Académique Documents
Professionnel Documents
Culture Documents
RNA Purification System
Transféré par
AlleleBiotechCopyright
Formats disponibles
Partager ce document
Partager ou intégrer le document
Avez-vous trouvé ce document utile ?
Ce contenu est-il inapproprié ?
Signaler ce documentDroits d'auteur :
Formats disponibles
RNA Purification System
Transféré par
AlleleBiotechDroits d'auteur :
Formats disponibles
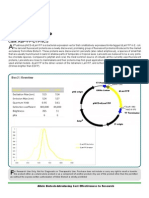
RNA Purification System,
Employs modified silica membranes as selective nucleic acid captures in a spin-column format.
he Allele RNA Purification Kit is designed for the purification of high-quality RNA from total RNA or RNA enzymatic reactions, such as in vitro transcription. This system employs modified silica membranes as selective nucleic acid captures in a spin-column format. RNA molecules (ssRNA > 80 bases, dsRNA > 150 bps), including RNAs from in vitro transcription reaction, capped RNAs, amino allyl-modified RNAs, Biotin and Cy-dye labeled RNAs can specifically interact with the modified silica membrane in the RNA Binding Buffer, while nucleotides, short oligonucleotides, salts, proteins and other contaminants are not bound and remain in the solution which is passed through. After washing the column with washing buffer to remove unbound material, the purified RNA can be easily eluted in 10 mM Tris Elution Buffer or water. Purified RNA can be used for any downstream application that requires high quality RNA, such as qRT-PCR, microarray, RNA-seq, RNase protection assay and other transcriptional RNA analysis.
Features
Fast and Easy Protocol: The procedure consists of three steps: RNA binding to membrane, contaminant being washed away and RNA elution from membrane. It takes less than 15 minutes to finish the purification procedure. High Yield and Quality: The modified membrane and RNA binding buffer system provides for maximum RNA capturing and release. The recovery is more than 75% of input RNA of high purity with efficient removal ofnucleotides, short oligonucleotides, salts and proteins. Technical data: RNA length of ssRNA > 80 bases or dsRNA > 150 bps is recommended for the procedure. RNA input from 1 g to 300 g can be purified with each purification.
General Precautions
Box 1 | Contents
Kit Contents
# of Purifications ABP-PP-RNAPUR025 ABP-PP-RNAPUR050
This kit is for research use only. All due care and attention should be exercised in the handling of the kits.
25 25 50 50
CC1:
Spin-Columns with Collection Tubes
RT1:
Recovery Tubes
25 5.0 mL 5.0 mL 6.25 mL
50 10 mL 10 mL 12.5 mL
Wear a laboratory coat, disposable gloves, and eye protection when handling reagents and tubes. Avoid ingestion and inhalation of reagents. In case of contact, wash thoroughly with water. See Material Safety Data Sheets (MSDS) for emergency procedures in case of accidental contact or ingestion. MSDS information is available upon request. Always use proper aseptic techniques to avoid nuclease contamination when working with RNA. Use only sterile, new pipette tips to prevent cross contamination.
RB4:
RNA Binding Buffer
WB2: EB3:
Washing Buffer II Elution Buffer
Preparation
WB2: For ABP-PP-RNAPUR025 kit, add 20 ml of
100% ethanol to Washing Buffer II (WB2) and mix well. For ABP-PP-RNAPUR050 kit, add 40 ml of 100% ethanol to Washing Buffer II (WB2) and mix well. Mark bottle that ethanol has been added. Store at room temperature and use Washing Buffer II (WB2) containing ethanol within six (6) months. Avoid nuclease contamination and clean lab bench with RNase inactivating agents such as RNase-Off or RNase-Away and wipe with 75%ethanol. Set a water baths or heat blocks at 94 C.
Additional Materials Needed 100% Ethanol (ACS grade or better) Pipettor and RNase-free tips Benchtop Centrifuge and Vortexer Nuclease-free 1. 7 ml microcentrifuge tubes Heating block or water bath
he components included with RNA Purification System are listed above. Upon receipt, store all components at room temperature with the exception of RNA Binding Buffer (RB4), which needs to be stored at 4 C..
Allele Biotech-Introducing Cost Effectiveness to Research
The protocol below is for the purification of RNA from 50 l reaction. The purification procedure may be scaled from 10100 l by proportionately adjusting all reagents throughout the procedure.
Protocols
Binding RNA to Spin Column 1. Bring the RNA sample to 50l with Elution Buffer (EB3) and mix well. 2. Add 170l RNA Binding Buffer (RB4) to the sample and mix well by pi petting solution up and down 7- 8 times to yield a homogenous solution. 3. Add 130l 100% ethanol to the sample and mix well by pipetting solution up and down 7-8 times to yield a homogenous solution. 4. Transfer 350l of the sample solution from step above to a Spin-Column with a Collection Tube (CCI). 5. Centrifuge the Spin-Column at 8000 x g for one minute at room temperature. Discard the filtrate in the Collection Tube and reinsert the Spin-Column back into the Collection Tube. Washing RNA Products 1. Add 400 l of Wash Buffer II (WB2) with ethanol to the Column. 2. Centrifuge the Spin-Column at 8000 g for 1 minute at room temperature. Discard the filtrate. 3. Repeat step 1 and 2 above for a total of two washes. Discard the filtrate. 4. Centrifuge the spin-column at maximum speed (10000- 15000 g, or 10000- 14000 rpm) for extra 1 minute to remove any residual wash buffer with ethanol. Discard Collection Tube. 5. Reinsert the Spin-Column into a new clean microcentrifuge Recovery Tube (RTI). Eluting RNA Products 1. Preheat 200-1000l Elution Buffer (EB3) in a 1.7 ml tube to 94 C with a heat block or a water bath. 2. Add 50l pre-heated Elution Buffer (EB3) into the center of the Spin-Column. 3. Incubate the column for 1 minute at room temperature. 4. Centrifuge the column at maximum speed for 1 minute at room temperature. The Recovery Tube (RTI) contains purified RNA.
5. To maximize RNA recovery, you may perform a second elution with 50l pre-heated Elution Buffer (EB3) with the same recovery tube, if desired. This can increase the total RNA yield by 10-30 %, but the final concentration of purified RNA in the recovery tube is reduced because of the increased volume. 6. Store the purified RNA on ice. At this point, samples are ready for desired downstream application such as qRT-PCR, or store the RNA samples at -80 C for long-term storage until further use.
Electrophoresis and Downstream Application Purified RNA can be examined by agarose gel electrophoresis. Yield can be measured with a spectrophotometer at 260 nm, fluorescent RNA assays or other quantification methods by diluting an aliquot of the purified RNA sample (usually 1:50- 1:200 dilution). For electrophoresis, loading 3-8 l purified RNA is recommended. The purified RNA is suitable for use in qRTPCR, microarray, RNA-seq, RNase protection assay and other transcriptional RNA analysis.
Allele Biotech-Introducing Cost Effectiveness to Research
Box 2 | Troubleshooting Problem Cause Amount of RNA used was less than recommended or poor quality of starting material Incomplete Mixing Low product yield Solution Check original RNA with gel electrophoresis or Agilent 2100 OR Add additional RNA sample. Mix RNA sample with RNA Binding Buffer and 100% Ethanol thoroughly before loading onto spin-column Make sure lab bench and pipettes are clean and RNase-free. Wear gloves at all times during the whole procedure and change gloves frequently to protect reagents and RNA from nuclease that are present on skin. Use RNase-free pipette tips and tubes to handle all solutions; avoid used tips or used tubes for reagents Be sure to add all components. Check positive and negative controls for RTPCR reaction.
Nuclease contamination
No RT-PCR product
Missing Component in the RT-PCR mixture
Welcome to Join in the Discussion at Alleles Network: Allele News: news.allelebiotech.com Allele Blog: blogs.allelebiotech.com Allele Facebook: http://www.facebook.com/ Allele Twitter: http://www.twitter.com/allele_biotech
www.allelebiotech.com
Allele Biotech-Introducing Cost Effectiveness to Research
Vous aimerez peut-être aussi
- Modified UTPDocument1 pageModified UTPAlleleBiotechPas encore d'évaluation
- Modified CTPDocument1 pageModified CTPAlleleBiotechPas encore d'évaluation
- M13KO7 Helper PhageDocument1 pageM13KO7 Helper PhageAlleleBiotechPas encore d'évaluation
- Anti-RFP (5F8) Monoclonal AntibodyDocument1 pageAnti-RFP (5F8) Monoclonal AntibodyAlleleBiotechPas encore d'évaluation
- PNCS dLanYFPDocument1 pagePNCS dLanYFPAlleleBiotechPas encore d'évaluation
- High Quality, Standard RGTP Suitable For Large Scale IVT.: DescriptionDocument1 pageHigh Quality, Standard RGTP Suitable For Large Scale IVT.: DescriptionAlleleBiotechPas encore d'évaluation
- High Quality, Standard rATP Suitable For Large Scale IVT.: DescriptionDocument1 pageHigh Quality, Standard rATP Suitable For Large Scale IVT.: DescriptionAlleleBiotechPas encore d'évaluation
- Allele AgaroseDocument1 pageAllele AgaroseAlleleBiotechPas encore d'évaluation
- Anti-RFP (3F5) Monoclonal AntibodyDocument1 pageAnti-RFP (3F5) Monoclonal AntibodyAlleleBiotechPas encore d'évaluation
- Luciferase PCR Template For IVTDocument1 pageLuciferase PCR Template For IVTAlleleBiotechPas encore d'évaluation
- pmTFP1 ClathrinDocument4 pagespmTFP1 ClathrinAlleleBiotechPas encore d'évaluation
- Mwasabi PCR Template For IVTDocument1 pageMwasabi PCR Template For IVTAlleleBiotechPas encore d'évaluation
- Custom Sub Cloning ServiceDocument1 pageCustom Sub Cloning ServiceAlleleBiotechPas encore d'évaluation
- mClavGR2 Fusion VectorsDocument3 pagesmClavGR2 Fusion VectorsAlleleBiotechPas encore d'évaluation
- 3' DC-CPGDocument1 page3' DC-CPGAlleleBiotechPas encore d'évaluation
- 3' DT-CPGDocument1 page3' DT-CPGAlleleBiotechPas encore d'évaluation
- 3' DG-CPGDocument1 page3' DG-CPGAlleleBiotechPas encore d'évaluation
- ChromoTek RFP BoosterDocument1 pageChromoTek RFP BoosterAlleleBiotechPas encore d'évaluation
- 3'-Thiol-Modifier C6 S-S CPGDocument1 page3'-Thiol-Modifier C6 S-S CPGAlleleBiotechPas encore d'évaluation
- Gryphon Selection MediumDocument1 pageGryphon Selection MediumAlleleBiotechPas encore d'évaluation
- 3'-Amino-Modifier C7 CPGDocument1 page3'-Amino-Modifier C7 CPGAlleleBiotechPas encore d'évaluation
- Shoe Dog: A Memoir by the Creator of NikeD'EverandShoe Dog: A Memoir by the Creator of NikeÉvaluation : 4.5 sur 5 étoiles4.5/5 (537)
- The Yellow House: A Memoir (2019 National Book Award Winner)D'EverandThe Yellow House: A Memoir (2019 National Book Award Winner)Évaluation : 4 sur 5 étoiles4/5 (98)
- The Subtle Art of Not Giving a F*ck: A Counterintuitive Approach to Living a Good LifeD'EverandThe Subtle Art of Not Giving a F*ck: A Counterintuitive Approach to Living a Good LifeÉvaluation : 4 sur 5 étoiles4/5 (5794)
- The Little Book of Hygge: Danish Secrets to Happy LivingD'EverandThe Little Book of Hygge: Danish Secrets to Happy LivingÉvaluation : 3.5 sur 5 étoiles3.5/5 (400)
- Grit: The Power of Passion and PerseveranceD'EverandGrit: The Power of Passion and PerseveranceÉvaluation : 4 sur 5 étoiles4/5 (588)
- Elon Musk: Tesla, SpaceX, and the Quest for a Fantastic FutureD'EverandElon Musk: Tesla, SpaceX, and the Quest for a Fantastic FutureÉvaluation : 4.5 sur 5 étoiles4.5/5 (474)
- A Heartbreaking Work Of Staggering Genius: A Memoir Based on a True StoryD'EverandA Heartbreaking Work Of Staggering Genius: A Memoir Based on a True StoryÉvaluation : 3.5 sur 5 étoiles3.5/5 (231)
- Hidden Figures: The American Dream and the Untold Story of the Black Women Mathematicians Who Helped Win the Space RaceD'EverandHidden Figures: The American Dream and the Untold Story of the Black Women Mathematicians Who Helped Win the Space RaceÉvaluation : 4 sur 5 étoiles4/5 (895)
- Team of Rivals: The Political Genius of Abraham LincolnD'EverandTeam of Rivals: The Political Genius of Abraham LincolnÉvaluation : 4.5 sur 5 étoiles4.5/5 (234)
- Never Split the Difference: Negotiating As If Your Life Depended On ItD'EverandNever Split the Difference: Negotiating As If Your Life Depended On ItÉvaluation : 4.5 sur 5 étoiles4.5/5 (838)
- The Emperor of All Maladies: A Biography of CancerD'EverandThe Emperor of All Maladies: A Biography of CancerÉvaluation : 4.5 sur 5 étoiles4.5/5 (271)
- Devil in the Grove: Thurgood Marshall, the Groveland Boys, and the Dawn of a New AmericaD'EverandDevil in the Grove: Thurgood Marshall, the Groveland Boys, and the Dawn of a New AmericaÉvaluation : 4.5 sur 5 étoiles4.5/5 (266)
- On Fire: The (Burning) Case for a Green New DealD'EverandOn Fire: The (Burning) Case for a Green New DealÉvaluation : 4 sur 5 étoiles4/5 (74)
- The Unwinding: An Inner History of the New AmericaD'EverandThe Unwinding: An Inner History of the New AmericaÉvaluation : 4 sur 5 étoiles4/5 (45)
- The Hard Thing About Hard Things: Building a Business When There Are No Easy AnswersD'EverandThe Hard Thing About Hard Things: Building a Business When There Are No Easy AnswersÉvaluation : 4.5 sur 5 étoiles4.5/5 (345)
- The World Is Flat 3.0: A Brief History of the Twenty-first CenturyD'EverandThe World Is Flat 3.0: A Brief History of the Twenty-first CenturyÉvaluation : 3.5 sur 5 étoiles3.5/5 (2259)
- The Gifts of Imperfection: Let Go of Who You Think You're Supposed to Be and Embrace Who You AreD'EverandThe Gifts of Imperfection: Let Go of Who You Think You're Supposed to Be and Embrace Who You AreÉvaluation : 4 sur 5 étoiles4/5 (1090)
- The Sympathizer: A Novel (Pulitzer Prize for Fiction)D'EverandThe Sympathizer: A Novel (Pulitzer Prize for Fiction)Évaluation : 4.5 sur 5 étoiles4.5/5 (121)
- Her Body and Other Parties: StoriesD'EverandHer Body and Other Parties: StoriesÉvaluation : 4 sur 5 étoiles4/5 (821)