Académique Documents
Professionnel Documents
Culture Documents
Carbon Steel
Transféré par
Tina MillerCopyright
Formats disponibles
Partager ce document
Partager ou intégrer le document
Avez-vous trouvé ce document utile ?
Ce contenu est-il inapproprié ?
Signaler ce documentDroits d'auteur :
Formats disponibles
Carbon Steel
Transféré par
Tina MillerDroits d'auteur :
Formats disponibles
Weldability of steels
Carbon steels
Low alloy steels (Cr - Mo steels)
Quenched tempered steels
High alloy steel
Problems
Hydrogen cracking
Solidification cracking
Lamellar tearing
Hydrogen induced crack
Cold crack
Underbead crack
Delayed crack
Hydrogen induced crack
Fillet and butt welds HAZ
Fillet and butt welds
Hydrogen cracking
Carbon equivalent C+Mn/4
Ductility
Hardness
Crack
sensitivity
Weldability of steels
Chemical composition of steel
Carbon content
Other compositions
C eq - C+Mn/6+(Cr+Mo+V)/5 + (Cu +Ni)/15
Welding procedures
Diffusible hydrogen
Susceptible microstructure
Tensile stress
cracking
Cracking mechanism
dissociation
super saturation
localisation
recombination
rupture
Hydrogen limited solubility in steel
solubility is high in liquid state
Upto the solubility limits present in
interstial solution
Beyond the solubility limit, retained in sites
called traps.
Localisation of hydrogen takes place
Adsorption of hydrogen reduces the
surface energy required for crack initiation
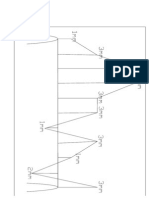
Temp C
L
o
g
H
i
n
c
m
3
/
1
0
0
g
o
f
f
e
a
t
S
T
P
Liquid Fe
Gamma Fe
Alpha Fe
1600 600 400 1000
Effect of temperature on solubility of hydrogen
1.2
1.6
0.8
0.4
0.0
0.2
Sources of hydrogen
Flux coating material in electrodes & SAW
welding Flux
Consumables exposed to atmosphere
Moisture in shielding gas
No/ improper baking of weld consumables
Hydrocarbons in base metal
Rusted consumable / base metal
Effect of hydrogen and stress on
damage caused by HIC
8 4 12 16 20 24
Time (h)
S
t
r
e
s
s
M
p
a
)
2 ppm
4 ppm
8ppm
Fig. Effect of hydrogen on the critical stress and the time
needed for HIC
Effect of preheat temp on rupture stress
Composition of steel
Processing route
Composition of weld metal
Welding conditions
Microstructure
Tensile stress
Restraint
Thickness
Improper fit up
Rapid cooling
To avoid hydrogen
cracking
Controlling diffusible hydrogen
content
basic coated Low hydrogen welding
electrodes
Low hydrogen welding process
Preheating
Post heating
Baking of electrodes
prior to welding
Rutile electrode 250 C
Basic electrode 350 C
Dont keep the
electrodes in open
atmosphere
keep the electrodes
in hand quivers
Preheat
Carbon equivalent
Thickness of the base metal
Diffusible hydrogen content
Restraint
Welding process, consumable &
conditions
Preheat
No welding on the base metal covered with ice
No welding shall be done at or below - 18 deg C
Raise the base metal temperature to min. 16 C
when the ambient temp is 0 to - 18 deg c
Preheat temperature shall be measured at a
distance of 2t or 4 which is higher
Preheat temperature shall be maintained during
welding
Solidification
cracking
Welding Research Institute
BHEL, Tiruchirappalli - 14
S. MANIMOZHI,
Dr R. NAGALAKSHMI &
Dr V. P. RAGHUPATHY
EFFECT OF HYDROGEN
ON
LAMELLAR TEARING
Lamellar Tearing
Lamellar
Tear Fillet Weld
Tee Joint Corner Joint
Causes
High tensile stresses parallel to weld
Stringer type inclusions in base plate
Presence of Diffusible Hydrogen
Lamellar Tear
Step like crack
in HAZ
Lamellar tearing
Lamellar Tearing - a Case study
Lamellar tearing in flange plate & root
crack in butt weld weakened the member
Poor profile of fillet reduced fatigue
strength
Failure of Alexander Kielland
Oil Drilling Platform on 27- 3-1980
in North Sea killed 123 of 213
people on board
Failure traced to cracking of 6 mm
length weld
(to hold a sonar device)
Lamellar
tear
Root
crack
Methods to avoid Lamellar Tearing
Joint configuration (Tee Joint)
Full Penetration
weld
X
Partial
Penetration weld
Buttering
layer
Use steel with guaranteed
Z thickness properties
- cross rolling
- calcium treatment
- inclusion shape control
Methods to avoid Lamellar Tearing
Change location & joint design
to reduce restraint
Reduce available hydrogen
Butter the surface with low H
2
Electrodes
Use higher pre-heat & interpass temperatures
Peen the welds
Substitute Forgings or Castings for the plates
Use steels with adequate Z thickness
properties
Poor Short Transverse ductility
Lamellar Tearing
Material
anisotropy
Restraint
stress
Joint Design
Welding Procedure
Joint thickness
Inclusions
Depends on
Type of steel
Deoxidation practice
Composition
Position of ingot
Inclusion content in steel
Inclusions
Type of inclusions
Sulphides
Oxides
Oxy sulphides
Silicates
Inclusion size & shape
Globular
Lamellar
Distribution of inclusion
Volume fraction
Mean spacing between inclusion
Grading of steel against Lamellar Tearing
Quality Recommend values for %
R.A
Grade
Mean Min.
Low LTR < 10 15 10
Medium LTR 20 25 15
LTR > 30 (high) 35 25
Test Methods
Z- direction impact test
L- direction notched tensile test
Z- Window type test
Z- direction tensile test
- Adopted for less restraint conditions
- Fully killed steels exhibit better correlation
- Semi killed steels show poor correlation
Z - Direction Charpy test
L- direction notched tensile test
Correlation not good
Z- Window type test
Severe Restraint conditions
Better correlation to incidence to LT
Section XX
X
X
Z- direction tensile test
% Elongation and percent reduction in area are
the parameters to evaluate the sensitivity to LT
Z
Tensile strength
in L & Z direction
do not have much
variation
Ductility in Z
direction inferior
to L- Direction
Experimental Plan
Base material API 5L
Design and development of experimental
electrodes to yield different levels of
hydrogen in weld metal
Determine diffusible hydrogen level as
per ISO 3690
Welding of cruciform joint
No preheat
No post heat
Specimen extraction
Tensile testing
Welding & Extraction of
Z direction specimen
Plate - 6 x 100 x 200mm
Material API -5L X 46
Welding process - SMAW
Electrode - E 25, E 35 & E45
Electrode dia. 3.15 mm
I = 90 to 100 A
Cruciform joint
Full penetration weld
Diffusible hydrogen level
Electrode
Hydrogen level
(ml/100g)
E 25 16.3
E 35 10.1
E 45 8.8
E 25, E35 & E45 corresponds to high
hydrogen, medium & low hydrogen
Measurements as per ISO 3690
Chemical composition of base metal &
All weld deposit of experimental electrode
(Wt.%)
Element Base Electrode Electrode Electrode
Metal E 25 E35 E45
C 0.33 0.05 0.05 0.06
Mn 0.91 1.30 1.28 1.25
Si < 0.01 0.28 0.23 0.30
S 0.016 0.025 0.021 0.023
P 0.018 0.010 0.010 0.008
Nb < 0.02 - - -
Ti < 0.05 - - -
V < 0.02 - - -
Tensile properties of base material
Base metal YS TS % E % RA
(Kg/mm
2
)
Transverse 42.5 50.3 28 42
direction
Long. 48.0 53.4 25 41
direction
All weld mechanical properties
experimental electrodes
Base metal YS TS % E % RA
(Kg/mm
2
)
E 25 42.3 52.8 28 66
E 35 43.5 58.0 29 66
E 45 42.8 53.4 28 64
Z-direction tensile test results - Electrode E25
% RA % Elongation UTS (Kg/mm
2
) Sl No.
11.4 14.0 44.1 1.
18.0 17.0 46.0 2.
18.6 17.0 46.8 3.
17.4 23.0 46.4 4
15.4 10.0 45.4 6.
11.6 15.0 49.4 5.
18.0 18.0 46.1 7.
13.0 20.0 45.3 8.
10.0 20.0 45.3 9.
22.1 13.0 45.3 10.
Z-Direction tensile test results - Electrode E35
% RA % Elongation UTS (Kg/mm
2
) Sl No.
19.0 20.0 40.8 1.
22.6 25.0 42.8 2.
28.9 21.0 42.1 3.
22.6 23.0 41.8 4
29.4 22.0 42.8 6.
15.4 12.0 43.3 5
17.8 19.0 43.3 7.
26.0 20.0 44.8 8.
26.0 21.0 44.3 9
19.0 15.0 34.1 10
Z-direction tensile test results - Electrode E45
% RA % Elongation UTS (Kg/mm
2
) Sl No.
16.0 22.0 50.1 1.
18.2 22.0 49.5 2.
16.0 20.0 49.0 3.
19.9 22.0 49.1 4
18.1 25.0 45.1 6.
19.6 22.0 44.6 5.
14.8 17.6 41.6 7.
19.8 24.0 46.0 8.
22.0 23.0 48.8 9.
21.0 23.0 47.8 10.
Inclusion - microstructure
Elongated inclusions
Mag. 100 X
Fractographs of broken specimens
fish eye, decohesion
E 25
E 35
E 45
Dimple fracture - inclusion
also seen
Dimple fracture -low level
inclusion
Correlation between H
2
& Ductility
%
E
l
o
n
g
a
t
i
o
n
a
n
d
R
A
log
e
[H]
15
17
19
21
23
25
Correlation between H
2
& % E is excellent
Correlation between H
2
& % E
Correlation between H
2
& % RA
2.0
2.2 2.4 2.6 2.8
% E = - 8.13 log
e
[H] + 39.238
CONCLUSION
Short transverse tensile tests were
carried out on API 5L material after
welding with three levels of hydrogen
The tensile ductility in the Short
transverse direction decreased with
the increased hydrogen level
CONCLUSION
The elongation in Z-direction
decreased as the hydrogen level
increased
The correlation between the Z-direction
elongation and hydrogen is very good than
the % reduction in area
No correlation between the tensile strength
and hydrogen was observed
CONCLUSION
Vous aimerez peut-être aussi
- Cover Letter Inspection EngineerDocument1 pageCover Letter Inspection EngineerTina MillerPas encore d'évaluation
- 5.6 Management Review: 5.6.1 GeneralDocument1 page5.6 Management Review: 5.6.1 GeneralTina MillerPas encore d'évaluation
- Coating SpecificationDocument14 pagesCoating SpecificationTina MillerPas encore d'évaluation
- Local Purchase Order: Amount in WordsDocument1 pageLocal Purchase Order: Amount in WordsTina MillerPas encore d'évaluation
- Trade LicenseDocument1 pageTrade LicenseTina MillerPas encore d'évaluation
- Specimen Details: A 150mm B 300mm For WPS and 150 MM For WQTDocument1 pageSpecimen Details: A 150mm B 300mm For WPS and 150 MM For WQTTina MillerPas encore d'évaluation
- Agenda of The PIMDocument1 pageAgenda of The PIMTina MillerPas encore d'évaluation
- PV Elite WebinarDocument24 pagesPV Elite WebinarAndrea Hank LattanzioPas encore d'évaluation
- Cover Letter Inspection EngineerDocument1 pageCover Letter Inspection EngineerTina MillerPas encore d'évaluation
- T-101-Roof Hand Railing Post-CorrosionDocument1 pageT-101-Roof Hand Railing Post-CorrosionTina MillerPas encore d'évaluation
- 2.0 Reference Standard/Code:: 3.0 Detailed Inspection ActivitiesDocument3 pages2.0 Reference Standard/Code:: 3.0 Detailed Inspection ActivitiesTina MillerPas encore d'évaluation
- Steam Trap InspectionDocument8 pagesSteam Trap InspectionMurrali Raj JeyagapalPas encore d'évaluation
- Engineering Calculation: 6.1cylindrical ShellDocument1 pageEngineering Calculation: 6.1cylindrical ShellTina MillerPas encore d'évaluation
- Plant Inspection-API InspectionDocument5 pagesPlant Inspection-API InspectionTina Miller100% (1)
- Nomenclature and Types of Heat ExchangersDocument1 pageNomenclature and Types of Heat ExchangersTina MillerPas encore d'évaluation
- Agenda of The PIMDocument1 pageAgenda of The PIMTina MillerPas encore d'évaluation
- Agenda of The PIMDocument1 pageAgenda of The PIMTina MillerPas encore d'évaluation
- Weld DefectsDocument78 pagesWeld DefectsjobsjamesPas encore d'évaluation
- Welding ErrorsDocument42 pagesWelding ErrorsTina Miller100% (1)
- DiscontinuitiesDocument78 pagesDiscontinuitiesTina Miller100% (1)
- Check List For Receiving Inspection of Manual Valves: Date-04/06/2014 QCIC/027 Rev-0Document1 pageCheck List For Receiving Inspection of Manual Valves: Date-04/06/2014 QCIC/027 Rev-0Tina MillerPas encore d'évaluation
- Low Hydrogen Electrodes To A5Document2 pagesLow Hydrogen Electrodes To A5Tina MillerPas encore d'évaluation
- Check List GalvanisingDocument1 pageCheck List GalvanisingTina MillerPas encore d'évaluation
- Basic MetallurgyDocument55 pagesBasic MetallurgyTina Miller100% (2)
- Low Hydrogen Electrodes To A5Document2 pagesLow Hydrogen Electrodes To A5Tina MillerPas encore d'évaluation
- Vertical Position ModelDocument1 pageVertical Position ModelTina MillerPas encore d'évaluation
- Amorphous Metal - Wikipedia, The Free EncyclopediaDocument5 pagesAmorphous Metal - Wikipedia, The Free EncyclopediaTina MillerPas encore d'évaluation
- Tank InspectionDocument7 pagesTank InspectionTina Miller100% (1)
- Implementation of ISO:IEC 17020-2012Document14 pagesImplementation of ISO:IEC 17020-2012Kin Kwan100% (3)
- The Subtle Art of Not Giving a F*ck: A Counterintuitive Approach to Living a Good LifeD'EverandThe Subtle Art of Not Giving a F*ck: A Counterintuitive Approach to Living a Good LifeÉvaluation : 4 sur 5 étoiles4/5 (5794)
- Shoe Dog: A Memoir by the Creator of NikeD'EverandShoe Dog: A Memoir by the Creator of NikeÉvaluation : 4.5 sur 5 étoiles4.5/5 (537)
- The Yellow House: A Memoir (2019 National Book Award Winner)D'EverandThe Yellow House: A Memoir (2019 National Book Award Winner)Évaluation : 4 sur 5 étoiles4/5 (98)
- Hidden Figures: The American Dream and the Untold Story of the Black Women Mathematicians Who Helped Win the Space RaceD'EverandHidden Figures: The American Dream and the Untold Story of the Black Women Mathematicians Who Helped Win the Space RaceÉvaluation : 4 sur 5 étoiles4/5 (895)
- The Hard Thing About Hard Things: Building a Business When There Are No Easy AnswersD'EverandThe Hard Thing About Hard Things: Building a Business When There Are No Easy AnswersÉvaluation : 4.5 sur 5 étoiles4.5/5 (344)
- The Little Book of Hygge: Danish Secrets to Happy LivingD'EverandThe Little Book of Hygge: Danish Secrets to Happy LivingÉvaluation : 3.5 sur 5 étoiles3.5/5 (399)
- Grit: The Power of Passion and PerseveranceD'EverandGrit: The Power of Passion and PerseveranceÉvaluation : 4 sur 5 étoiles4/5 (588)
- The Emperor of All Maladies: A Biography of CancerD'EverandThe Emperor of All Maladies: A Biography of CancerÉvaluation : 4.5 sur 5 étoiles4.5/5 (271)
- Devil in the Grove: Thurgood Marshall, the Groveland Boys, and the Dawn of a New AmericaD'EverandDevil in the Grove: Thurgood Marshall, the Groveland Boys, and the Dawn of a New AmericaÉvaluation : 4.5 sur 5 étoiles4.5/5 (266)
- Never Split the Difference: Negotiating As If Your Life Depended On ItD'EverandNever Split the Difference: Negotiating As If Your Life Depended On ItÉvaluation : 4.5 sur 5 étoiles4.5/5 (838)
- A Heartbreaking Work Of Staggering Genius: A Memoir Based on a True StoryD'EverandA Heartbreaking Work Of Staggering Genius: A Memoir Based on a True StoryÉvaluation : 3.5 sur 5 étoiles3.5/5 (231)
- On Fire: The (Burning) Case for a Green New DealD'EverandOn Fire: The (Burning) Case for a Green New DealÉvaluation : 4 sur 5 étoiles4/5 (73)
- Elon Musk: Tesla, SpaceX, and the Quest for a Fantastic FutureD'EverandElon Musk: Tesla, SpaceX, and the Quest for a Fantastic FutureÉvaluation : 4.5 sur 5 étoiles4.5/5 (474)
- Team of Rivals: The Political Genius of Abraham LincolnD'EverandTeam of Rivals: The Political Genius of Abraham LincolnÉvaluation : 4.5 sur 5 étoiles4.5/5 (234)
- The World Is Flat 3.0: A Brief History of the Twenty-first CenturyD'EverandThe World Is Flat 3.0: A Brief History of the Twenty-first CenturyÉvaluation : 3.5 sur 5 étoiles3.5/5 (2259)
- The Unwinding: An Inner History of the New AmericaD'EverandThe Unwinding: An Inner History of the New AmericaÉvaluation : 4 sur 5 étoiles4/5 (45)
- The Gifts of Imperfection: Let Go of Who You Think You're Supposed to Be and Embrace Who You AreD'EverandThe Gifts of Imperfection: Let Go of Who You Think You're Supposed to Be and Embrace Who You AreÉvaluation : 4 sur 5 étoiles4/5 (1090)
- The Sympathizer: A Novel (Pulitzer Prize for Fiction)D'EverandThe Sympathizer: A Novel (Pulitzer Prize for Fiction)Évaluation : 4.5 sur 5 étoiles4.5/5 (120)
- Her Body and Other Parties: StoriesD'EverandHer Body and Other Parties: StoriesÉvaluation : 4 sur 5 étoiles4/5 (821)
- Volume-7. Essential Knowledge Required For Design and Manufacturing of Hydraulic PressesDocument62 pagesVolume-7. Essential Knowledge Required For Design and Manufacturing of Hydraulic PressesQ.S. Khan100% (5)
- Ocr As Level Chemistry A: Answer All Questions Max 77 MarksDocument14 pagesOcr As Level Chemistry A: Answer All Questions Max 77 MarksdhruviniPas encore d'évaluation
- Engineering MaterialsDocument16 pagesEngineering MaterialsYosef Ganang Jati NugrohoPas encore d'évaluation
- ASTM Volume 02.05, May 2013 Metallic and Inorganic Coatings Metal Powders and Metal Powder ProductsDocument5 pagesASTM Volume 02.05, May 2013 Metallic and Inorganic Coatings Metal Powders and Metal Powder ProductsCicero Gomes de SouzaPas encore d'évaluation
- Samson Eco HopperDocument8 pagesSamson Eco HopperMauricio MpintoPas encore d'évaluation
- Conversion of Ore Into It's OxideDocument21 pagesConversion of Ore Into It's OxideGeeta BhattPas encore d'évaluation
- Section - 08 11 16 - Aluminum Doors and FramesDocument2 pagesSection - 08 11 16 - Aluminum Doors and FramesEliseo LagmanPas encore d'évaluation
- 10 11648 J Ijmpem 20190402 11Document8 pages10 11648 J Ijmpem 20190402 11ShantyPas encore d'évaluation
- V 082 N 04 P 112Document6 pagesV 082 N 04 P 112Romi FadliPas encore d'évaluation
- GalvInfoNote 1 4Document4 pagesGalvInfoNote 1 4rplata63Pas encore d'évaluation
- Ferro AlloysDocument13 pagesFerro Alloysvjkumar7378397Pas encore d'évaluation
- Float Collar Rev1Document3 pagesFloat Collar Rev1Dilip ReddyPas encore d'évaluation
- Synopsis Rfcs Projects 2015-17Document158 pagesSynopsis Rfcs Projects 2015-17znrobson internetPas encore d'évaluation
- APPDocument25 pagesAPPDiego MarquinaPas encore d'évaluation
- Catalog-Reductoare-Manuale PTR Vane PlaneDocument59 pagesCatalog-Reductoare-Manuale PTR Vane PlanezenogredinaPas encore d'évaluation
- Asme Ix QW PDFDocument183 pagesAsme Ix QW PDFAnonymous 4e7GNjzGW100% (4)
- Casting ProcessDocument17 pagesCasting ProcessThulasi Doss100% (1)
- Material Safety Data Sheet: Welding Alloys USA IncDocument4 pagesMaterial Safety Data Sheet: Welding Alloys USA IncShenny AldanaPas encore d'évaluation
- Industrial Training ReportDocument19 pagesIndustrial Training ReportRam PandeyPas encore d'évaluation
- A 732/a732m-02Document8 pagesA 732/a732m-02mutluPas encore d'évaluation
- Building Materials of Ancient IndiaDocument118 pagesBuilding Materials of Ancient IndiaNihakriPas encore d'évaluation
- CPD 25 - Perforated Metal Facades - Magazine Features - BuildingDocument6 pagesCPD 25 - Perforated Metal Facades - Magazine Features - BuildingTommy YapPas encore d'évaluation
- MCDR 2017 PDFDocument142 pagesMCDR 2017 PDFANKURPas encore d'évaluation
- Acids and Bases Review MCQDocument2 pagesAcids and Bases Review MCQAnna DixonPas encore d'évaluation
- Nano Ceramic Coating FormulationDocument16 pagesNano Ceramic Coating FormulationBobby AndreasPas encore d'évaluation
- C.25LAS01.026.01 Sesi 6 - Pengelasan SMAW (2 JP)Document38 pagesC.25LAS01.026.01 Sesi 6 - Pengelasan SMAW (2 JP)vnrfjrnPas encore d'évaluation
- 2022 Science Stage 9 - Paper 2Document18 pages2022 Science Stage 9 - Paper 2KomronbekPas encore d'évaluation
- Bis StandardsDocument5 pagesBis StandardsSaurabh GuptaPas encore d'évaluation
- SOM-BMT Lab Manual FinalDocument48 pagesSOM-BMT Lab Manual FinalHarish T S Gowda100% (1)
- TYPD ExercisesDocument10 pagesTYPD ExercisesConstance Lynn'da GPas encore d'évaluation