Académique Documents
Professionnel Documents
Culture Documents
Stirling Cycle
Transféré par
Christian_Joe__9147Description originale:
Copyright
Formats disponibles
Partager ce document
Partager ou intégrer le document
Avez-vous trouvé ce document utile ?
Ce contenu est-il inapproprié ?
Signaler ce documentDroits d'auteur :
Formats disponibles
Stirling Cycle
Transféré par
Christian_Joe__9147Droits d'auteur :
Formats disponibles
Gas Power Cycles Mallikarjuna
Prof. U.S.P. Shet , Prof. T. Sundararajan and Prof. J.M .
4.2 Stirling Cycle (Regenerative Cycle) :
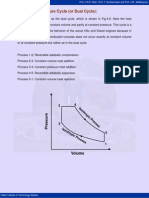
The Carnot cycle has a low mean effective pressure because of its very low work output. Hence, one of the modified forms of the cycle to produce higher mean effective pressure whilst theoretically achieving full Carnot cycle efficiency is the Stirling cycle. It consists of two isothermal and two constant volume processes. The heat rejection and addition take place at constant temperature. The p-v and T-s diagrams for the Stirling cycle are shown in Fig.4.2.
3 4 2 1
Volume
Temperature T3=T4
3 4
T2=T1
Entropy
Fig.4.2. Stirling cycle processes on p-v and T-s diagrams
Indian Institute of Technology Madras
Gas Power Cycles Mallikarjuna
Prof. U.S.P. Shet , Prof. T. Sundararajan and Prof. J.M .
Stirling Cycle Processes:
(a) The air is compressed isothermally from state 1 to 2 (TL to TH). (b) The air at state-2 is passed into the regenerator from the top at a temperature T1. The air passing through the regenerator matrix gets heated from TL to TH. (c) The air at state-3 expands isothermally in the cylinder until it reaches state-4. (d) The air coming out of the engine at temperature TH (condition 4) enters into regenerator from the bottom and gets cooled while passing through the regenerator matrix at constant volume and it comes out at a temperature TL, at condition 1 and the cycle is repeated. (e) It can be shown that the heat absorbed by the air from the regenerator matrix during the process 2-3 is equal to the heat given by the air to the regenerator matrix during the process 4-1, then the exchange of heat with external source will be only during the isothermal processes.
Now we can write, Net work done = W = Qs - QR Heat supplied = QS = heat supplied during the isothermal process 3-4.
V v = P 3 3ln 4 ; r = 4 = CR V v3 V3 = mRTH ln ( r )
Heat rejected = QR = Heat rejected during the isothermal compression process, 1-2. v = P1V1 ln 1 v2 = mR TL ln ( r ) Wnet = m R ln ( r ) [ T - T ] H L
Indian Institute of Technology Madras
Gas Power Cycles Mallikarjuna
Prof. U.S.P. Shet , Prof. T. Sundararajan and Prof. J.M .
Now, th = Wnet Qs and th =1TL TH m R ln ( r ) = ( TH - TL ) TH - TL TH
m R ln ( r ) TH
Thus the efficiency of Stirling cycle is equal to that of Carnot cycle efficiency when both are working with the same temperature limits. It is not possible to obtain 100% efficient regenerator and hence there will be always 10 to 20 % loss of heat in the regenerator, which decreases the cycle efficiency. Considering regenerator efficiency, the efficiency of the cycle can be written as,
th =
R TH ln ( r ) + (1 - R ) C V ( TH - TL )
R ln ( r )( TH - TL )
Where, R is the regenerator efficiency.
Indian Institute of Technology Madras
Vous aimerez peut-être aussi
- Understanding the Stirling Cycle Gas Power ProcessDocument3 pagesUnderstanding the Stirling Cycle Gas Power ProcessRon QuerubinPas encore d'évaluation
- Otto CycleDocument5 pagesOtto CycleSaraju NandiPas encore d'évaluation
- Nptel - Ac.in Courses IIT-MADRAS Applied Thermodynamics Module 4 6 AsdcDocument4 pagesNptel - Ac.in Courses IIT-MADRAS Applied Thermodynamics Module 4 6 AsdcSanjeev PrakashPas encore d'évaluation
- Limited Pressure CycleDocument4 pagesLimited Pressure Cyclecasu19Pas encore d'évaluation
- IC Engines 2012 Edition Theory & QuestionsDocument178 pagesIC Engines 2012 Edition Theory & Questionskumarrohit91Pas encore d'évaluation
- Stirling and Ericsson Cycles 2016 PDFDocument13 pagesStirling and Ericsson Cycles 2016 PDFArvin Loui Bascon100% (1)
- 2 - RankinecycleDocument4 pages2 - RankinecycleAnonymous BHBWQIcClPas encore d'évaluation
- Rankine cycle analysis and efficiency calculationDocument4 pagesRankine cycle analysis and efficiency calculationSami UllahPas encore d'évaluation
- Rankine cycle analysis and efficiency calculationDocument4 pagesRankine cycle analysis and efficiency calculationMantript SinghPas encore d'évaluation
- 2 RankinecycleDocument4 pages2 Rankinecyclesoubhadra nagPas encore d'évaluation
- 5.2 Rankine Cycle:: BoilerDocument4 pages5.2 Rankine Cycle:: BoilerVidyut KumarPas encore d'évaluation
- 2 RankinecycleDocument4 pages2 RankinecycleAmit PandeyPas encore d'évaluation
- 2 Rankinecycle-2Document4 pages2 Rankinecycle-2Anonymous gS6JtOfaPas encore d'évaluation
- 5.2 Rankine Cycle:: BoilerDocument4 pages5.2 Rankine Cycle:: BoilerSasi GunaPas encore d'évaluation
- Rankine cycle analysis and efficiency calculationDocument4 pagesRankine cycle analysis and efficiency calculationJohn FredyPas encore d'évaluation
- Stirling and Ericsson Cycles 2016 PDFDocument13 pagesStirling and Ericsson Cycles 2016 PDFMarion Villamor100% (7)
- RAC TutorialDocument30 pagesRAC Tutorialkhushal bhanderiPas encore d'évaluation
- 2 Carnot CycleDocument6 pages2 Carnot CycleSamujawal Kumar SumanPas encore d'évaluation
- Lesson 9 Air cycle refrigeration systemsDocument163 pagesLesson 9 Air cycle refrigeration systemsali105Pas encore d'évaluation
- Cycles PDFDocument10 pagesCycles PDFratchagarajaPas encore d'évaluation
- Air Cycle Refrigeration Systems (Indian Institute of Technology Kharagpur)Document15 pagesAir Cycle Refrigeration Systems (Indian Institute of Technology Kharagpur)emawz21Pas encore d'évaluation
- 2 - RankinecycleDocument4 pages2 - RankinecycletpplantPas encore d'évaluation
- Rankine CycleDocument4 pagesRankine Cyclerishav43Pas encore d'évaluation
- 2 - RankinecycleDocument4 pages2 - RankinecycleamdevaPas encore d'évaluation
- Rankine Cycle Vapour PowerDocument4 pagesRankine Cycle Vapour PowerEnny Rosmawar PurbaPas encore d'évaluation
- Rankine Cycle Vapour PowerDocument4 pagesRankine Cycle Vapour PowerAtanu Brainless RoyPas encore d'évaluation
- Rankine Cycle Vapour PowerDocument4 pagesRankine Cycle Vapour PowerPrasad KhatiPas encore d'évaluation
- 2 - RankinecycleDocument4 pages2 - RankinecycleRishabh VermaPas encore d'évaluation
- 2 - RankinecycleDocument4 pages2 - Rankinecycledurga345Pas encore d'évaluation
- 2 - RankinecycleDocument4 pages2 - RankinecycleamdevaPas encore d'évaluation
- 2 - RankinecycleDocument4 pages2 - RankinecycleRishabh VermaPas encore d'évaluation
- 2 - RankinecycleDocument4 pages2 - RankinecycleamdevaPas encore d'évaluation
- 2 - RankinecycleDocument4 pages2 - RankinecyclePrasad KhatiPas encore d'évaluation
- Applied Termo 122424Document4 pagesApplied Termo 122424rjrahul25Pas encore d'évaluation
- COP vs cooling load characteristics irreversible air cycleDocument9 pagesCOP vs cooling load characteristics irreversible air cycleYalçın DurmuşoğluPas encore d'évaluation
- Lecture Notes OnDocument200 pagesLecture Notes Onananth k r100% (3)
- 07 Gas Turbine Power PlantsDocument37 pages07 Gas Turbine Power PlantsLozada JohnPas encore d'évaluation
- SPPU Sem 6 ARAC 1, 2& 3 NOTESDocument52 pagesSPPU Sem 6 ARAC 1, 2& 3 NOTESAshish RejikumarPas encore d'évaluation
- Turbine: The Effect of Friction On The Joule CycleDocument13 pagesTurbine: The Effect of Friction On The Joule CycleGeorge AniborPas encore d'évaluation
- Air Standard Cycles - BasicsDocument19 pagesAir Standard Cycles - Basicsexpressive87Pas encore d'évaluation
- Brayton CycleDocument5 pagesBrayton CycleAnonymous yorzHjDBd100% (1)
- Ideal Engine CycleDocument20 pagesIdeal Engine CycleMulugeta WoldePas encore d'évaluation
- Experiment 2: A Model Energy Conversion System: Operation and Analysis of A Stirling EngineDocument11 pagesExperiment 2: A Model Energy Conversion System: Operation and Analysis of A Stirling Enginetimur_okPas encore d'évaluation
- Air Standard Cycles - BasicsDocument34 pagesAir Standard Cycles - Basicsrazvan66m100% (1)
- Engineering Council Certificate Level Thermodynamic, Fluid and Process Engineering C106 Tutorial 5 - Ideal Engine CyclesDocument14 pagesEngineering Council Certificate Level Thermodynamic, Fluid and Process Engineering C106 Tutorial 5 - Ideal Engine CyclesDipeshPas encore d'évaluation
- Vapour Power Cycles and Steam NozzlesDocument48 pagesVapour Power Cycles and Steam NozzlesGurpreet MeeluPas encore d'évaluation
- Gas TurbineDocument7 pagesGas TurbineVishal BediPas encore d'évaluation
- Finite Physical Dimensions Optimal Thermodynamics 1: FundamentalsD'EverandFinite Physical Dimensions Optimal Thermodynamics 1: FundamentalsPas encore d'évaluation
- Treatise on Irreversible and Statistical Thermodynamics: An Introduction to Nonclassical ThermodynamicsD'EverandTreatise on Irreversible and Statistical Thermodynamics: An Introduction to Nonclassical ThermodynamicsÉvaluation : 1 sur 5 étoiles1/5 (1)
- Power Electronics Applied to Industrial Systems and Transports, Volume 2: Power Converters and their ControlD'EverandPower Electronics Applied to Industrial Systems and Transports, Volume 2: Power Converters and their ControlÉvaluation : 5 sur 5 étoiles5/5 (1)
- Working Guide to Vapor-Liquid Phase Equilibria CalculationsD'EverandWorking Guide to Vapor-Liquid Phase Equilibria CalculationsÉvaluation : 5 sur 5 étoiles5/5 (1)
- “Foundations to Flight: Mastering Physics from Curiosity to Confidence: Cipher 4”: “Foundations to Flight: Mastering Physics from Curiosity to Confidence, #4D'Everand“Foundations to Flight: Mastering Physics from Curiosity to Confidence: Cipher 4”: “Foundations to Flight: Mastering Physics from Curiosity to Confidence, #4Pas encore d'évaluation
- 18mba0044 SCM Da2Document4 pages18mba0044 SCM Da2Prabhu ShanmugamPas encore d'évaluation
- Troublesshooting Manual 3b6 PDFDocument50 pagesTroublesshooting Manual 3b6 PDFAgnieszka Wawrzyniak Rybka100% (1)
- Intangible Capital: Key Factor of Sustainable Development in MoroccoDocument8 pagesIntangible Capital: Key Factor of Sustainable Development in MoroccojournalPas encore d'évaluation
- List of Non-Scheduled Urban Co-Operative Banks: Sr. No. Bank Name RO Name Head Office Address PincodeDocument65 pagesList of Non-Scheduled Urban Co-Operative Banks: Sr. No. Bank Name RO Name Head Office Address PincodemanojPas encore d'évaluation
- Aartv Industrial Training ReportDocument48 pagesAartv Industrial Training ReportRupal NaharPas encore d'évaluation
- Final Eligible Voters List North Zone 2017 118 1Document12 pagesFinal Eligible Voters List North Zone 2017 118 1Bilal AhmedPas encore d'évaluation
- Applying problem solving techniques to routine computer issuesDocument2 pagesApplying problem solving techniques to routine computer issuesBirhanu Atnafu0% (1)
- Cladding & Hardfacing ProcessesDocument16 pagesCladding & Hardfacing ProcessesMuhammed SulfeekPas encore d'évaluation
- Manufacturing and Service TechnologiesDocument18 pagesManufacturing and Service Technologiesajit123ajitPas encore d'évaluation
- Opentbs Demo: Merge Data With A ChartDocument4 pagesOpentbs Demo: Merge Data With A Charteduarditom1Pas encore d'évaluation
- Chapter 9 PowerPointDocument33 pagesChapter 9 PowerPointYusrah JberPas encore d'évaluation
- House Bill 470Document9 pagesHouse Bill 470Steven DoylePas encore d'évaluation
- CH 5 Audit-Planning MCQDocument3 pagesCH 5 Audit-Planning MCQEhab hoba0% (1)
- Visual Basic Is Initiated by Using The Programs OptionDocument141 pagesVisual Basic Is Initiated by Using The Programs Optionsaroj786Pas encore d'évaluation
- PT-E - Fundamentals of Process Plant Layout and Piping DesignDocument14 pagesPT-E - Fundamentals of Process Plant Layout and Piping DesignNofrizal HasanPas encore d'évaluation
- 280Document6 pages280Alex CostaPas encore d'évaluation
- Presentasi AkmenDocument18 pagesPresentasi AkmenAnonymous uNgaASPas encore d'évaluation
- Design and Construction of Water Supply SchemeDocument126 pagesDesign and Construction of Water Supply SchemeShreyansh SharmaPas encore d'évaluation
- Office of The Integrity Commissioner - Investigation Report Regarding The Conduct of Councillor Mark Grimes (July 05, 2016)Document44 pagesOffice of The Integrity Commissioner - Investigation Report Regarding The Conduct of Councillor Mark Grimes (July 05, 2016)T.O. Nature & DevelopmentPas encore d'évaluation
- MAINTENANCE AND RELIABILITY ENGINEERING - Lecture 1Document24 pagesMAINTENANCE AND RELIABILITY ENGINEERING - Lecture 1K ULAGANATHANPas encore d'évaluation
- Ciphertext-Policy Attribute-Based EncryptionDocument15 pagesCiphertext-Policy Attribute-Based EncryptionJ_RameshPas encore d'évaluation
- Theory of Elinor OstromDocument5 pagesTheory of Elinor OstromSanjana KrishnakumarPas encore d'évaluation
- m100 Stopswitch ManualDocument12 pagesm100 Stopswitch ManualPatrick TiongsonPas encore d'évaluation
- Business Process Reengineering and Performance Improvement in The BankDocument12 pagesBusiness Process Reengineering and Performance Improvement in The BankYakut Rumani SultanPas encore d'évaluation
- Marie Bjerede and Tzaddi Bondi 2012 - Learning Is Personal, Stories of Android Tablet Use in The 5th GradeDocument50 pagesMarie Bjerede and Tzaddi Bondi 2012 - Learning Is Personal, Stories of Android Tablet Use in The 5th Gradeluiz carvalhoPas encore d'évaluation
- Introduction To Global Business 2nd Edition Gaspar Test BankDocument26 pagesIntroduction To Global Business 2nd Edition Gaspar Test BankJerryGarrettmwsi100% (56)
- VR Headset QR CodesDocument35 pagesVR Headset QR CodesAbdelsabour Ahmed100% (1)
- Kokichi OmaSprite Gallery Danganronpa Wiki FDocument3 pagesKokichi OmaSprite Gallery Danganronpa Wiki Fmzcvxf9fwpPas encore d'évaluation
- Digital Payments in IndiaDocument6 pagesDigital Payments in IndiaInternational Journal of Innovative Science and Research TechnologyPas encore d'évaluation
- Chemical Content of FertilizersDocument22 pagesChemical Content of FertilizersJohn DAcquisto, D.Sc.100% (1)