authority Reporting Form should be used. Comments may be added to the report to maintain its confidentiality or to prevent public disclosure. For example, Still under investigation, do not disclose through access to information, or Do not release to public. Also, where possible and helpful, electronically include any background information such as a Dear Doctor letter or company letter. In order to minimize the risk of confusion, the NCAR Secretariat, on behalf of GHTF, will tentatively review the form for completeness, the correct sequential references and track the reports. Content is not edited. The form will then be forwarded by email to countries participating in the exchange. In rare circumstances, such as when there are time critical issues of significant public health threat or concern, NCAs may send reports directly to countries participating in the exchange. In such circumstances, the issuing NCA should ensure that the form is completed fully and contains the correct sequential reference, preferably by contacting the NCAR Secretariat. The issuing NCAR participant is the originator of the report unless the report says otherwise. Receiving NCAs may contact the source country of the report for more information if they wish. This shall be the first point of contact for incidents still under investigation.
5)
6)
7) 8)
__________________________________________________________________________________________ February 2009 12 / 13
Medical Devices: Post Market Surveillance: National Competent Authority Report Exchange Criteria and Report Form : GHTF/SG2/N79R11:2009 Study Group 2 Final Document
___________________________________________________________________________ Appendix A: Competent Authority Reporting Flowchart
Note: Please refer to section 4 of this document for guidance on using this chart.
__________________________________________________________________________________________ February 2009 13 / 13
Vous aimerez peut-être aussi
- Medical Devices and IVDs: Fit for the new EU-Regulations: Your complete seminar for projekt, study and jobD'EverandMedical Devices and IVDs: Fit for the new EU-Regulations: Your complete seminar for projekt, study and jobPas encore d'évaluation
- Post Market Surveillance: Global Guidance For Adverse Event Reporting For Medical DevicesDocument37 pagesPost Market Surveillance: Global Guidance For Adverse Event Reporting For Medical DevicesSachin KumarPas encore d'évaluation
- QP19-Vigilance Report - CE MarkDocument18 pagesQP19-Vigilance Report - CE Markanusha shankarPas encore d'évaluation
- Australia Post Market Activity GuidelinesDocument31 pagesAustralia Post Market Activity Guidelinesspenceblack7999Pas encore d'évaluation
- Guide Psur Apr 2021Document55 pagesGuide Psur Apr 2021ifrahPas encore d'évaluation
- EU NB-MED - 2.12 - Rec1 - Rev 11 - Post-Marketing Surveillance - PMSDocument9 pagesEU NB-MED - 2.12 - Rec1 - Rev 11 - Post-Marketing Surveillance - PMSAKSPas encore d'évaluation
- Psur Apr 2021Document14 pagesPsur Apr 2021Mauro CostaPas encore d'évaluation
- 2.PMS Report Template GRF - 25-46a - Rev - 1.2 - PMS - Appplication - FormDocument8 pages2.PMS Report Template GRF - 25-46a - Rev - 1.2 - PMS - Appplication - Formdelal karakuPas encore d'évaluation
- Sop VigilanceDocument7 pagesSop VigilanceJane BrownPas encore d'évaluation
- Template PMS Report TL01 V01Document9 pagesTemplate PMS Report TL01 V01Sandra SilvaPas encore d'évaluation
- Post Market Vigilance and FSCA With HSADocument8 pagesPost Market Vigilance and FSCA With HSASubashiиy PяabakaяaиPas encore d'évaluation
- PMS PMCF CER RelationshipDocument1 pagePMS PMCF CER RelationshipMohammed HammedPas encore d'évaluation
- Procedure For Complaint Handling-AOCDocument3 pagesProcedure For Complaint Handling-AOCMohamed EzzatPas encore d'évaluation
- Post-Marketing Surveillance PDFDocument10 pagesPost-Marketing Surveillance PDFscjofyWFawlroa2r06YFVabfbajPas encore d'évaluation
- EU MDR - Pinnacle Software Technologies LimitedDocument6 pagesEU MDR - Pinnacle Software Technologies LimitedPinnacle Software Technologies Limited100% (1)
- Fall Management Technical File DoC EU MDRDocument3 pagesFall Management Technical File DoC EU MDRvicPas encore d'évaluation
- PSUR Action Plan and Follow Up (R1)Document6 pagesPSUR Action Plan and Follow Up (R1)Bo Ram KimPas encore d'évaluation
- 510 (K) SUBSTANTIAL EQUIVALENCE DETERMINATIONDocument2 pages510 (K) SUBSTANTIAL EQUIVALENCE DETERMINATIONAlex JPas encore d'évaluation
- CE Marking RulesDocument46 pagesCE Marking RulesMusyoka UrbanusPas encore d'évaluation
- MDR - GAP ANALYSIS - TS - Q& E - Rev01Document53 pagesMDR - GAP ANALYSIS - TS - Q& E - Rev01Hariom Shukla100% (1)
- Example of MD PMS ActivitiesDocument6 pagesExample of MD PMS ActivitiesAKSPas encore d'évaluation
- Post Market Surveillance PlanDocument5 pagesPost Market Surveillance PlanBiolytic LifesciencesPas encore d'évaluation
- Risk Management ISO 14971 - 2019 r10v1.0 (01-08-20)Document84 pagesRisk Management ISO 14971 - 2019 r10v1.0 (01-08-20)AelindahPas encore d'évaluation
- Clinical Practice Evaluation 2Document21 pagesClinical Practice Evaluation 2api-477510991Pas encore d'évaluation
- Periodic Safety Update Report: ProductDocument5 pagesPeriodic Safety Update Report: ProductMauro CostaPas encore d'évaluation
- EMEA EtO GuidelineDocument3 pagesEMEA EtO GuidelineSpectre SpectrePas encore d'évaluation
- Language Implications of MDR For LabellingDocument15 pagesLanguage Implications of MDR For LabellingChan StephenPas encore d'évaluation
- Applicable Standards and Common SpecificationsDocument4 pagesApplicable Standards and Common Specificationsaymohamad2020Pas encore d'évaluation
- NEWS CENTER Maine (NCM) Sent A List of Questions To The FDA and These Were Their ResponsesDocument2 pagesNEWS CENTER Maine (NCM) Sent A List of Questions To The FDA and These Were Their ResponsesNEWS CENTER MainePas encore d'évaluation
- 1Document11 pages1Jonathan McloughlinPas encore d'évaluation
- MDCG 2020 - 5 Clinical Evaluation - Equivalence - April 2020Document20 pagesMDCG 2020 - 5 Clinical Evaluation - Equivalence - April 2020Kangjin JeonPas encore d'évaluation
- DePuy ASR PMCF StudiesDocument9 pagesDePuy ASR PMCF StudiesKaiser Gornick LLPPas encore d'évaluation
- Clarity COVID-19 Antigen Rapid Test - EC Declaration of Conformity - CE Marking and ApprovalDocument1 pageClarity COVID-19 Antigen Rapid Test - EC Declaration of Conformity - CE Marking and ApprovalCuong NguyenPas encore d'évaluation
- Post-Marketing Surveillance of Drugs: HapterDocument26 pagesPost-Marketing Surveillance of Drugs: HapterPalak AgarwalPas encore d'évaluation
- 06 MDCG 2020-6 Guidance On Sufficient Clinical Evidence For Legacy DevicesDocument22 pages06 MDCG 2020-6 Guidance On Sufficient Clinical Evidence For Legacy DevicesSantiago IbañezPas encore d'évaluation
- Design Dossier Technical Documentation EU R2 5 1-5 Rev4Document24 pagesDesign Dossier Technical Documentation EU R2 5 1-5 Rev4Andreas WeinfurterPas encore d'évaluation
- Design Dossiers ClassIIIDocument4 pagesDesign Dossiers ClassIIISanti PerezPas encore d'évaluation
- Clinical Evluation GiudenceDocument20 pagesClinical Evluation GiudenceabcPas encore d'évaluation
- Declaration of Interest - Clinical EvaluationDocument1 pageDeclaration of Interest - Clinical EvaluationAlex StiefenhoferPas encore d'évaluation
- 26 Vigilance Control System-R5Document18 pages26 Vigilance Control System-R5hitham shehataPas encore d'évaluation
- Risk Manangement For Medical DevicesDocument73 pagesRisk Manangement For Medical Deviceshamed IranpourPas encore d'évaluation
- Japan MHLW Ordinance 136 EnglishDocument16 pagesJapan MHLW Ordinance 136 Englishroxy_floxyPas encore d'évaluation
- Rules On Vigilance of Medical Devices PDFDocument8 pagesRules On Vigilance of Medical Devices PDFsks27974Pas encore d'évaluation
- EU PMS PSUR Requirements MDR PDFDocument9 pagesEU PMS PSUR Requirements MDR PDFHiral PatelPas encore d'évaluation
- Spentys - Technical File (Face Shield Mask)Document20 pagesSpentys - Technical File (Face Shield Mask)hitham shehataPas encore d'évaluation
- Post MarketSurveillancePlanTemplateDocument3 pagesPost MarketSurveillancePlanTemplateVomanh HealthcareandFitness100% (1)
- As 4369.1-1996 Surgical Dressings Single-Use X-Ray Detectable Abdominal SpongesDocument6 pagesAs 4369.1-1996 Surgical Dressings Single-Use X-Ray Detectable Abdominal SpongesSAI Global - APACPas encore d'évaluation
- Guidance Technical Documentation and Design Dossiers Fornon Active Medical DevicesDocument25 pagesGuidance Technical Documentation and Design Dossiers Fornon Active Medical DevicesCamila CamposPas encore d'évaluation
- Aggregate Reporting Psurs / Dsurs Best PracticesDocument26 pagesAggregate Reporting Psurs / Dsurs Best Practicesyousra yatrebPas encore d'évaluation
- Periodic Safety Update Reports: Adel Alrwisan B.SC, M.SC, PPCDocument16 pagesPeriodic Safety Update Reports: Adel Alrwisan B.SC, M.SC, PPCHala MohamedPas encore d'évaluation
- Medical Device Import PoliciesDocument4 pagesMedical Device Import PoliciesRavia SharmaPas encore d'évaluation
- Risk Management PlanDocument4 pagesRisk Management PlanprabhatgoaPas encore d'évaluation
- Sample SoP For Vigilance System PDFDocument4 pagesSample SoP For Vigilance System PDFhitham shehataPas encore d'évaluation
- Post Marketing SurveillanceDocument19 pagesPost Marketing SurveillanceRamanjeet SinghPas encore d'évaluation
- Executive SummaryDocument38 pagesExecutive Summaryimpexcoscopharmacist100% (1)
- Instrument Validation and Inspection MethodsDocument13 pagesInstrument Validation and Inspection MethodsHeena BhojwaniPas encore d'évaluation
- Med-Info: Biological EvaluationDocument4 pagesMed-Info: Biological EvaluationRand OmPas encore d'évaluation
- BSI Technical Guide Medical Protective Clothing en UkDocument6 pagesBSI Technical Guide Medical Protective Clothing en Uksutu ram100% (1)
- MDR Conformity Assessment Procedure: Class IibDocument1 pageMDR Conformity Assessment Procedure: Class IibDoaa KhalilPas encore d'évaluation
- Bsi VigilanceDocument19 pagesBsi VigilanceHazalPas encore d'évaluation
- PMA 2015 2016 Issue 3 PDFDocument36 pagesPMA 2015 2016 Issue 3 PDFLyndon Mercado TolentinoPas encore d'évaluation
- Shoulder Joint Position Sense Improves With ElevationDocument10 pagesShoulder Joint Position Sense Improves With ElevationpredragbozicPas encore d'évaluation
- Meditran SX Sae 15w 40 API CH 4Document1 pageMeditran SX Sae 15w 40 API CH 4Aam HudsonPas encore d'évaluation
- URICA TestDocument3 pagesURICA TestCristy PagalanPas encore d'évaluation
- Starbucks Reconciliation Template & Instructions v20231 - tcm137-84960Document3 pagesStarbucks Reconciliation Template & Instructions v20231 - tcm137-84960spaljeni1411Pas encore d'évaluation
- Planificare Săptămânală - Clasa A II-a (Fairyland 2)Document6 pagesPlanificare Săptămânală - Clasa A II-a (Fairyland 2)Oxy Oxy100% (1)
- BRS PDFDocument14 pagesBRS PDFGautam KhanwaniPas encore d'évaluation
- DET Tronics: Unitized UV/IR Flame Detector U7652Document2 pagesDET Tronics: Unitized UV/IR Flame Detector U7652Julio Andres Garcia PabolaPas encore d'évaluation
- MCS120 220 Error Ref - GAA30082DAC - RefDocument21 pagesMCS120 220 Error Ref - GAA30082DAC - RefCoil98Pas encore d'évaluation
- Governance StructureDocument1 pageGovernance StructureJoydip MukhopadhyayPas encore d'évaluation
- Chapter 1 Fundamentals of Taxation by Cruz, Deschamps, Miswander, Prendergast, Schisler, and TroneDocument25 pagesChapter 1 Fundamentals of Taxation by Cruz, Deschamps, Miswander, Prendergast, Schisler, and TroneReese Parker100% (4)
- AACO 7th Aviation Fuel Forum: AttendanceDocument3 pagesAACO 7th Aviation Fuel Forum: AttendanceJigisha Vasa0% (1)
- Why Men Want Sex and Women Need Love by Barbara and Allen Pease - ExcerptDocument27 pagesWhy Men Want Sex and Women Need Love by Barbara and Allen Pease - ExcerptCrown Publishing Group62% (34)
- Content of An Investigational New Drug Application (IND)Document13 pagesContent of An Investigational New Drug Application (IND)Prathamesh MaliPas encore d'évaluation
- Internationalresidential Code 2009 Edition Fuel Gas SectionDocument49 pagesInternationalresidential Code 2009 Edition Fuel Gas SectionZarex BorjaPas encore d'évaluation
- Upper Limb OrthosesDocument29 pagesUpper Limb OrthosesMaryam KhalidPas encore d'évaluation
- The Development of Attachment in Separated and Divorced FamiliesDocument33 pagesThe Development of Attachment in Separated and Divorced FamiliesInigo BorromeoPas encore d'évaluation
- MGMTDocument19 pagesMGMTMakrant MohanPas encore d'évaluation
- Fortune Falls - Lou VaneDocument402 pagesFortune Falls - Lou VanetestePas encore d'évaluation
- Unit Two The Health Benefits of Physical ActivityDocument10 pagesUnit Two The Health Benefits of Physical ActivityYoseph DefaruPas encore d'évaluation
- 00516-CLIA-Newsletter Jan 06Document4 pages00516-CLIA-Newsletter Jan 06losangelesPas encore d'évaluation
- Clinical Features, Evaluation, and Diagnosis of Sepsis in Term and Late Preterm Infants - UpToDateDocument42 pagesClinical Features, Evaluation, and Diagnosis of Sepsis in Term and Late Preterm Infants - UpToDatedocjime9004Pas encore d'évaluation
- Impact of Job Design On Employee Engagement: A Theoretical and Literature ReviewDocument6 pagesImpact of Job Design On Employee Engagement: A Theoretical and Literature ReviewAnonymous CwJeBCAXpPas encore d'évaluation
- Aquamine 50.01Document17 pagesAquamine 50.01Armando RelajoPas encore d'évaluation
- Ca 2013 39Document40 pagesCa 2013 39singh1699Pas encore d'évaluation
- Method Statement For Lifting WorksDocument12 pagesMethod Statement For Lifting WorksRachel Flores85% (26)
- PMEGP Revised Projects (Mfg. & Service) Vol 1Document260 pagesPMEGP Revised Projects (Mfg. & Service) Vol 1Santosh BasnetPas encore d'évaluation
- Transmission Lines SMART EDGE VILLARUEL For April 2024 v1Document89 pagesTransmission Lines SMART EDGE VILLARUEL For April 2024 v1mayandichoso24Pas encore d'évaluation
- Cisco - Level 45Document1 pageCisco - Level 45vithash shanPas encore d'évaluation